Abstract
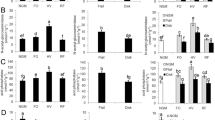
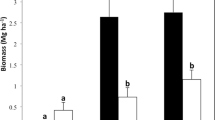
Black walnut (Juglans nigra) and honeylocust (Gleditsia triacanthos) have gained particular interest for use in silvopastures and other temperate agroforestry systems. However, measures of soil health indicators in these deciduous hardwood silvopastures over time have received limited attention. This study assessed soil health indicators in 25-year-old black walnut- (BSP) and honeylocust-based silvopastures (HSP) and compared with those from adjacent open pasture (OP) systems. Soil samples collected from 0 to 10 cm depth were analyzed for soil organic matter (OM), carbon and nitrogen fractions, glomalin-related soil protein, soil enzymatic activities, and microbial community structure. The soil OM content in HSP (64 g kg−1) was greater (p < 0.05) than BSP and OP (55 g kg−1). The BSP soils had about 45% and 52% more (p < 0.05) microbial biomass carbon and nitrogen than OP soils. The HSP soils had 60% greater (p < 0.05) microbial biomass nitrogen than OP soils. The BSP supported greater (p < 0.05) β-glucosidase and urease activities than the HSP and OP treatments, while greater (p < 0.05) β-glucosidase activity was measured in HSP than OP. Total phospholipid-derived fatty acid and actinomycetes abundance were greater (p < 0.001) in OP compared to BSP and HSP while the arbuscular mycorrhiza fungi abundance was greater (p < 0.05) in OP than in HSP. These findings indicate that black walnut and honeylocust trees in the silvopastures with proper management can improve certain soil health parameters over time depending on tree characteristics and the age of the system.


Similar content being viewed by others
References
Akinnifesi FK, Kang BT, Ladipo DO (1999) Structural root form and fine root distribution of some woody species evaluated for agroforestry systems. Agrofor Syst 42(2):121–138. https://doi.org/10.1023/A:1006178002019
Alfaia SS, Ribeiro GA, Nobre AD, Luizao RC, Luizao FJ (2004) Evaluation of soil fertility in smallholder agroforestry system and pastures in western Amazonia. Agric Ecosyst Environ 102:409–414. https://doi.org/10.1016/j.agee.2003.08.011
Beck T, Joergensen RG, Kandeler E, Makeschin F, Nuss E, Oberholzer HR, Scheu S (1997) An inter-laboratory comparison of ten different ways of measuring soil microbial biomass C. Soil Biol Biochem 29(7):1023–1032. https://doi.org/10.1016/S0038-0717(97)00030-8
Belsky AJ, Mwonga SM, Duxbury JM (1993) Effects of widely spaced trees and livestock grazing on understory environments in tropical savannas. Agrofor Syst 24:1–20
Bendfeldt ES, Feldhake CM, Burger JA (2001) Establishing trees in an Appalachian silvopasture: response to shelters, grass control, mulch, and fertilization. Agrofor Syst 53:291–295. https://doi.org/10.1023/A:1013367224860
Bosi C, Pezzopane JRM, Sentelhas PC (2020) Soil water availability in a full sun pasture and in a silvopastoral system with eucalyptus. Agrofor Syst 94:429–440. https://doi.org/10.1007/s10457-019-00402-7
Buresh RJ, Tian G (1998) Soil improvement in sub-Saharan Africa. Agrofor Syst 38:51–76. https://doi.org/10.1007/978-94-015-9008-2_2
Campo J, Maass M, Jaramillo VJ, Martínez-Yrízar A, Sarukhán J (2001) Phosphorus cycling in a Mexican tropical dry forest ecosystem. Biogeochemistry 53(2):161–179. https://doi.org/10.1023/A:1010663516029
Casals P, Romero J, Rusch GM, Ibrahim M (2014) Soil organic C and nutrient contents under trees with different functional characteristics in seasonally dry tropical silvopastures. Plant Soil 374(1):643–659. https://doi.org/10.1007/s11104-013-1884-9
Clapperton M, Lacey M, Hanson K, Hamel C (2005) Analysis of phospholipid and neutral lipid fatty acids extracted from soils. Research Newsletter. Swift Current, SK: SPARC‐AAFC December 12
Clark AM, Jurgens TM, Hufford CD (1990) Antimicrobial activity of juglone. Phytother Res 4(1):11–14. https://doi.org/10.1002/ptr.2650040104
Cornelissen JHC (1996) An experimental comparison of leaf decomposition rates in a wide range of temperate plant species and types. J Ecol. https://doi.org/10.2307/2261479
Cubillos AM, Vallejo VE, Arbeli Z, Teran W, Dick RP, Molina CH, Molina E, Roldan F (2016) Effect of the conversion of conventional pasture to intensive silvopastoral systems on edaphic bacterial and ammonia oxidizer communities in Colombia. Eur J Soil Biol 72:42–50. https://doi.org/10.1016/j.ejsobi.2015.12.003
Eivazi F, Tabatabai MA (1977) Phosphatases in soils. Soil Biol Biochem 9(3):167–172
Eivazi F, Tabatabai MA (1988) Glucosidases and galactosidases in soils. Soil Biol Biochem 20(5):601–606
Fannon AG, Fike JH, Greiner SP, Feldhake CM, Wahlberg MA (2017) Hair sheep performance in a mid-stage deciduous Appalachian silvopasture. Agrofor Syst. https://doi.org/10.1007/s10457-017-0154-x
Fierer N, Strickland MS, Liptzin D, Bradford MA, Cleveland CC (2009) Global patterns in belowground communities. Ecol Lett 12:1238–1249. https://doi.org/10.1111/j.1461-0248.2009.01360.x
Garrett HE, Rietveld WJ, Fisher RF (2000) North American agroforestry: an integrated science and practice. American Society of Agronomy, Madison
Ghani A, Dexter M, Perrott KW (2003) Hot-water extractable carbon in soils: a sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol Biochem 35(9):1231–1243. https://doi.org/10.1016/S0038-0717(03)00186-X
Gregorich EG, Wen G, Voroney RP, Kachanoski RG (1990) Calibration of a rapid direct chloroform extraction method for measuring soil microbial biomass C. Soil Biol Biochem 22(7):1009–1011. https://doi.org/10.1016/0038-0717(90)90148-S
Haile SG, Nair PR, Nair VD (2008) Carbon storage of different soil-size fractions in Florida silvopastoral systems. J Environ Qual 37(5):1789–1797. https://doi.org/10.2134/jeq2007.0509
Hawken P (2017) Drawdown: the most comprehensive plan ever proposed to reverse global warming. Penguin, London
Hopkins MF, Filley RT, Gleixner G, Lange M, Top MS, Trumbore ES (2014) Increased belowground carbon inputs and warming promote loss of soil organic carbon through complementary microbial responses. Soil Biol Biochem 76:57–69. https://doi.org/10.1016/j.soilbio.2014.04.028
Jackson RB, Schenk HJ, Jobbagy EG, Canadell J, Colello GD, Dickinson RE, Field CB, Friedlingstein P, Heimann M, Hibbard K, Kicklighter DW (2000) Belowground consequences of vegetation change and their treatment in models. Ecol Appl 10(2):470–483. https://doi.org/10.1890/1051-0761(2000)010[0470:BCOVCA]2.0.CO;2
Janzen HH, Janzen DW, Gregorich EG (2021) The ‘soil health’ metaphor: Illuminating or illusory? Soil Biol Biochem 159:108167. https://doi.org/10.1016/j.soilbio.2021.108167
Johnston AE, Poulton PR, Coleman K (2009) Soil organic matter: its importance in sustainable agriculture and carbon dioxide fluxes. Adv Agron 101:1–57. https://doi.org/10.1016/S0065-2113(08)00801-8
Kahlon MS, Khurana K (2017) Effect of land management practices on physical properties of soil and water productivity in the wheat-maize system of Northwest India. Appl Ecol Environ Res 15(4):1–13. https://doi.org/10.15666/aeer/1504_001013
Kandeler E, Gerber H (1988) Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol Fertil Soils 6(1):68–72. https://doi.org/10.1007/BF00257924
Karki S, Shange R, Ankumah R, McElhenney W, Idehen O, Poudel S, Karki U (2021) Comparative assessment of soil health indicators in response to woodland and silvopasture land use systems. Agrofor Syst 95(1):227–240. https://doi.org/10.1007/s10457-020-00577-4
Liang C (2020) Soil microbial carbon pump: mechanism and appraisal. Soil Ecol Lett 2(4):241–254. https://doi.org/10.1007/s42832-020-0052-4
Lundquist EJ, Jackson LE, Scow KM, Hsu C (1999) Changes in microbial biomass and community composition, and soil carbon and nitrogen pools after incorporation of rye into three California agricultural soils. Soil Biol Biochem 31:221–236. https://doi.org/10.1016/S0038-0717(98)00093-5
Malik AA, Chowdhury S, Schlager V, Oliver A, Puissant J, Vazquez PG, Jehmlich N, Von Bergen M, Griffiths RI, Gleixner G (2016) Soil fungal: bacterial ratios are linked to altered carbon cycling. Front Microbiol 7:1247. https://doi.org/10.3389/fmicb.2016.01247
Marschner P, Kandeler E, Marschner B (2003) Structure and function of the soil microbial community in a long-term fertilizer experiment. Soil Biol Biochem 35(3):453–461
Nair VD, Nair PKR, Kalmbacher RS, Ezenwa IV (2007) Reducing nutrient loss from farms through silvopastoral practices in coarse-textured soils of Florida, USA. Ecol Eng 29(2):192–199. https://doi.org/10.1016/j.ecoleng.2006.07.003
Nelson DW, Sommers LE (1996) Total carbon, organic carbon and organic matter. In: Sparks DL (ed) Methods of soil analysis. Part 3. Chemical methods. SSSA Book Ser. 5. SSSA, Madison, pp 961–1010
Peichl M, Thevathasan NV, Gordon A, Huss J, Abohassan RA (2006) Carbon sequestration potentials in temperature tree-based intercrop** systems, southern Ontario, Canada. Agrofor Syst 66:243–257
Pent GJ (2020) Over-yielding in temperate silvopastures: a meta-analysis. Agrofor Syst 94(5):1741–2175. https://doi.org/10.1007/s10457-020-00494-6
Pent GJ, Greiner SP, Munsell JF, Tracy BF, Fike JH (2020) Lamb performance in hardwood silvopastures, I: animal gains and forage measures in summer. Transl Anim Sci 4(1):385–399. https://doi.org/10.1093/tas/txz154
Ramakrishnan S, Kumar S, Chaudhary M, Govindasamy P, Yadav M, Prasad M, Srivastava R, Kumari B, Prajapati K (2020) Silvopastoral system for resilience of key soil health indicators in semi-arid environment. Arch Agron Soil Sci. https://doi.org/10.1080/03650340.2020.1814954
Rhoades CC (1997) Single-tree influences on soil properties in agro-forestry: lessons from natural forest and savanna ecosystems. Agrofor Syst 35:71–94. https://doi.org/10.1007/BF02345330
Schroth G (1995) Tree root characteristics as criteria for species selection and systems design in agroforestry. Agrofor Syst 30:125–143. https://doi.org/10.1007/978-94-017-0681-0_6
Sharrow SH, Ismail S (2004) Carbon and nitrogen storage in agroforests, tree plantations, and pastures in western Oregon, USA. Agrofor Syst 60(2):123–130. https://doi.org/10.1023/B:AGFO.0000013267.87896.41
Staley TE, Gonzalez JM, Neel JP (2008) Conversion of deciduous forest to silvopasture produces soil properties indicative of rapid transition to improved pasture. Agrofor Syst 74(3):267. https://doi.org/10.1007/s10457-008-9139-0
Tabatabai MA, Bremner JM (1972) Assay of urease activity in soils. Soil Biol Biochem 4(4):479–487. https://doi.org/10.1016/0038-0717(72)90064-8
Tonucci RG, Nair PR, Nair VD, Garcia R, Bernardino FS (2011) Soil carbon storage in silvopasture and related land-use systems in the Brazilian Cerrado. J Environ Qual 40(3):833–841. https://doi.org/10.2134/jeq2010.0162
USDA NRCS (Natural Resources Conservation Service) (2014). Soil health home. https://www.nrcs.usda.gov/wps/portal/nrcs/main/soils/health/. Accessed 23 April 2022
Vallejo VE, Arbeli Z, Terán W, Lorenz N, Dick RP, Roldan F (2012) Effect of land management and Prosopis juliflora (Sw.) DC trees on soil microbial community and enzymatic activities in intensive silvopastoral systems of Colombia. Agric Ecosyst Environ 150:139–148. https://doi.org/10.1016/j.agee.2012.01.022
Watson ME, Brown JR (1998) pH and lime requirement. Recommended chemical soil test procedures for the North Central Region. North Central Regional Research Publication No. 221 (Revised). Missouri Agricultural Experiment Station SB 1001, Columbia
Weintraub SR, Wieder WR, Cleveland CC, Townsend AR (2013) Organic matter inputs shift soil enzyme activity and allocation patterns in a wet tropical forest. Biogeochemistry 114(1):313–326. https://doi.org/10.1007/s10533-012-9812-2
Wright SF, Upadhyaya A (1998) A survey of soils for aggregate stability and glomalin, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant Soil 198(1):97–107. https://doi.org/10.1023/A:1004347701584
Zantua MI, Dumenil LC, Bremner JM (1977) Relationships between soil urease activity and other soil properties. Soil Sci Soc Am J 41(2):350–352. https://doi.org/10.2136/sssaj1977.03615995004100020036x
Acknowledgements
The authors would like to acknowledge Virginia Tech Soil Testing Lab, Virginia Tech Environmental Quality Lab, and Soil Physics Lab at South Dakota State University for help with all the lab analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Poudel, S., Bansal, S., Podder, S. et al. Conversion of open pasture to hardwood silvopasture enhanced soil health of an ultisol. Agroforest Syst 96, 1237–1247 (2022). https://doi.org/10.1007/s10457-022-00783-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-022-00783-2