Abstract
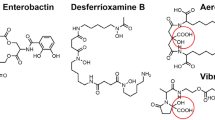
Acyl peptidic siderophores are produced by a variety of bacteria and possess unique amphiphilic properties. Amphiphilic siderophores are generally produced in a suite where the iron(III)-binding headgroup remains constant while the fatty acid appendage varies by length and functionality. Acyl peptidic siderophores are commonly synthesized by non-ribosomal peptide synthetases; however, the method of peptide acylation during biosynthesis can vary between siderophores. Following biosynthesis, acyl siderophores can be further modified enzymatically to produce a more hydrophilic compound, which retains its ferric chelating abilities as demonstrated by pyoverdine from Pseudomonas aeruginosa and the marinobactins from certain Marinobacter species. Siderophore hydrophobicity can also be altered through photolysis of the ferric complex of certain β-hydroxyaspartic acid-containing acyl peptidic siderophores.











Similar content being viewed by others
References
Barbeau K, Rue E, Bruland K, Butler A (2001) Photochemical cycling of iron in the surface ocean mediated by microbial iron(III)-binding ligands. Nature 413(6854):409–413. doi:10.1038/35096545
Barbeau K, Rue E, Trick C, Bruland K, Butler A (2003) Photochemical reactivity of siderophores produced by marine heterotrophic bacteria and cyanobacteria based on characteristic Fe(III) binding groups. Limnol Oceanogr 48(3):1069–1078
Bokhove M, Jimenez P, Quax W, Dijkstra B (2010) The quorum-quenching N-acyl homoserine lactone acylase PvdQ is an Ntn-hydrolase with an unusual substrate-binding pocket. Proc Natl Acad Sci USA 107(2):686–691. doi:10.1073/pnas.0911839107
Butler A, Theisen R (2010) Iron(III)-siderophore coordination chemistry: reactivity of marine siderophores. Coord Chem Rev 254(3–4):288–296. doi:10.1016/j.ccr.2009.09.010
Challis G, Ravel J, Townsend C (2000) Predictive, structure-based model of amino acid recognition by nonribosomal peptide synthetase adenylation domains. Chem Biol 7(3):211–224. doi:10.1016/S1074-5521(00)00091-0
Cisar J, Tan D (2008) Small molecule inhibition of microbial natural product biosynthesis—an emerging antibiotic strategy. Chem Soc Rev 37(7):1320–1329
Clevenger K, Wu R, Er J, Liu D, Fast W (2013) Rational design of a transition state analogue with picomolar affinity for Pseudomonas aeruginosa PvdQ, a siderophore biosynthetic enzyme. ACS Chem Biol 8(10):2192–2200. doi:10.1021/cb400345h
Crosa J, Walsh C (2002) Genetics and assembly line enzymology of siderophore biosynthesis in bacteria. Microbiol Mol Biol Rev 66(2):223–249. doi:10.1128/MMBR.66.2.223-249.2002
Drake E, Gulick A (2011) Structural characterization and high-throughput screening of inhibitors of PvdQ, an NTN hydrolase involved in pyoverdine synthesis. ACS Chem Biol 6(11):1277–1286. doi:10.1021/cb2002973
Engelhart C, Aldrich C (2013) Synthesis of chromone, quinolone, and benzoxazinone sulfonamide nucleosides as conformationally constrained inhibitors of adenylating enzymes required for siderophore biosynthesis. J Org Chem 78(15):7470–7481
Ferreras J, Ryu J-S, Di Lello F, Tan D, Quadri L (2005) Small-molecule inhibition of siderophore biosynthesis in Mycobacterium tuberculosis and Yersinia pestis. Nat Chem Biol 1(1):29–32. doi:10.1038/nchembio706
Gauglitz J, Butler A (2013) Amino acid variability in the peptide composition of a suite of amphiphilic peptide siderophores from an open ocean Vibrio species. J Biol Inorg Chem 18(5):489–497. doi:10.1007/s00775-013-0995-3
Gauglitz J, Inishi A, Ito Y, Butler A (2014) Microbial tailoring of acyl peptidic siderophores. Biochemistry 53(16):2624–2631. doi:10.1021/bi500266x
Gehring A, Mori I, Walsh C (1998) Reconstitution and characterization of the Escherichia coli enterobactin synthetase from EntB, EntE, and EntF. Biochemistry 37(8):2648–2659. doi:10.1021/bi9726584
Gobin J, Horwitz M (1996) Exochelins of Mycobacterium tuberculosis remove iron from human iron-binding proteins and donate iron to mycobactins in the M. tuberculosis cell wall. J Exp Med 183(4):1527–1532. doi:10.1084/jem.183.4.1527
Gobin J, Moore C, Reeve J, Wong D, Gibson B, Horwitz M (1995) Iron acquisition by Mycobacterium tuberculosis: isolation and characterization of a family of iron-binding exochelins. Proc Natl Acad Sci USA 92(11):5189–5193. doi:10.1073/pnas.92.11.5189
Grunewald J, Marahiel M (2006) Chemoenzymatic and template-directed synthesis of bioactive macrocyclic peptides. Microbiol Mol Biol Rev 70(1):121–146. doi:10.1128/MMBR.70.1.121-146.2006
Guillon L, El Mecherki M, Altenburger S, Graumann P, Schalk I (2012) High cellular organization of pyoverdine biosynthesis in Pseudomonas aeruginosa: clustering of PvdA at the old cell pole. Environ Microbiol 14(8):1982–1994. doi:10.1111/j.1462-2920.2012.02741.x
Hannauer M, Schafer M, Hoegy F, Gizzi P, Wehrung P, Mislin G, Budzikiewicz H, Schalk I (2012) Biosynthesis of the pyoverdine siderophore of Pseudomonas aeruginosa involves precursors with a myristic or a myristoleic acid chain. FEBS Lett 586(1):96–101. doi:10.1016/j.febslet.2011.12.004
Homann V, Sandy M, Tincu J, Templeton A, Tebo B, Butler A (2009) Loihichelins A–F, a suite of amphiphilic siderophores produced by the marine bacterium Halomonas LOB-5. J Nat Prod 72(5):884–888. doi:10.1021/np800640h
Huang J, Han J, Zhang L, Leadbetter J (2003) Utilization of acyl-homoserine lactone quorum signals for growth by a soil pseudomonad and Pseudomonas aeruginosa PAO1. Appl Environ Microbiol 69(10):5941–5949. doi:10.1128/AEM.69.10.5941-5949.2003
Ito Y, Butler A (2005) Structure of synechobactins, new siderophores of the marine cyanobacterium Synechococcus sp. PCC 7002. Limnol Oceanogr 50(6):1918–1923
Jimenez P, Koch G, Papaioannou E, Wahjudi M, Krzeslak J, Coenye T, Cool R, Quax W (2010) Role of PvdQ in Pseudomonas aeruginosa virulence under iron-limiting conditions. Microbiol SGM 156:49–59. doi:10.1099/mic.0.030973-0
Kem M, Zane H, Springer S, Gauglitz J, Butler A (2014) Amphiphilic siderophore production by oil-associating microbes. Metallomics 6(6):1150–1155. doi:10.1039/c4mt00047a
Kem M, Naka H, Iinishi A, Haygood M, Butler A (2015) Fatty acid hydrolysis of acyl marinobactin siderophores by Marinobacter acylases. ACS Biochem. doi:10.1021/bi5013673
Koch G, Jimenez P, Muntendam R, Chen Y, Papaioannou E, Heeb S, Camara M, Williams P, Cool R, Quax W (2010) The acylase PvdQ has a conserved function among fluorescent Pseudomonas spp. Environ Microbiol Rep 2(3):433–439. doi:10.1111/j.1758-2229.2010.00157.x
Koch G, Nadal-Jimenez P, Reis C, Muntendam R, Bokhove M, Melillo E, Dijkstra B, Cool R, Quax W (2014) Reducing virulence of the human pathogen Burkholderia by altering the substrate specificity of the quorum-quenching acylase PvdQ. Proc Natl Acad Sci USA 111(4):1568–1573. doi:10.1073/pnas.1311263111
Kraas F, Helmetag V, Wittmann M, Strieker M, Marahiel M (2010) Functional dissection of surfactin synthetase initiation module reveals insights into the mechanism of lipoinitiation. Chem Biol 17(8):872–880. doi:10.1016/j.chembiol.2010.06.015
Kreutzer M, Nett M (2012) Genomics-driven discovery of taiwachelin, a lipopeptide siderophore from Cupriavidus taiwanensis. Org Biomol Chem 10(47):9338–9343. doi:10.1039/c2ob26296g
Kreutzer M, Kage H, Nett M (2012) Structure and biosynthetic assembly of cupriachelin, a photoreactive siderophore from the bioplastic producer Cupriavidus necator H16. J Am Chem Soc 134(11):5415–5422. doi:10.1021/ja300520z
Krithika R, Marathe U, Saxena P, Ansari M, Mohanty D, Gokhale R (2006) A genetic locus required for iron acquisition in Mycobacterium tuberculosis. Proc Natl Acad Sci USA 103(7):2069–2074. doi:10.1073/pnas.0507924103
Kupper F, Carrano C, Kuhn J, Butler A (2006) Photoreactivity of iron(III)–Aerobactin: photoproduct structure and iron(III) coordination. Inorg Chem 45(15):6028–6033. doi:10.1021/ic0604967
Lane S, Marshall P, Upton R, Ratledge C, Ewing M (1995) Novel extracellular mycobactins, the carboxymycobactins from Mycobacterium avium. Tetrahedron Lett 36(23):4129–4132. doi:10.1016/0040-4039(95)00676-4
Lin Y, Xu J, Hu J, Wang L, Ong S, Leadbetter J, Zhang L (2003) Acyl-homoserine lactone acylase from Ralstonia strain XJ12B represents a novel and potent class of quorum-quenching enzymes. Mol Microbiol 47(3):849–860. doi:10.1046/j.1365-2958.2003.03351.x
Luo M, Lin H, Fischbach M, Liu D, Walsh C, Groves J (2006) Enzymatic tailoring of enterobactin alters membrane partitioning and iron acquisition. ACS Chem Biol 1(1):29–32. doi:10.1021/cb0500034
Madigan C, Cheng T, Layre E, Young D, McConnell M, Debono C, Murry J, Wei J, Barry C, Rodriguez G, Matsunaga I, Rubin E, Moody D (2012) Lipidomic discovery of deoxysiderophores reveals a revised mycobactin biosynthesis pathway in Mycobacterium tuberculosis. Proc Natl Acad Sci USA 109(4):1257–1262. doi:10.1073/pnas.1109958109
Marahiel M, Stachelhaus T, Mootz H (1997) Modular peptide synthetases involved in nonribosomal peptide synthesis. Chem Rev 97(7):2651–2673. doi:10.1021/cr960029e
Martin J, Ito Y, Homann V, Haygood M, Butler A (2006) Structure and membrane affinity of new amphiphilic siderophores produced by Ochrobactrum sp. SP18. J Biol Inorg Chem 11(5):633–641. doi:10.1007/s00775-006-0112-y
Martinez J, Butler A (2007) Marine amphiphilic siderophores: marinobactin structure, uptake, and microbial partitioning. J Inorg Biochem 101(11–12):1692–1698. doi:10.1016/j.**orgbio.2007.07.007
Martinez J, Zhang G, Holt P, Jung H, Carrano C, Haygood M, Butler A (2000) Self-assembling amphiphilic siderophores from marine bacteria. Science 287(5456):1245–1247. doi:10.1126/science.287.5456.1245
McMahon M, Rush J, Thomas M (2012) Analyses of MbtB, MbtE, and MbtF suggest revisions to the mycobactin biosynthesis pathway in Mycobacterium tuberculosis. J Bacteriol 194(11):2809–2818. doi:10.1128/JB.00088-12
Neres J, Wilson D, Celia L, Beck B, Aldrich C (2008) Aryl acid adenylating enzymes involved in siderophore biosynthesis: fluorescence polarization assay, ligand specificity, and discovery of non-nucleoside inhibitors via high-throughput screening. Biochemistry 47(45):11735–11749
Quadri L, Sello J, Keating T, Weinreb P, Walsh C (1998) Identification of a Mycobacterium tuberculosis gene cluster encoding the biosynthetic enzymes for assembly of the virulence-conferring siderophore mycobactin. Chem Biol 5(11):631–645. doi:10.1016/S1074-5521(98)90291-5
Ratledge C (2004) Iron, mycobacteria and tuberculosis. Tuberculosis 84(1–2):110–130. doi:10.1016/j.tube.2003.08.012
Rosconi F, Davyt D, Martinez V, Martinez M, Abin-Carriquiry J, Zane H, Butler A, de Souza E, Fabiano E (2013) Identification and structural characterization of serobactins, a suite of lipopeptide siderophores produced by the grass endophyte Herbaspirillum seropedicae. Environ Microbiol 15(3):916–927. doi:10.1111/1462-2920.12075
Sandy M, Butler A (2009) Microbial iron acquisition: marine and terrestrial siderophores. Chem Rev 109(10):4580–4595. doi:10.1021/cr9002787
Schalk I, Guillon L (2013) Pyoverdine biosynthesis and secretion in Pseudomonas aeruginosa: implications for metal homeostasis. Environ Microbiol 15(6):1661–1673. doi:10.1111/1462-2920.12013
Shen B (2003) Polyketide biosynthesis beyond the type I, II and III polyketide synthase paradigms. Curr Opin Chem Biol 7(2):285–295. doi:10.1016/S1367-5931(03)00020-6
Sio C, Otten L, Cool R, Diggle S, Braun P, Bos R, Daykin M, Camara M, Williams P, Quax W (2006) Quorum quenching by an N-acyl-homoserine lactone acylase from Pseudomonas aeruginosa PAO1. Infect Immun 74(3):1673–1682. doi:10.1128/IAI.74.3.1673-1682.2006
Snow G (1965) Isolation and structure of mycobactin T, a growth factor from Mycobacterium tuberculosis. Biochem J 97(1):166–175
Somu R, Boshoff H, Qiao C, Bennett E, Barry C III, Aldrich C (2006) Rationally designed nucleoside antibiotics that inhibit siderophore biosynthesis of Mycobacterium tuberculosis. J Med Chem 49(1):31–34
Stachelhaus T, Mootz H, Marahiel M (1999) The specificity-conferring code of adenylation domains in nonribosomal peptide synthetases. Chem Biol 6(8):493–505. doi:10.1016/S1074-5521(99)80082-9
Staunton J, Weissman K (2001) Polyketide biosynthesis: a millennium review. Nat Prod Rep 18(4):380–416. doi:10.1039/a909079g
Vergnolle O, Xu H, Blanchard J (2013) Mechanism and regulation of mycobactin fatty acyl-AMP ligase FadD33. J Biol Chem 288(39):28116–28125. doi:10.1074/jbc.M113.495549
Visca P, Imperi F, Lamont I (2007) Pyoverdine siderophores: from biogenesis to biosignificance. Trends Microbiol 15(1):22–30. doi:10.1016/j.tim.2006.11.004
Vraspir J, Holt P, Butler A (2011) Identification of new members within suites of amphiphilic marine siderophores. Biometals 24(1):85–92. doi:10.1007/s10534-010-9378-1
Wahjudi M, Papaioannou E, Hendrawati O, van Assen A, van Merkerk R, Cool R, Poelarends G, Ouax W (2011) PA0305 of Pseudomonas aeruginosa is a quorum quenching acylhomoserine lactone acylase belonging to the Ntn hydrolase superfamily. Microbiol SGM 157:2042–2055. doi:10.1099/mic.0.043935-0
Walsh C, Chen H, Keating T, Hubbard B, Losey H, Luo L, Marshall C, Miller D, Patel H (2001) Tailoring enzymes that modify nonribosomal peptides during and after chain elongation on NRPS assembly lines. Curr Opin Chem Biol 5(5):525–534. doi:10.1016/S1367-5931(00)00235-0
Wurst J, Drake E, Theriault J, Jewett I, VerPlank L, Perez J, Dandapani S, Palmer M, Moskowitz S, Schreiber S, Munoz B, Gulick A (2014) Identification of inhibitors of PvdQ, an enzyme involved in the synthesis of the siderophore pyoverdine. ACS Chem Biol 9(7):1536–1544. doi:10.1021/cb5001586
Xu G, Martinez J, Groves J, Butler A (2002) Membrane affinity of the amphiphilic marinobactin siderophores. J Am Chem Soc 124(45):13408–13415. doi:10.1021/ja026768w
Yeterian E, Martin L, Guillon L, Journet L, Lamont I, Schalk I (2010) Synthesis of the siderophore pyoverdine in Pseudomonas aeruginosa involves a periplasmic maturation. Aminoacids 38(5):1447–1459. doi:10.1007/s00726-009-0358-0
Zane H, Naka H, Rosconi F, Sandy M, Haygood M, Butler A (2014) Biosynthesis of amphi-enterobactin siderophores by Vibrio harveyi BAA-1116: identification of a bifunctional nonribosomal peptide synthetase condensation domain. J Am Chem Soc 136(15):5615–5618. doi:10.1021/ja5019942
Acknowledgments
Funding from National Science Foundation Grant CHE-1411941 (A.B.) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kem, M.P., Butler, A. Acyl peptidic siderophores: structures, biosyntheses and post-assembly modifications. Biometals 28, 445–459 (2015). https://doi.org/10.1007/s10534-015-9827-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-015-9827-y