Abstract
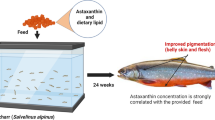
This study examined the effects of dietary esterified astaxanthin concentration on coloration, accumulation of carotenoids, and the composition of carotenoids over time in the skin of Amphiprion ocellaris. Juveniles of 30 days-post-hatch were fed 40, 60, 80, or 160 mg esterified astaxanthin per kg diet (mg kg−1) for 90 days. Skin coloration was analyzed using the hue, saturation, and luminosity model. Increased astaxanthin concentrations and duration on diet lead to improvements in skin color, that is, lower hues (~27–29 to ~14–17; redder fish), higher saturation (~77 to ~87 %), and lower luminosity (~43 to ~35 %). Fish fed 80 and 160 mg kg−1 astaxanthin feed showed significant coloration improvements over fish fed lower astaxanthin feeds. Increasing both dietary astaxanthin concentration and time on the feed resulted in significant increases in total skin carotenoid concentration (0.033–0.099 μg mm−2). Furthermore, there was a significant linear relationship between hue and total skin carotenoid concentration. Compositionally, free astaxanthin and 4-hydroxyzeaxanthin were the major skin carotenoids. 4-hydroxyzeaxanthin was previously unreported for A. ocellaris. Carotenoid composition was affected by duration on diet. Fraction 4-hydroxyzeaxanthin increased by ~15 %, while free astaxanthin decreased equivalently. The transition from 4-hydroxyzeaxanthin to free astaxanthin appears to follow a reductive pathway. Results suggest that managing coloration in the production of A. ocellaris juveniles requires manipulation of both dietary astaxanthin concentration and period of exposure to astaxanthin containing diet. In order to achieve more orange–red-colored fish, feeding 80–160 mg kg−1 esterified astaxanthin for an extended duration is recommended.






Similar content being viewed by others
References
Ako H, Tamaru CS, Asano L, Yuen B, Yamamoto M (2000) Achieving natural coloration in fish under culture. In: Tamaru CC-T, Tamaru CS, McVey J, Ikuta K (eds) Spawning and maturation of aquatic species, UJNR Technical Report No 28. University of Hawaii Sea Grant College Program, Hawaii
Andrews C (1990) The ornamental fish trade and fish conservation. J Fish Biol 37(sA):53–59
Avella MA, Olivotto I, Gioacchini G, Maradonna F, Carnevali O (2007) The role of fatty acids enrichments in the larviculture of false percula clownfish Amphiprion ocellaris. Aquaculture 273(1):87–95
Bjerkeng B, Hamre K, Hatlen B, Wathne E (1999) Astaxanthin deposition in fillets of Atlantic salmon Salmo salar L. fed two dietary levels of astaxanthin in combination with three levels of α-tocopheryl acetate. Aquacult Res 30(9):637–646
Blount J, Surai P, Nager R, Houston D, Møller A, Trewby M, Kennedy M (2002) Carotenoids and egg quality in the lesser blackbacked gull Larus fuscus: a supplemental feeding study of maternal effects. Proc R Soc B 269(1486):29–36
Booth MA, Warner-Smith RJ, Allan GL, Glencross BD (2004) Effects of dietary astaxanthin source and light manipulation on the skin colour of Australian snapper Pagrus auratus (Bloch & Schneider, 1801). Aquacult Res 35(5):458–464
Chatzifotis S, Pavlidis M, Jimeno C, Vardanis G, Sterioti A, Divanach P (2005) The effect of different carotenoid sources on skin coloration of cultured red porgy (Pagrus pagrus). Aquacult Res 36(15):1517–1525
Chen H-M, Meyers SP (1984) A rapid quantitative method for determination of astaxanthin pigment concentration in oil extracts. J Am Oil Chem Soc 61(6):1045–1047
Goodwin T (1984) The biochemistry of the carotenoids: Animals, vol 2, 2nd edn. Chapman and Hall, London
Goto S, Kogure K, Abe K, Kimata Y, Kitahama K, Yamashita E, Terada H (2001) Efficient radical trap** at the surface and inside the phospholipid membrane is responsible for highly potent antiperoxidative activity of the carotenoid astaxanthin. Biochim Biophys Acta 1512(2):251–258
Gouveia L, Rema P (2005) Effect of microalgal biomass concentration and temperature on ornamental goldfish (Carassius auratus) skin pigmentation. Aquacult Nutr 11(1):19–23
Gouveia L, Rema P, Pereira O, Empis J (2003) Colouring ornamental fish (Cyprinus carpio and Carassius auratus) with microalgal biomass. Aquacult Nutr 9(2):123–129
Hawkes JW (1974) The structure of fish skin II. The chromatophore unit. Cell Tiss Res 149(2):159–172
Hill G, Inouye C, Montgomerie R (2002) Dietary carotenoids predict plumage coloration in wild house finches. Proc R Soc B 269(1496):1119–1124
Hoff F (1996) Conditioning, spawning and rearing of fish with emphasis on marine clownfish. Aquaculture Consultants Inc, Dade City
Hudon J, Grether G, Millie D (2003) Marginal differentiation between the sexual and general carotenoid pigmentation of guppies (Poecilia reticulata) and a possible visual explanation. Physiol Biochem Zool 76(6):776–790
Kalinowski CT, Robaina LE, Fernández-Palacios H, Schuchardt D, Izquierdo MS (2005) Effect of different carotenoid sources and their dietary levels on red porgy (Pagrus pagrus) growth and skin colour. Aquaculture 244(1–4):223–231
Kitahara T (1983) Behavior of carotenoids in the chum salmon (Oncorhynchus keta) during anadromous migration. Comp Biochem Physiol B 76(1):97–101
Li H, Tyndale ST, Heath DD, Letcher RJ (2005) Determination of carotenoids and all-trans-retinol in fish eggs by liquid chromatography–electrospray ionization–tandem mass spectrometry. J Chromatogr B 816(1–2):49–56
Matsuno T (2001) Aquatic animal carotenoids. Fish Sci 67(5):771–783
McGraw KJ, Ardia DR (2003) Carotenoids, immunocompetence, and the information content of sexual colors: an experimental test. Am Nat 162(6):704–712
Miki W (1991) Biological functions and activities of animal carotenoids. Pure Appl Chem 63(1):141–146
Ohkubo M, Tsushima M, Maoka T, Matsuno T (1999) Carotenoids and their metabolism in the goldfish Carassius auratus (Hibuna). Comp Biochem Physiol B 124(3):333–340
Ostrander G (ed) (2000) Handbook of experimental animals: The laboratory fish, 1st edn. Academic Press, London
Palmer L (1915) Xanthophyll, the principal natural yellow pigment of the egg yolk, body fat, and blood serum of the hen. The physiological relation of the pigment to the xanthophylls of plants. J Biol Chem 23:261–279
Rhyne AL, Tlusty MF, Schofield PJ, Kaufman L, Morris JA Jr, Bruckner AW (2012) Revealing the appetite of the marine aquarium fish trade: the volume and biodiversity of fish imported into the United States. PLoS One 7(5):e35808
Schiedt K, Vecchi M, Glinz E, Storebakken T (1988) Metabolism of carotenoids in salmonids. Part 3. Metabolites of astaxanthin and canthaxanthin in the skin of Atlantic salmon (Salmo salar, L.). Helv Chim Acta 71(4):887–896
Tanaka Y, Yamamoto A, Kamata T, Simpson K (1992) Biochemical study on the carotenoids in the anemonefish, Amphiprion spp. Mem Fac Fish Kagoshima Univ 41:1–8
van Nieuwerburgh L, Wänstrand I, Liu J, Snoeijs P (2005) Astaxanthin production in marine pelagic copepods grazing on two different phytoplankton diets. J Sea Res 53(3):147–160
Wabnitz C, Taylor M, Green E, Razak T (2003) From ocean to aquarium. UNEP-WCMC, Cambridge
Wallat GK, Lazur AM, Chapman FA (2005) Carotenoids of different types and concentrations in commercial formulated fish diets affect color and its development in the skin of the red oranda variety of goldfish. N Am J Aquacult 67(1):42–51
Wathne E, Bjerkeng B, Storebakken T, Vassvik V, Odland AB (1998) Pigmentation of Atlantic salmon (Salmo salar) fed astaxanthin in all meals or in alternating meals. Aquaculture 159(3–4):217–231
White D, Page G, Swaile J, Moody A, Davies S (2002) Effect of esterification on the absorption of astaxanthin in rainbow trout, Oncorhynchus mykiss (Walbaum). Aquacult Res 33(5):343–350
Wittenrich ML (2007) The complete illustrated breeder’s guide to marine aquarium fishes. Publications, Neptune City, T.F.H
Wood E (2001) Collection of coral reef fish for aquaria: Global trade, conservation issues and management strategies. Marine Conservation Society, UK
Yasir I, Qin JG (2009a) Effect of light intensity on color performance of false clownfish, Amphiprion ocellaris Cuvier. J World Aqua Soc 40(3):337–350
Yasir I, Qin JG (2009b) Impact of background on color performance of false clownfish, Amphiprion ocellaris, Cuvier. J World Aqua Soc 40(6):724–734
Yasir I, Qin JG (2010) Effect of dietary carotenoids on skin color and pigments of false clownfish, Amphiprion ocellaris, Cuvier. J World Aqua Soc 41(3):308–318
Acknowledgments
This study was conducted by the first author as his undergraduate senior thesis to partially meet the requirements for his Honors degree at Roger Williams University. The authors would like to thank Bradford D. Bourque, Kevin G. Jackson, Amanda C. White, Stacey A. Helming, Tim L. Arcand, Eric J. Payne, and Andrew R. Porteus for various aspects of the grow-out phase and chemical analyses. The authors would also like to extend our gratitude toward David L. Taylor for his help with statistics, Nancy E. Breen for her help with the analytical process, and to Dale F. Leavitt for his help with diet analysis. The authors would like to thank Waters Corporation for their aid in the identification of the specific carotenoids, and also the anonymous reviewers who provided valuable comments on this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ho, A.L.F.C., O’Shea, S.K. & Pomeroy, H.F. Dietary esterified astaxanthin effects on color, carotenoid concentrations, and compositions of clown anemonefish, Amphiprion ocellaris, skin. Aquacult Int 21, 361–374 (2013). https://doi.org/10.1007/s10499-012-9558-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-012-9558-9