Abstract
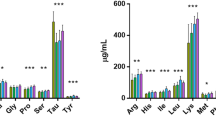
The experiment aimed at determining the efficient use of phytase (Phy) in Atlantic salmon diets that had low (4.5%) fishmeal and contained 60% soy protein concentrate (SPC). Phytase was either included at 250, 500, 1,000 or 4,000 U Phy kg−1 diet or the SPC was pre-treated prior to making diets using 250, 500 or 1,000 U Phy kg−1 SPC. Fish were fed the experimental diets for 12 weeks, and there were no differences in survival among treatments nor were there differences in growth performance between the phytase-pre-treated SPC diets. Feed intake and weight gain were significantly lower for diets supplemented below 1,000 U Phy kg−1 compared to all other diets. Apparent digestibility (AD) of phosphorus was significantly lower without the use of phytase (45.43 ± 2.06%) than for all other treatments. AD phosphorus increased from 55.70 ± 1.81% at the lowest phytase supplementation (250 U Phy kg−1) to 80.87 ± 2.12% at the highest (4,000 U Phy kg−1). There was no difference in AD phosphorus between the diet with the highest supplementation (4,000 U Phy kg−1) and the pre-treated diets. There were no differences in whole-body dry material, crude protein or total lipid, whereas bone ash was significantly lower for diets supplemented below 1,000 U Phy kg−1. Ash and phosphorus in the whole body and bone increased with increasing added phytase. At and above an inclusion of 1,000 U Phy kg−1, bone ash (51.26 ± 0.12% bone weight) and bone phosphorus (11.21 ± 0.04% bone weight) reached concentrations that were no different to the pre-treated diets. In conclusion, phytase improved Atlantic salmon’s growth performance fed low fishmeal diets containing SPC, and at least 1,000 U Phy kg−1 diet was required to have the same effect as pre-treatment of SPC with 250 U Phy kg−1 SPC.



Similar content being viewed by others
Abbreviations
- AD:
-
Apparent digestibility
- FER:
-
Feed efficiency ratio
- FI:
-
Feed intake
- Phy:
-
Phytase
- SPC:
-
Soy protein concentrate
References
AOAC (1995) Official methods of analysis of Official Analytical Chemists International, 16th edn. Association of Official Analytical Chemists, Arlington
Bedford MR (2000) Exogenous enzymes in monogastric nutrition—their current value and future benefits. Anim Feed Sci Technol 86:1–13
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Cain KD, Garling DL (1995) Pretreatment of soybean meal with phytase for salmonid diets to reduce P concentrations in hatchery effluents. Progr Fish Cult 57:114–119
Cao L, Wang WM, Yang CT, Yang Y, Diana J, Yakupitiyage A, Luo Z, Li DP (2007) Application of microbial phytase in fish feed. Enzyme Microb Technol 40:497–507
Carter CG, Hauler RC (2000) Fish meal replacement by plant meals in extruded feeds for Atlantic salmon, Salmo salar L. Aquaculture 185:299–311
Carter CG, Houlihan DF, McCarthy ID (1992) Feed utilisation efficiencies of Atlantic salmon (Salmo salar L.) parr: effect of a single supplementary enzyme. Comp Biochem Physiol 101A:369–374
Carter CG, Houlihan DF, Buchanan B, Mitchell AI (1994) Growth and feed utilization efficiencies of seawater Atlantic salmon. Salmo salar L., fed a diet containing supplementary enzymes. Aquacult Fish Manag 25:37–46
Carter CG, Bransden MP, Lewis TE, Nichols PD (2003) Potential of thraustochytrids to partially replace fish oil in Atlantic salmon feeds. Mar Biotechnol 5:480–492
Carter CG, Katersky RS, Barnes J, Irwin K, Hauler RC (2005) Interactions between temperature and nutrition in Atlantic salmon Salmo salar and barramundi Lates calcarifer. In: Lessons from the past to optimise the future, European Aquaculture Society Special Publication, vol 35, pp 156–157
Denstadli V, Skrede A, Krogdahl A, Sahlstrom S, Storebakken T (2006) Feed intake, growth, feed conversion, digestibility, enzyme activities and intestinal structure in Atlantic salmon (Salmo salar L.) fed graded levels of phytic acid. Aquaculture 256:365–376
Denstadli V, Storebakken T, Svihus B, Skrede A (2007) A comparison of online phytase pre-treatment of vegetable feed ingredients and phytase coating in diets for Atlantic salmon (Salmo salar L.) reared in cold water. Aquaculture 269:414–426
Forster I, Higgs DA, Dosanjh BS, Rowshandeli M, Parr J (1999) Potential for dietary phytase to improve the nutritive value of canola protein concentrate and decrease phosphorus output in rainbow trout (Oncorhynchus mykiss) held in 11°C fresh water. Aquaculture 179:109–125
Gatlin DM, Wilson RP (1984) Zinc supplementation of practical channel catfish diets. Aquaculture 41:31–36
Hauler RC, Carter CG (1997) Phytase stimulates appetite in Atlantic salmon (Salmo salar L.) parr fed soybean meal. Proceedings of the Nutrition Society of Australia, vol 21. The Nutrition Society of Australia, Victoria, p 139
Jackson LS, Li MH, Robinson EH (1996) Use of microbial phytase in channel catfish Ictalurus punctatus diets to improve utilization of phytate phosphorus. J World Aquacult Soc 27:309–313
Kornegay ET (2001) Digestion of phosphorus and other nutrients: the role of phytases and factors influencing their activity. In: Bedford MR, Partridge GG (eds) Enzymes in farm animal nutrition. CAB International, Wallingford, pp 237–271
Lanari D, D’Agaro E, Turri C (1998) Use of nonlinear regression to evaluate the effects of phytase enzyme treatment of plant protein diets for rainbow trout (Oncorhynchus mykiss). Aquaculture 161:345–356
Li MH, Robinson EH (1997) Microbial phytase can replace inorganic phosphorus supplements in channel catfish Ictalurus punctatus diets. J World Aquacult Soc 28:402–406
Masumoto T, Tamura B, Shimeno S (2001) Effects of phytase on bioavailability of phosphorus in soybean meal-based diets for Japanese flounder Paralichthys olivaceus. Fish Sci 67:1075–1080
Maynard LA, Loosli JK (1969) Animal nutrition. McGraw-Hill, New York, p 533
Miller MR, Nichols PD, Barnes J, Davies NW, Peacock EJ, Carter CG (2006) Regiospecificity profiles of storage and membrane lipids from the gill and muscle tissue of Atlantic salmon (Salmo salar L.) grown at elevated temperature. Lipids 41:865–876
Naylor RL, Hardy RW, Bureau DP, Chui A, Elliott M, Farrell AP, Forster I, Gatlin DM, Goldburg RJ et al (2009) Feeding aquaculture in an era of finite resources. Proc Natl Acad Sci USA 106:15103–15110
NRC (National Research Council) (1993) Nutrient requirements of fish. National Academy Press, Washington DC, p 114
Nwanna LC, Kolahsa M, Eisenreich R, Schwarz FJ (2008) Pre-treatment of dietary plant feedstuffs with phytase and its effect on growth and mineral concentration in common carp (Cyprinus carpio L.). J Anim Physiol Anim Nutr 92:677–682
Rao D, Rao KV, Reddy TP, Reddy VD (2009) Molecular characterization, physicochemical properties, known and potential applications of phytases: an overview. Crit Rev Biotechnol 29:182–198
Roberts RJ, Hardy RW, Sugiura SH (2001) Screamer disease in Atlantic salmon, Salmo salar L., in Chile. J Fish Dis 24:543–549
Rodehutscord M, Pfeffer E (1995) Effects of supplemental microbial phytase on phosphorus digestibility and utilization in rainbow trout Oncorhynchus mykiss. Water Sci Technol 31:143–147
Sajjadi M, Carter CG (2004a) Effect of phytic acid and phytase on feed intake, growth, digestibility and trypsin activity in Atlantic salmon (Salmo salar L.). Aquacult Nutr 10:135–142
Sajjadi M, Carter CG (2004b) Dietary phytase supplementation and the utilization of phosphorus by Atlantic salmon (Salmo salar L.) fed a canola meal based diet. Aquaculture 240:417–431
Sales J (2009) The effect of fish meal replacement by soyabean products on fish growth: a meta-analysis. Brit J Nutr 102:1709–1722
Satoh S, Poe WE, Wilson RP (1989) Effect of supplemental phytate and/or tricalcium phosphate on weight gain, feed efficiency and zinc content in vertebrae of channel catfish. Aquaculture 80:155–161
Schäfer A, Koppe WM, Meyer-Burgdorrf KH, Günther KD (1995) Effects of a microbial phytase on the utilization of native phosphorus by carp in diet based on soybean meal. Water Sci Technol 31:149–155
Storebakken T, Shearer KD, Roem AJ (1998) Availability of protein, phosphorus and other elements in fish meal, soy-protein-concentrate and phytase-treated soy-protein-concentrate-based diets to Atlantic salmon, Salmo salar. Aquaculture 161:365–379
Sugiura SH, Dong FM, Hardy RW (1998) Effects of dietary supplements on the availability of minerals in fish meal; preliminary observations. Aquaculture 160:283–303
Sugiura SH, Gabaudan J, Dong FM, Hardy RW (2001) Dietary microbial phytase supplementation and the utilization of phosphorus, trace minerals and protein by rainbow trout [Oncorhynchus mykiss (Walbaum)] fed soybean meal-based diets. Aquacult Res 32:583–592
Van Weerd JH, Khalaf KA, Aartsen FJ, Tijssen PAT (1999) Balance trials with African catfish Clarias gariepinus fed phytase-treated soybean meal-based diets. Aquacult Nutr 5:135–142
Vandecasteele C, Block CB (1993) Modern methods for trace element determination. Wiley, Brisbane, p 330
Vielma J, Lall SP, Koskela J, Schöner F-J, Mattila P (1998) Effects of dietary phytase and cholecalciferol on phosphorus bioavailability in rainbow trout, Oncorhynchus mykiss. Aquaculture 163:307–321
Vielma J, Ruohonen K, Peisker M (2002) Dephytinization of two soy proteins increases phosphorus and protein utilization by rainbow trout, Oncorhynchus mykiss. Aquaculture 204:145–156
Wang F, Yang YH, Han ZZ, Dong HW, Yang CH, Zou ZY (2009) Effects of phytase pre-treatment of soybean meal and phytase-sprayed in diets on growth, apparent digestibility coefficient and nutrient excretion of rainbow trout (Oncorhynchus mykiss Walbaum). Aquacult Int 17:143–157
Ward DA, Carter CG, Townsend AT (2005) The use of yttrium oxide and the effect of faecal collection timing for determining the apparent digestibility of minerals and trace elements in Atlantic salmon (Salmo salar L.) feeds. Aquacult Nutr 11:49–59
Acknowledgments
The authors gratefully acknowledge P. Selle and C. Roach (BASF Australia Ltd.) for providing phytase, L. Browning (ex Roche Vitamins Australia Ltd. Sydney) for supplying vitamins C and E used in this study and H. King (SALTAS Hatchery, Wayatinah, Tasmania) for supplying the fish.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carter, C.G., Sajjadi, M. Low fishmeal diets for Atlantic salmon, Salmo salar L., using soy protein concentrate treated with graded levels of phytase. Aquacult Int 19, 431–444 (2011). https://doi.org/10.1007/s10499-010-9358-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-010-9358-z