Abstract
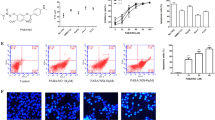
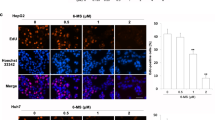
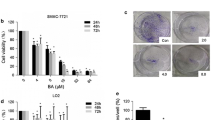
Hepatocellular carcinoma (HCC) is one of the main causes of death in cancer. Some naphthalimide derivatives exert high anti-proliferative effects on HCC. In this study, it is confirmed that 3-nitro-naphthalimide and nitrogen mustard conjugate (NNM-25), a novel compound conjugated by NNM-25, displayed more potent therapeutic action on HCC, both in vivo and in vitro, than amonafide, a naphthalimide drug in clinical trials. More importantly, preliminary toxicological evaluation also supported that NNM-25 exhibited less systemic toxicity than amonafide at the therapeutic dose. The antitumor mechanism of conjugates of naphthalimides with nitrogen mustard remains poorly understood up to now. Here, we first reported that apoptosis might be the terminal fate of cancer cells treated with NNM-25. Inhibition of p53 by siRNA resulted in a significant decrease of NNM-25-induced apoptosis, which corroborated that p53 played a vital role in the cell apoptosis triggered by NNM-25. NNM-25 inhibited the PARP-1 activity, AKT phosphorylation, up-regulated the protein expression of p53, Bad, and mTOR as well as down-regulating the protein expression of Bcl-2 and decreasing mitochondrial membrane potential. It also facilitated cytochrome c release from mitochondria to cytoplasm, activated caspase 8, caspase 9, and caspase 3 in HepG2 cells in vitro, as also authenticated in H22 tumor-bearing mice in vivo. Collectively, the conjugation of naphthalimides with nitrogen mustard provides favorable biological activity and thus is a valuable strategy for future drug design in HCC therapy.





Similar content being viewed by others
References
Alves RC, Alves D, Guz B, Matos C, Viana M, Harriz M (2011) Advanced hepatocellular carcinoma. Review of targeted molecular drugs. Ann Hepatol 10:21–27
Duffy A, Greten T (2010) Develo** better treatments in hepatocellular carcinoma. Expert Rev Gastroenterol Hepatol 4:551–560
Ingrassia L, Lefranc F, Kiss R, Mijatovic T (2010) Naphthalimides and azonafides as promising anti-cancer agents. Curr Med Chem 16:1192–1213
Tian ZY, **e SQ, Du YW, Ma YF, Zhao J, Gao WY, Wang CJ (2009) Synthesis, cytotoxicity and apoptosis of naphthalimide polyamine conjugates as antitumor agents. Eur J Med Chem 44:393–399
Tian ZY, **e SQ, Mei ZH, Zhao J, Gao WY, Wang CJ (2010) Conjugation of substituted naphthalimides to polyamines as cytotoxic agents targeting the Akt/mTOR signal pathway. Org Biomol Chem 7:4651–4660
Pain A, Samanta S, Dutta S, Saxena AK, Shanmugavel M, Kampasi H, Quazi GN, Sanyal U (2003) Synthesis and evaluation of substituted naphthalimide nitrogen mustards as rationally designed anticancer compounds. Acta Pol Pharm 60:285–291
Horton JK, Stefanick DF, Zeng JY, Carrozza MJ, Wilson SH (2011) Requirement for NBS1 in the S phase checkpoint response to DNA methylation combined with PARP inhibitory. DNA Repair 10:225–234
Powell C, Mikropoulos C, Kaye SB, Nutting CM, Bhide SA, Newbold K (2010) Pre-clinical and clinical evaluation of PARP inhibitors as tumour-specific radiosensitisers. Cancer Treat Rev 36:566–575
Rodon J, Iniesta MD, Papadopoulos K (2009) Development of PARP inhibitors in oncology. Expert Opin Investig Drugs 18:31–43
Heacock ML, Stefanick DF, Horton JK, Wilson SH (2010) Alkylation DNA damage in combination with PARP inhibitory results in formation of S-phase-dependent double-strand breaks. DNA Repair 9:929–936
Wang S, Jia L, Zhou H, Shi W, Zhang J (2009) Knockdown of caveolin-1 by siRNA inhibits the transformation of mouse hepatoma H22 cells in vitro and in vivo. Oligonucleotides 19:81–88
Kuang DM, Peng C, Zhao Q, Wu Y, Chen MS, Zheng L (2010) Activated monocytes in peritumoral stroma of hepatocellular carcinoma promote expansion of memory T helper 17 cells. Hepatology 51:154–164
**e SQ, Li Q, Zhang YH, Wang JH, Mei ZH, Zhao J, Wang CJ (2011) NPC-16, a novel naphthalimide-polyamine conjugate, induced apoptosis and autophagy in human hepatoma HepG2 cells and Bel-7402 cells. Apoptosis 16:27–34
Alano CC, Kauppinen TM, Valls AV, Swanson RA (2006) Minocycline inhibits poly(ADP-ribose) polymerase-1 at nanomolar concentrations. Proc Natl Acad Sci USA 103:9685–9690
Bergamaschi D, Gasco M, Hiller L, Sullivan A, Syed N, Trigiante G, Yulug I, Merlano M, Numico G, Comino A, Attard M, Reelfs O, Gusterson B, Bell AK, Heath V, Tavassoli M, Farrell PJ, Smith P, Lu X, Crook T (2003) p53 polymorphism influences response in cancer chemotherapy via modulation of p73-dependent apoptosis. Cancer Cell 3:387–402
Van Quaquebeke E, Mahieu T, Dumont P, Dewelle J, Ribaucour F, Simon G, Sauvage S, Gaussin JF, Tuti J, El Yazidi M, Van Vynckt F, Mijatovic T, Lefranc F, Darro F, Kiss R (2007) 2,2,2-Trichloro-N-({2-[2-(dimethylamino)ethyl]-1,3-dioxo-2,3-dihydro-1H-benzo[de]isoquinolin-5-yl}carbamoyl)acetamide (UNBS3157), a novel non-hematotoxic naphthalimide derivative with potent antitumor activity. J Med Chem 50:4122–4134
Pain A, Samanta S, Dutta S, Saxena AK, Shanmugavel M, Kampasi H (2003) Evaluation of naphthalmustine, a nitrogen mustard derivative of naphthalimide as a rationally-designed anticancer agent. J Exp Clin Cancer Res 22:411–418
Sandhu SK, Yap TA, de Bono JS (2010) Poly(ADP-ribose) polymerase inhibitors in cancer treatment: a clinical perspective. Eur J Cancer 46:9–20
Löser DA, Shibata A, Shibata AK, Woodbine LJ, Jeggo PA (2010) Chalmers, sensitization to radiation and alkylating agents by inhibitors of poly(ADP-ribose) polymerase is enhanced in cells deficient in DNA double-strand break repair. Mol Cancer Ther 9:1775–1787
Huang SH, **ong M, Chen XP, **ao ZY, Zhao YF, Huang ZY (2008) PJ34, an inhibitor of PARP-1, suppresses cell growth and enhances the suppressive effects of cisplatin in liver cancer cells. Oncol Rep 20:567–572
Fiorillo C, Ponziani V, Giannini L, Cecchi C, Celli A, Nassi N, Lanzilao L, Caporale R, Nassi P (2006) Protective effects of the PARP-1 inhibitor PJ34 in hypoxic-reoxygenated cardiomyoblasts. Cell Mol Life Sci 63:3061–3071
Pu T, Zhang XP, Liu F, Wang W (2010) Coordination of the nuclear and cytoplasmic activities of p53 in response to DNA damage. Biophys J 99:1696–1705
Smith ML, Kumar MA (2010) The “two faces” of tumor suppressor p53-revisited. Mol Cell Pharmacol 2:117–119
Acknowledgments
This study was funded by the National Natural Science Foundation of China (No. 90913001) for Chao-jie Wang, Projects of Science and Technology of Henan (No. 0821022700; 102300410095) and China Postdoctoral Science Foundation Funded Project (No. 20090450092; 201003395) for Song-qiang **e.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
**e, Sq., Zhang, Yh., Li, Q. et al. 3-Nitro-naphthalimide and nitrogen mustard conjugate NNM-25 induces hepatocellular carcinoma apoptosis via PARP-1/p53 pathway. Apoptosis 17, 725–734 (2012). https://doi.org/10.1007/s10495-012-0712-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-012-0712-7