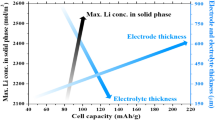
Lithium-ion batteries are considered as a promising energy source due to the high energy density and long cycle life. In this paper, the authors propose a model of a lithium-ion battery capacity fade and the electrochemical reactions in the battery. To improve the properties of the electrolyte and the lithium/electrolyte interface, the additive tris-(2,2,2-trifluoroethyl)-borate (TTFEB) is added into the carbonatebased electrolyte. The authors also study the factors influencing the battery capacity, including the resistance of solid electrolyte interface (SEI), the active material particle radius, the discharging rate, and the ambient temperature. The simulation results show that the lithium-ion battery life is determined by the capacity decay rate, the electrochemical mechanism, and the physicochemical parameters.











Similar content being viewed by others
References
R. U. Khalid, S. Seuring, and R. Wagner, “Evaluating supply chain constructs in the base of the pyramid environment,” J. Clean. Prod., 207,122415 (2020).
I. A. Hunt, Y. Patel, M. Szczygielski, L. Kabacik, and G. J. Offer, “Lithium-sulfur battery nail penetration test under load,” J. Energy Stor., 2, 25-29 (2015).
J. Yi and H. Han, “Analysis of factors affecting response for mixed potential gas sensors,” Electrochim. Acta, 379,138129 (2021).
Z. Chen, Q. Zhang, and J. Lu, “Optimization-based method to develop practical driving cycle for application in electric vehicle power management: A case study in Shenyang, China,” Energy, 186(C),115766.1-115766.13 (2019).
F. Shu, Z. Lu, J. Lin, et al., “Alternating iterative secure structure between beamforming and power allocation for UAVaided directional modulation networks,” Phys. Comm., 33, 46-53 (2019).
Z. Wang, S. Sankarasubramanian, J. Willey, H. Feng, and V. Ramani, “Engineering block co-polymer anion exchange membrane domains for highly efficient electrode-decoupled redox flow batteries,” Sustain. Energy Fuels, 5(14), 3606-3616 (2021).
V. Kumar, C. Sachdeva, K. M. Waidha, S. Sharma, and B. Saha, “In vitro and in silico anti-plasmodial evaluation of newly synthesized β-carboline derivatives,” Chem. Select, 6(21), 5338-5342 (2021).
P. Ramadass, B. Haran, P. M. Gomadam, R. White, and B. N. Popov, “Development of first principles capacity fade model for Li-ion cells,” J. Electrochem. Soc.,151(2), A196–A203 (2014).
W. S. Jung, W. H. Lee, H. S. Oh, et al., “Highly stable and ordered intermetallic PtCo alloy catalyst supported on graphitized carbon containing Co@CN for oxygen reduction reaction,” J. Mater. Chem. A, 8,19833-19842 (2020).
G. Ning, R. E. White, and B. N. Popov, “A generalized cycle life model of rechargeable Li-ion batteries,” Electrochim. Acta, 51(10), 2012-2022 (2006).
W. He, N. Williard, C. Chen, and M. Pecht, “State of charge estimation for electric vehicle batteries using unscented Kalman filtering,” Microelectron. Rel., 53(6), 840-847 (2013).
J. Guo, Z. Li, and M. Pecht, “A Bayesian approach for Li-ion battery capacity fade modeling and cycles to failure prognostics,” J. Pow. Sour., 281,173-184 (2015).
I. Bloom, B. W. Cole, J. J. Sohn, et al., “An accelerated calendar and cycle life study of Li-ion cells,” J. Pow. Sour., 101, 238-247 (2001).
J. Wang, P. Liu, J. Hicks-Garner, et al., “Cycle-life model for graphite-LiFePO4 cells,” J. Pow. Sour., 196, 3942-3948 (2011).
S. Saxena, C. Hendricks, and M. Pecht, “Cycle life testing and modeling of graphite/LiCoO2 cells under different state of charge ranges,” J. Pow. Sour., 327, 394-400 (2016).
M. Doyle and J. Newman, “The use of mathematical modeling in the design of lithium/polymer battery systems,” Electrochim. Acta, 40, 2191-2196 (1995).
T. F. Fuller, M. Doyle, and J. Newman, “Simulation and optimization of the dual lithium ion insertion cell,” J. Electrochem. Soc., 141,1-10 (1994).
W. Huo, H. He, and F. Sun, “Electrochemical-thermal modeling for a ternary lithium ion battery during discharging and driving cycle testing,” RSC Adv., 5, 57599-57607 (2015).
Acknowledgment
This work is supported by the Science and Technology Program of the Bei**g Municipal Education Commission (KM201911232023).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya i Tekhnologiya Topliv i Masel, No. 3, pp. 92–99 May–June, 2022.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, Z., Yang, L., Yang, Y. et al. Application of Electrochemical Model of a Lithium-Ion Battery. Chem Technol Fuels Oils 58, 519–529 (2022). https://doi.org/10.1007/s10553-022-01416-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10553-022-01416-1