Abstract
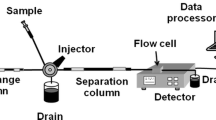
An ion-exclusion chromatography (IELC) comparison between a conventional ion-exchange column and an ultra high-performance liquid chromatography (UHPLC) dynamically surfactant modified C18 column for the separation of an aliphatic carboxylic acid and two aromatic carboxylic acids is presented. Professional software is used to optimize the conventional IELC separation conditions for acetylsalicylic acid and the hydrolysis products: salicylic acid and acetic acid. Four different variables are simultaneously optimized including H2SO4 concentration, pH, flow rate, and sample injection volume. Thirty different runs are suggested by the software. The resolutions and the time of each run are calculated and feed back to the software to predict the optimum conditions. Derringer’s desirability functions are used to evaluate the test conditions and those with the highest desirability value are utilized to separate acetylsalicylic acid, salicylic acid, and acetic acid. These conditions include using a 0.35 mM H2SO4 (pH 3.93) eluent at a flow rate of 1 mL min−1 and an injection volume of 72 μL. To decrease the run time and improve the performance, a UHPLC C18 column is used after dynamic modification with sodium dodecyl sulfate. Using pure water as a mobile phase, a shorter analysis time and better resolution are achieved. In addition, the elution order is different from the IELC method which indicates the contribution of the reversed-phase mode to the separation mechanism.







Similar content being viewed by others
References
Wheaton RM, Bauman WC (1953) Ann N Y Acad Sci 57:159–176
Ohta K, Towata A, Ohashi M (2003) J Chromatogr A 997:95–106
Fritz JS, Gjerde DT (2009) Ion chromatography. WILEY-VCH Verlag GmbH & Co. KGA, Weinheim
Ohta K, Tanaka K, Haddad PR (1997) J Chromatogr A 782:33–40
Mansour FR, Kirkpatrick CL, Danielson ND (2013) J Chromatogr Sci. doi:10.1093/chromsci/bmt007
Ohta K, Tanaka K (1999) J Chromatogr A 850:177–185
Ohta K, Ohashi M, ** JY, Takeuchi T, Fujimoto C, Choi SH, Ryoo JJ, Lee KP (2003) J Chromatogr A 997:117–125
Zapała W, Kaczmarski K (2010) Acta Chromatographica 22:1–25
Tanaka K, Ding MY, Takahashi H, Helaleh MIH, Taoda H, Hu W, Hasebe K, Haddad PR, Mori M, Fritz JS, Sarzanini C (2002) Anal Chim Acta 474:31–35
Tanaka K, Ding MY, Helaleh MIH, Taoda H, Takahashi H, Hu W, Hasebe K, Haddad PR, Fritz JS, Sarzanini C (2002) J Chromatogr A 956:209–214
Helaleh MIH, Tanaka K, Mori M, Xu Q, Taoda H, Ding MY, Hu W, Hasebe K, Haddad PR (2003) J Chromatogr A 997:133–138
Helaleh MIH, Tanaka K, Mori M, Xu Q, Taoda H, Ding MY (2003) J Chromatogr A 997:139–144
Mori M, Itabashi H, Helaleh MIH, Kaczmarski K, Głod B, Kowalska T (2006) J Chromatogr A 1118:41–45
Zapała W, Kostka J, Kaczmarski K (2011) Acta Chromatographica 2:377–388
Mori M, Helaleh MIH, Xu Q, Hu W, Ikedo M, Ding MY, Taoda H, Tanaka K (2004) J Chromatogr A 1039:129–133
Widiastuti R, Haddad PR, Jackson PE (1992) J Chromatogr 602:43–50
Glod BK, Haddad PR, Alexander PW (1992) J Chromatogr 595:149–154
Watson JR, Crescuolo P, Matsui F (1971) J Pharm Sci 60:454–458
Hobl EL, Jilma B, Ebner J, Schmid RW (2013) Biomed Chromatogr. doi:10.1002/bmc.2846
Mullangi R, Sharma K, Srinivas NR (2012) Biomed Chromatogr 26:906–941
Bharathi DV, Hotha KK, Kolagatla PRR, Venkateswarlu V (2013) Biomed Chromatogr. doi:10.1002/bmc.2832
Shaw LH, Tsai TH (2012) J Chromatogr B 895:31–38
Wudarska E, Chrzescijanska E, Kusmierek E, Rynkowski J (2013) Electrochim Acta 93:189–194
Hansen SH, Jensen ME, Bjørnsdottir I (1998) J Pharm Biomed Anal 17:1155–1160
Šlais K (1989) J Chromatogr 469:223–229
Lee JH, Hyung S-W, Mun D-G, Jung H-J, Kim H, Lee H, Kim S-J, Park KS, Moore RJ, Smith RD, Lee S-W (2012) J Proteome Res 11:4373–4381
Myers RH, Montgomery DC, Anderson-Cook CM (2009) Response surface methodology: process and product optimization using designed experiments. Wiley, Hoboken
Lucie N, Hana V, Solich P (2012) Talanta 93:99–105
Hu W, Hasebe K, Reynolds DM, Haraguchi H (1997) Anal Chim Acta 353:143–149
Berky R, Fekete S, Fekete J (2012) Chromatographia 75:305–312
Fekete S, Berky R, Fekete J, Veuthey J-L, Guillarme D (2012) J Chromatogr A 1252:90–103
Acknowledgments
Funding for the UHPLC instrument was made possible primarily by NIH AREA and ARRA grants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mansour, F.R., Kirkpatrick, C.L. & Danielson, N.D. Separation of Aliphatic and Aromatic Carboxylic Acids by Conventional and Ultra High-Performance Ion-Exclusion Chromatography. Chromatographia 76, 603–609 (2013). https://doi.org/10.1007/s10337-013-2461-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-013-2461-3