Abstract
Purpose
To evaluate the relationships between optic nerve head blood flow, expressed as mean blur rate (MBR) measured by laser speckle flowgraphy (LSFG), and photocoagulation therapy in neonates with retinopathy of prematurity (ROP).
Study design
Case series study.
Methods
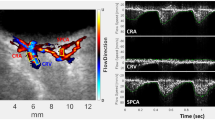
We studied 5 ROP neonates either during sleep or under sedation both before and after photocoagulation, and evaluated 8 eyes in which the circulation could be measured three times consecutively. Correlations between the MBR-A (mean of all values), MBR-V (vessel mean) and MBR-T (tissue mean) and postmenstrual age were evaluated using Spearman’s rank correlation coefficient. In addition, correlations between the relative MBR (-A, -V, -T) value and number of photocoagulation burns and the NV score were evaluated. Differences between post-treatment MBR in ROP subjects and normal neonates’ MBR were estimated using analysis of covariance (ANCoVA), with adjustment for postmenstrual age.
Results
The relative MBR (-A, -V, -T) values after photocoagulation were 69.6 ± 16.0%, 66.7 ± 17.0% and 74.3 ± 14.6%, respectively. Postmenstrual age was significantly correlated with post-treatment MBR-A (r = 0.83, p = 0.0101), MBR-V (r = 0.85, p = 0.007) and MBR-T (r = 0.76, p = 0.0282). The relative MBR-T value was significantly correlated with the number of photocoagulation burns (r = −0.75, p = 0.033) and NV score (r = −0.72, p = 0.0437). The ANCoVA results showed no significant difference between post-treatment MBR and normal neonates’ MBR.
Conclusions
Photocoagulation improved the dilation of veins and tortuosity of arteries and reduced ocular blood flow in ROP subjects. Since the post-treatment MBR was not different from a normal neonate’s MBR, it is suggested that the pre-treatment MBR was higher in severe ROP cases.




Similar content being viewed by others
References
Early Treatment For Retinopathy of Prematurity Cooperative Group. Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol. 2003;121:1684–94.
Gelman R, Martinez-Perez ME, Vanderveen DK, Moskowitz A, Fulton AB. Diagnosis of plus disease in retinopathy of prematurity using Retinal Image multiScale Analysis. Investig Ophthalmol Vis Sci. 2005;46:4734–8.
Koreen S, Gelman R, Martinez-Perez ME, Jiang L, Berrocal AM, Hess DJ, et al. Evaluation of a computer-based system for plus disease diagnosis in retinopathy of prematurity. Ophthalmology. 2007;114:e59–67.
Chiang MF, Gelman R, Williams SL, Lee JY, Casper DS, Martinez-Perez ME, et al. Plus disease in retinopathy of prematurity: development of composite images by quantification of expert opinion. Investig Ophthalmol Vis Sci. 2008;49:4064–70.
Wittenberg LA, Jonsson NJ, Chan RV, Chiang MF. Computer-based image analysis for plus disease diagnosis in retinopathy of prematurity. J Pediatr Ophthalmol Strabismus. 2012;49:11–9; quiz 0, 20.
Campbell JP, Ataer-Cansizoglu E, Bolon-Canedo V, Bozkurt A, Erdogmus D, Kalpathy-Cramer J, et al. Expert diagnosis of plus disease in retinopathy of prematurity from computer-based image analysis. JAMA Ophthalmol. 2016;134:651–7.
Baerts W, Blecourt-Devilee MWD, Sauer PJ. Ambient light, ophthalmic artery blood flow velocities and retinopathy of prematurity. Acta Paediatr. 1993;82:719–22.
Holland DR, Saunders RA, Kagemann LE, Bluestein EC, Hutchinson AK, Corson DW, et al. Color Doppler imaging of the central retinal artery in premature infants undergoing examination for retinopathy of prematurity. J AAPOS. 1999;3:194–8.
Niwald A, Gralek M. Evaluation of blood flow in the ophthalmic artery and central retinal artery in children with retinopathy of prematurity. Klin Oczna. 2006;108:32–5.
Neely D, Harris A, Hynes E, McNulty L, McCranor L, Siesky B, et al. Longitudinal assessment of plus disease in retinopathy of prematurity using color Doppler imaging. J AAPOS. 2009;13:509–11.
Hartenstein S, Muller B, Metze B, Czernik C, Buhrer C. Blood flow assessed by color Doppler imaging in retinopathy of prematurity. J Perinatol. 2015;35:745–7.
Tamaki Y, Araie M, Kawamoto E, Eguchi S, Fujii H. Noncontact, two-dimensional measurement of retinal microcirculation using laser speckle phenomenon. Investig Ophthalmol Vis Sci. 1994;35:3825–34.
Tamaki Y, Araie M, Kawamoto E, Eguchi S, Fujii H. Non-contact, two-dimensional measurement of tissue circulation in choroid and optic nerve head using laser speckle phenomenon. Exp Eye Res. 1995;60:373–83.
Sugiyama T, Araie M, Riva CE, Schmetterer L, Orgul S. Use of laser speckle flowgraphy in ocular blood flow research. Acta Ophthalmol. 2010;88:723–9.
Aizawa N, Yokoyama Y, Chiba N, Omodaka K, Yasuda M, Otomo T, et al. Reproducibility of retinal circulation measurements obtained using laser speckle flowgraphy-NAVI in patients with glaucoma. Clin Ophthalmol. 2011;5:1171–6.
Sugiyama T. Basic technology and clinical applications of the updated model of laser speckle flowgraphy to ocular diseases. Photonics. 2014;1:220–34.
Fujii H. Visualisation of retinal blood flow by laser speckle flowgraphy. Med Biol Eng Comput. 1994;32:302–4.
Aizawa N, Nitta F, Kunikata H, Sugiyama T, Ikeda T, Araie M, et al. Laser speckle and hydrogen gas clearance measurements of optic nerve circulation in albino and pigmented rabbits with or without optic disc atrophy. Investig Ophthalmol Vis Sci. 2014;55:7991–6.
Matsumoto T, Itokawa T, Shiba T, Katayama Y, Arimura T, Mizukaki N, et al. Reproducibility of neonate ocular circulation measurements using laser speckle flowgraphy. Biomed Res Int. 2015;2015:693056.
Matsumoto T, Itokawa T, Shiba T, Hine K, Hori Y. A change in ocular circulation after photocoagulation for retinopathy of prematurity in a neonate. Case Rep Ophthalmol. 2017;8:91–8.
Matsumoto T, Itokawa T, Shiba T, Katayama Y, Arimura T, Hine K, et al. Ocular blood flow values measured by laser speckle flowgraphy correlate with the postmenstrual age of normal neonates. Graefes Arch Clin Exp Ophthalmol. 2016;254:1631–6.
Romagnoli C, Papacci P, Zecca E, Giannantonio C, De Carolis MP, Tortorolo G. Normal neonatal values of ophthalmic and central retinal artery blood flow velocities. J Pediatr Ophthalmol Strabismus. 2001;38:213–7.
Gunay M, Tuten A, Sancak S, Celik G, Bardak H, Dincer E, et al. Effect of single intravitreal bevacizumab on ophthalmic and middle cerebral arterial blood flow in retinopathy of prematurity. Ophthalmic Res. 2016;55:165–71.
Yalin Imamoglu E, Gunay M, Gursoy T, Imamoglu S, Ekmekci OB, Celik G, et al. Effect of laser photocoagulation on plasma levels of VEGF-A, VEGFR-2, and Tie2 in infants with retinopathy of prematurity. J AAPOS. 2014;18:466–70.
Iwase T, Kobayashi M, Yamamoto K, Ra E, Terasaki H. Effects of photocoagulation on ocular blood flow in patients with severe non-proliferative diabetic retinopathy. PLoS ONE. 2017;12:e0174427.
Pechauer AD, Hwang TS, Hagag AM, Liu L, Tan O, Zhang X, et al. Assessing total retinal blood flow in diabetic retinopathy using multiplane en face Doppler optical coherence tomography. Br J Ophthalmol. 2017. doi:10.1136/bjophthalmol-2016-310042.
Srinivas S, Tan O, Nittala MG, Wu JL, Fazwi AA, Huang D, et al. Assessment of retinal blood flow in diabetic retinopathy using Doppler Fourier-domain optical coherence tomography. Retina. 2017. doi:10.1097/IAE.0000000000001479.
Feke GT, Green GJ, Goger DG, McMeel JW. Laser Doppler measurements of the effect of panretinal photocoagulation on retinal blood flow. Ophthalmology. 1982;89:757–62.
Grunwald JE, Riva CE, Brucker AJ, Sinclair SH, Petrig BL. Effect of panretinal photocoagulation on retinal blood flow in proliferative diabetic retinopathy. Ophthalmology. 1986;93:590–5.
Ishida S, Usui T, Yamashiro K, Kaji Y, Amano S, Ogura Y, et al. VEGF164-mediated inflammation is required for pathological, but not physiological, ischemia-induced retinal neovascularization. J Exp Med. 2003;198:483–9.
Ishida S, Usui T, Yamashiro K, Kaji Y, Ahmed E, Carrasquillo KG, et al. VEGF164 is proinflammatory in the diabetic retina. Investig Ophthalmol Vis Sci. 2003;44:2155–62.
Lee JC, Wong BJ, Tan O, Srinivas S, Sadda SR, Huang D, et al. Pilot study of Doppler optical coherence tomography of retinal blood flow following laser photocoagulation in poorly controlled diabetic patients. Investig Ophthalmol Vis Sci. 2013;54:6104–11.
Okamoto M, Matsuura T, Ogata N. Effects of panretinal photocoagulation on choroidal thickness and choroidal blood flow in patients with severe nonproliferative diabetic retinopathy. Retina. 2016;36:805–11.
Novellas R, Ruiz de Gopegui R, Espada Y. Effects of sedation with midazolam and butorphanol on resistive and pulsatility indices in healthy dogs. Vet Radiol Ultrasound. 2007;48:276–80.
Acknowledgements
This work was supported by Toho University project Grants (#26-34), a Toho University Nukada scholarship and a Toho University Shibata scholarship. The authors would like to express appreciation for advice on the statistical analysis by Mar S. Oba (Department of Medical Statistics Faculty of Medicine, Toho University).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
T. Matsumoto, None; T. Itokawa, None; T. Shiba, None; M. Tomita, None; K. Hine, None; N. Mizukaki, None; H. Yoda, None; Y. Hori, None.
About this article
Cite this article
Matsumoto, T., Itokawa, T., Shiba, T. et al. Decreased ocular blood flow after photocoagulation therapy in neonatal retinopathy of prematurity. Jpn J Ophthalmol 61, 484–493 (2017). https://doi.org/10.1007/s10384-017-0536-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-017-0536-7