Abstract
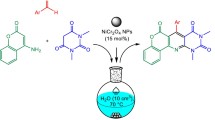
Benzimidazole derivatives have attracted a significant attention in recent years because of their medicinal applications as antiviral, antiulcer, antifungal, antihypertensive, anticancer, and antihistamine compounds. The one-pot synthesis of benzimidazole derivatives via oxidative condensation of aromatic aldehydes with o-phenylenediamines under mild conditions was successfully accomplished using a cobalt(II) supported on mesoporous silica-type material. The supported cobalt catalyst could be easily recovered after reaction completion and reused seven times with an excellent durability and without any noticeable loss in activity.
Graphical Abstract




Similar content being viewed by others
References
O’Sullivan DG, Wallis AK (1972) J Med Chem 15:103
Lohray BB, Lohray VB, Guntupalli P, Kommireddi NR, Mamoor PK, Ramanujam R (2000) US Patent No. 6051570
El-Masry AH, Fahmy HH, Abdelwahed SHA (2000) Molecules 5:1429
Kohara Y, Kubo K, Imamiya E, Wada T, Inada Y, Naka T (1996) J Med Chem 39:5228
Hanan H, Refaat M (2010) Eur J Med Chem 45:2949
Orjales A, Rubio V, Bordell M (1999) US Patent No. 5877187
Hasegawa E, Yoneoka A, Suzuki K, Kato T, Kitazume T, Yanagi K (1999) Tetrahedron 55:12957
Bai Y, Lu J, Shi Z, Yang B (2001) Synlett 12:544
Middleton RW, Wibberley DG (1980) J Heterocycl Chem 17:1757
Czarny A, Wilson WD, Boykin DW (1996) J Heterocycl Chem 33:1393
Sharghi H, Asemani O, Khalifeh R (2008) Synth Commun 38:1128
Ruiz VR, Corma A, Sabater MJ (2010) Tetrahedron 66:730
Parghi KD, Jayaram RV (2010) Catal Commun 11:1205
Raghavendra GM, Ramesha AB, Revanna CN, Nandeesh KN, Mantelingu K, Rangappa KS (2011) Tetrahedron Lett 52:5571
Chari MA, Shobha D, Kenawy E-R, Al-Deyab SS, Reddy BVS, Vinu A (2010) Tetrahedron Lett 51:5195
Eren B, Erdogan G (2012) React Kinet Mech Cat 107:333
Chari MA, Shobha D, Sasaki T (2011) Tetrahedron Lett 52:5575
Alinezhad H, Salehian F, Biparva P (2012) Synth Commun 42:102
Inamdar SM, More VK, Mandal SK (2013) Tetrahedron Lett 54:579
Jafarpour M, Rezaeifard A, Ghahramaninezhad M, Tabibi T (2013) New J Chem 37:2087
Heravi MM, Sadjadi S, Oskooie HA, Shoar RH, Bamoharram FF (2008) Catal Commun 9:504
Fazaeli R, Aliyan H (2009) Appl Catal A: Gen 353:74
Rafiee E, Rahpeima N, Eavani S (2014) Acta Chim Slov 61:177
Khanday WA, Tomar R (2014) Catal Commun 43:141
Das SS, Konwar D (2009) Synth Commun 39:980
Hao L, Zhao Y, Yu B, Zhang H, Xu H, Liu Z (2014) Green Chem 16:3039
Dutta A, Mondal J, Patra AK, Bhaumik A (2012) Chem Eur J 18:13372
Rajabi F, Luque R, Clark JH, Karimi B, Macquarrie DJ (2011) Catal Commun 12:510
Rajabi F, Raessi M, Arancon RAD, Saidi MR, Luque R (2015) Catal Commun 59:122
Acknowledgments
FR is grateful to Payame Noor University for support of this work. SD gratefully acknowledges University Grants Commission (UGC), India for the financial support during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajabi, F., De, S. & Luque, R. An Efficient and Green Synthesis of Benzimidazole Derivatives Using SBA-15 Supported Cobalt Nanocatalysts. Catal Lett 145, 1566–1570 (2015). https://doi.org/10.1007/s10562-015-1546-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-015-1546-z