Abstract
The electrocatalytic reduction of nitrate waste into ammonia allows both the removal of nitrate contaminants and an alternative production of ammonia compared to the classical Haber–Bosch industrial process. Ammonia is useful in agriculture for manufacturing fertilizers, and as a reagent in pharmaceuticals, metallurgy, explosives, and the textile industry; ammonia is also an energy carrier in the automobile industry for next-generation fuel cells. Here we review the nitrate-to-ammonia conversion by electrocatalysis of industrial and agricultural waste, with focus on catalysts, reaction intermediates, side reactions, and reaction conditions. Electron transfer is facilitated by electrocatalysts with transition metals having occupied d-orbitals with similar energy levels to that of the nitrate lowest unoccupied molecular orbital. Green electro-conversion using carbon-based materials is also discussed. Results show nitrate conversion from 53 to 99.8% and ammonia selectivity from 70 to 97.4%.

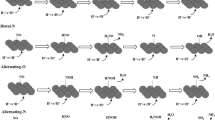
Reproduced from reference (Hasan et al. 2021) with permission from MDPI



Reproduced from reference (Jia et al. 2020) with permission from American Chemical Society

Reproduced from reference (Yin et al. 2021) with permission from Elsevier

Reproduced from reference (Jia et al. 2021) with permission from American Chemical Society. NSs—nanoshuttles; PANSs—porous and amorphous nanoshuttles


Similar content being viewed by others
References
Balaji TE, Tanaya Das H, Maiyalagan T (2021) Recent trends in bimetallic oxides and their composites as electrode materials for supercapacitor applications. ChemElectroChem 8(10):1723–1746. https://doi.org/10.1002/celc.202100098
Barrera L, Bala Chandran R (2021) Harnessing photoelectrochemistry for wastewater nitrate treatment coupled with resource recovery. ACS Sustain Chem Eng 9(10):3688–3701. https://doi.org/10.1021/acssuschemeng.0c07935
Binaeian E, Li Y, Yuan D (2021) Improving ammonia uptake performance of zirconium-based metal-organic frameworks through open metal site insertion strategy. Chem Eng J 421:129655. https://doi.org/10.1016/j.cej.2021.129655
Casella IG, Contursi M (2014) Highly dispersed rhodium particles on multi-walled carbon nanotubes for the electrochemical reduction of nitrate and nitrite ions in acid medium. Electrochim Acta 138:447–453. https://doi.org/10.1016/j.electacta.2014.05.125
Casella IG, Contursi M, Toniolo R (2014) A non-enzymatic carbohydrate sensor based on multiwalled carbon nanotubes modified with adsorbed active gold particles. Electroanalysis 26(5):988–995. https://doi.org/10.1002/elan.201300644
Chen M, Bi J, Huang X, Wang T, Wang Z, Hao H (2021) Bi2O3 nanosheets arrays in-situ decorated on carbon cloth for efficient electrochemical reduction of nitrate. Chemosphere 278:130386. https://doi.org/10.1016/j.chemosphere.2021.130386
Cheng H, Ding LX, Chen GF, Zhang L, Xue J, Wang H (2018) Molybdenum carbide nanodots enable efficient electrocatalytic nitrogen fixation under ambient conditions. Adv Mater 30(46):e1803694. https://doi.org/10.1002/adma.201803694
Choi H, Peters AW, Noh H, Gallington LC, Platero-Prats AE, DeStefano MR, Rimoldi M, Goswami S, Chapman KW, Farha OK, Hupp JT (2019) Vapor-phase fabrication and condensed-phase application of a MOF-node-supported iron thiolate photocatalyst for nitrate conversion to ammonium. ACS Appl Energy Mater 2(12):8695–8700. https://doi.org/10.1021/acsaem.9b01664
Das HT, Balaji TE, Dutta S, Das N, Maiyalagan T (2022) Recent advances in MXene as electrocatalysts for sustainable energy generation: a review on surface engineering and compositing of MXene. Int J Energy Res. https://doi.org/10.1002/er.7847
de Groot MT, Koper MTM (2004) The influence of nitrate concentration and acidity on the electrocatalytic reduction of nitrate on platinum. J Electroanal Chem 562(1):81–94. https://doi.org/10.1016/j.jelechem.2003.08.011
Duan W, Li G, Lei Z, Zhu T, Xue Y, Wei C, Feng C (2019) Highly active and durable carbon electrocatalyst for nitrate reduction reaction. Water Res 161:126–135. https://doi.org/10.1016/j.watres.2019.05.104
Fu Y, Richardson P, Li K, Yu H, Yu B, Donne S, Kisi E, Ma T (2020) Transition metal aluminum boride as a new candidate for ambient-condition electrochemical ammonia synthesis. Nano-Micro Lett 12(1):65. https://doi.org/10.1007/s40820-020-0400-z
Fu W, Hu Z, Zheng Y, Su P, Zhang Q, Jiao Y, Zhou M (2022) Tuning mobility of intermediate and electron transfer to enhance electrochemical reduction of nitrate to ammonia on Cu2O/Cu interface. Chem Eng J 433:133680. https://doi.org/10.1016/j.cej.2021.133680
Furtado AMB, Liu J, Wang Y, LeVan MD (2011) Mesoporous silica–metal organic composite: synthesis, characterization, and ammonia adsorption. J Mater Chem 21(18):6698–6706. https://doi.org/10.1039/c1jm10451a
Gao R, Zhang G, Ru X, Xu C, Li M, Lin R, Wang Z (2021a) Morphology control of metal-organic frameworks by Co-competitive coordination strategy for low-temperature selective catalytic reduction of NO with NH3. J Solid State Chem 297:122031. https://doi.org/10.1016/j.jssc.2021.122031
Gao Z, Lai Y, Tao Y, **ao L, Zhang L, Luo F (2021b) Constructing well-defined and robust Th-MOF-supported single-site copper for production and storage of ammonia from electroreduction of nitrate. ACS Cent Sci 7(6):1066–1072. https://doi.org/10.1021/acscentsci.1c00370
Garcia-Gil J (1996) Brunet RC, Garcia-Gil LJ. Sulfide-induced dissimilatory nitrate reduction to ammonia in anaerobic freshwater sediments. FEMS Microbiol Ecol 21: 131–138. FEMS Microbiol Ecol 21:131–138
Garcia-Segura S, Lanzarini-Lopes M, Hristovski K, Westerhoff P (2018) Electrocatalytic reduction of nitrate: fundamentals to full-scale water treatment applications. Appl Catal B Environ 236:546–568. https://doi.org/10.1016/j.apcatb.2018.05.041
Gootzen JFE, Peeters PGJM, Dukers JMB, Lefferts L, Visscher W, van Veen JAR (1997) The electrocatalytic reduction of NO3− on Pt, Pd and Pt + Pd electrodes activated with Ge. J Electroanal Chem 434(1–2):171–183. https://doi.org/10.1016/s0022-0728(97)00093-4
Guo Y, Zhang R, Zhang S, Zhao Y, Yang Q, Huang Z, Dong B, Zhi C (2021) Pd do**-weakened intermediate adsorption to promote electrocatalytic nitrate reduction on TiO2 nanoarrays for ammonia production and energy supply with zinc–nitrate batteries. Energy Environ Sci 14(7):3938–3944. https://doi.org/10.1039/D1EE00806D
Hasan MH, Mahlia TM, Mofijur M, Rizwanul Fattah IM, Handayani F, Ong HC, Silitonga AS (2021) A comprehensive review on the recent development of ammonia as a renewable energy carrier. Energies. https://doi.org/10.3390/en14133732
He W, Zhang J, Dieckhöfer S, Varhade S, Brix AC, Lielpetere A, Seisel S, Junqueira JRC, Schuhmann W (2022) Splicing the active phases of copper/cobalt-based catalysts achieves high-rate tandem electroreduction of nitrate to ammonia. Nat Commun 13(1):1129. https://doi.org/10.1038/s41467-022-28728-4
Hong Q-L, Zhou J, Zhai Q-G, Jiang Y-C, Hu M-C, **ao X, Li S-N, Chen Y (2021a) Cobalt phosphide nanorings towards efficient electrocatalytic nitrate reduction to ammonia. Chem Commun 57(88):11621–11624. https://doi.org/10.1039/D1CC04952F
Hong QL, Zhou J, Zhai QG, Jiang YC, Hu MC, **ao X, Li SN, Chen Y (2021b) Cobalt phosphide nanorings towards efficient electrocatalytic nitrate reduction to ammonia. Chem Commun (camb) 57(88):11621–11624. https://doi.org/10.1039/d1cc04952f
Hu Q, Qin Y, Wang X, Zheng H, Gao K, Yang H, Zhang P, Shao M, He C (2021) Grain boundaries engineering of hollow copper nanoparticles enables highly efficient ammonia Electrosynthesis from Nitrate. CCS Chem. https://doi.org/10.31635/ccschem.021.202101042
Huang Y, Long J, Wang Y, Meng N, Yu Y, Lu S, **ao J, Zhang B (2021) Engineering nitrogen vacancy in polymeric carbon nitride for nitrate electroreduction to ammonia. ACS Appl Mater Interfaces 13(46):54967–54973. https://doi.org/10.1021/acsami.1c15206
Humphreys L, Wilson I, McAteer D, Pons J (2018) Development of metal-organic framework (MOF) sensors for landmine detection. https://doi.org/10.5162/IMCS2018/P1AP.8
Huo S, Yang S, Niu Q, Song Z, Yang F, Song L (2020) Fabrication of porous configurated Ni2P/Ni foam catalyst and its boosted properties for ph-universal hydrogen evolution reaction and efficient nitrate reduction. ChemCatChem 12(18):4600–4610. https://doi.org/10.1002/cctc.2020a00426
Huo S, Yang S, Niu Q, Yang F, Song L (2020) Synthesis of functional Ni2P/CC catalyst and the robust performances in hydrogen evolution reaction and nitrate reduction. Int J Hydrog Energy 45(7):4015–4025. https://doi.org/10.1016/j.ijhydene.2019.11.210
Jia R, Wang Y, Wang C, Ling Y, Yu Y, Zhang B (2020) Boosting selective nitrate electroreduction to ammonium by constructing oxygen vacancies in TiO2. ACS Catal 10(6):3533–3540. https://doi.org/10.1021/acscatal.9b05260
Jia Y, Ji YG, Xue Q, Li FM, Zhao GT, ** PJ, Li SN, Chen Y (2021) Efficient nitrate-to-ammonia electroreduction at cobalt phosphide nanoshuttles. ACS Appl Mater Interfaces 13(38):45521–45527. https://doi.org/10.1021/acsami.1c12512
Jiang G, Peng M, Hu L, Ouyang J, Lv X, Yang Z, Liang X, Liu Y, Liu H (2022) Electron-deficient Cuδ+ stabilized by interfacial Cu–O-Al bonding for accelerating electrocatalytic nitrate conversion. Chem Eng J 435:134853. https://doi.org/10.1016/j.cej.2022a.134853
Jiang M, Su J, Song X, Zhang P, Zhu M, Qin L, Tie Z, Zuo J-L, ** Z (2022b) Interfacial reduction nucleation of noble metal nanodots on redox-active metal–organic frameworks for high-efficiency electrocatalytic conversion of nitrate to ammonia. Nano Lett 22(6):2529–2537. https://doi.org/10.1021/acs.nanolett.2c00446
Jiang M, Su J, Song X, Zhang P, Zhu M, Qin L, Tie Z, Zuo JL, ** Z (2022c) Interfacial reduction nucleation of noble metal nanodots on redox-active metal-organic frameworks for high-efficiency electrocatalytic conversion of nitrate to ammonia. Nano Lett 22(6):2529–2537. https://doi.org/10.1021/acs.nanolett.2c00446
Kibsgaard J, Tsai C, Chan K, Benck JD, Nørskov JK, Abild-Pedersen F, Jaramillo TF (2015) Designing an improved transition metal phosphide catalyst for hydrogen evolution using experimental and theoretical trends. Energy Environ Sci 8(10):3022–3029. https://doi.org/10.1039/c5ee02179k
Lan Y, Chen J, Zhang H, Zhang W-x, Yang J (2020) Fe/Fe3C nanoparticle-decorated N-doped carbon nanofibers for improving the nitrogen selectivity of electrocatalytic nitrate reduction. J Mater Chem A 8(31):15853–15863. https://doi.org/10.1039/D0TA02317E
Laursen AB, Patraju KR, Whitaker MJ, Retuerto M, Sarkar T, Yao N, Ramanujachary KV, Greenblatt M, Dismukes GC (2015) Nanocrystalline Ni5P4: a hydrogen evolution electrocatalyst of exceptional efficiency in both alkaline and acidic media. Energy Environ Sci 8(3):1027–1034. https://doi.org/10.1039/c4ee02940b
Lee J, Kwak SY (2018) Mn-doped maghemite (gamma-Fe2O3) from Metal-organic framework accompanying redox reaction in a bimetallic system: the structural phase transitions and catalytic activity toward NOx removal. ACS Omega 3(3):2634–2640. https://doi.org/10.1021/acsomega.7b01865
Li C, Shi Y, Zhang H, Zhao Q, Xue F, Li X (2016a) Cu-BTC metal-organic framework as a novel catalyst for low temperature selective catalytic reduction (SCR) of NO by NH3: promotional effect of activation temperature. Integr Ferroelectr 172(1):169–179. https://doi.org/10.1080/10584587.2016.1177385
Li L, Tang C, Cui X, Zheng Y, Wang X, Xu H, Zhang S, Shao T, Davey K, Qiao S-Z (2021) Efficient nitrogen fixation to ammonia through integration of plasma oxidation with electrocatalytic reduction. Angewandte Chem Int Edition 60(25):14131–14137. https://doi.org/10.1002/anie.2021a04394
Li X, Gu Y, Wu S, Chen S, Quan X, Yu H (2021b) Selective reduction of nitrate to ammonium over charcoal electrode derived from natural wood. Chemosphere 285:131501. https://doi.org/10.1016/j.chemosphere.2021.131501
Li Y, Ma J, Waite TD, Hoffmann MR, Wang Z (2021c) Development of a mechanically flexible 2D-MXene membrane cathode for selective electrochemical reduction of nitrate to N2: mechanisms and implications. Environ Sci Technol 55(15):10695–10703. https://doi.org/10.1021/acs.est.1c00264
Li Y, Yamaguchi A, Yamamoto M, Takai K, Nakamura R (2016b) Molybdenum sulfide: a bioinspired electrocatalyst for dissimilatory ammonia synthesis with geoelectrical current. J Phys Chem C 121(4):2154–2164. https://doi.org/10.1021/acs.jpcc.6b08343
Li Z, Wen G, Liang J, Li T, Luo Y, Kong Q, Shi X, Asiri AM, Liu Q, Sun X (2021d) High-efficiency nitrate electroreduction to ammonia on electrodeposited cobalt–phosphorus alloy film. Chem Commun 57(76):9720–9723. https://doi.org/10.1039/D1CC02612G
Liang J, Hu W-F, Song B, Mou T, Zhang L, Luo Y, Liu Q, Alshehri AA, Hamdy MS, Yang L-M, Sun X (2022) Efficient nitric oxide electroreduction toward ambient ammonia synthesis catalyzed by a CoP nanoarray. Inorgan Chem Front 9(7):1366–1372. https://doi.org/10.1039/d2qi00002d
Liu H, Park J, Chen Y, Qiu Y, Cheng Y, Srivastava K, Gu S, Shanks BH, Roling LT, Li W (2021a) Electrocatalytic nitrate reduction on oxide-derived silver with tunable selectivity to nitrite and ammonia. ACS Catal 11(14):8431–8442. https://doi.org/10.1021/acscatal.1c01525
Liu T, Li A, Wang C, Zhou W, Liu S, Guo L (2018) Interfacial electron transfer of Ni2 P-NiP2 polymorphs inducing enhanced electrochemical properties. Adv Mater 30(46):e1803590. https://doi.org/10.1002/adma.201803590
Liu Y, Deng B, Li K, Wang H, Sun Y, Dong F (2022a) Metal-organic framework derived carbon-supported bimetallic copper-nickel alloy electrocatalysts for highly selective nitrate reduction to ammonia. J Colloid Interface Sci 614:405–414. https://doi.org/10.1016/j.jcis.2022.01.127
Liu Z, Wang C, Chen C, Li C, Guo C (2021) Selective electroreduction of nitrate to ammonia with high Faradaic efficiency on nanocrystalline silver. Electrochem Commun 131:107121. https://doi.org/10.1016/j.elecom.2021b.107121
Lu X, Song H, Cai J, Lu S (2021) Recent development of electrochemical nitrate reduction to ammonia: a mini review. Electrochem Commun 129:107094. https://doi.org/10.1016/j.elecom.2021.107094
Madhura L, Singh S, Kanchi S, Sabela M, Bisetty K (2019) Nanotechnology-based water quality management for wastewater treatment. Environ Chem Lett 17(1):65–121. https://doi.org/10.1007/s10311-018-0778-8
Martínez J, Ortiz A, Ortiz I (2017) State-of-the-art and perspectives of the catalytic and electrocatalytic reduction of aqueous nitrates. Appl Catal B Environ 207:42–59. https://doi.org/10.1016/j.apcatb.2017.02.016
McEnaney JM, Crompton JC, Callejas JF, Popczun EJ, Read CG, Lewis NS, Schaak RE (2014) Electrocatalytic hydrogen evolution using amorphous tungsten phosphide nanoparticles. Chem Commun (camb) 50(75):11026–11028. https://doi.org/10.1039/c4cc04709e
Min B, Gao Q, Yan Z, Han X, Hosmer K, Campbell A, Zhu H (2021) Powering the remediation of the nitrogen cycle: progress and perspectives of electrochemical nitrate reduction. Ind Eng Chem Res 60(41):14635–14650. https://doi.org/10.1021/acs.iecr.1c03072
Morin-Crini N, Lichtfouse E, Fourmentin M, Ribeiro ARL, Noutsopoulos C, Mapelli F, Fenyvesi É, Vieira MGA, Picos-Corrales LA, Moreno-Piraján JC, Giraldo L, Sohajda T, Huq MM, Soltan J, Torri G, Magureanu M, Bradu C, Crini G (2022) Removal of emerging contaminants from wastewater using advanced treatments. Rev Environ Chem Lett 20(2):1333–1375. https://doi.org/10.1007/s10311-021-01379-5
Mounfield WP, Taborga Claure M, Agrawal PK, Jones CW, Walton KS (2016) Synergistic effect of mixed oxide on the adsorption of ammonia with metal–organic frameworks. Ind Eng Chem Res 55(22):6492–6500. https://doi.org/10.1021/acs.iecr.6b01045
Mudhoo A, Paliya S, Goswami P, Singh M, Lofrano G, Carotenuto M, Carraturo F, Libralato G, Guida M, Usman M, Kumar S (2020) Fabrication, functionalization and performance of doped photocatalysts for dye degradation and mineralization: a review. Environ Chem Lett 18(6):1825–1903. https://doi.org/10.1007/s10311-020-01045-2
Petit C, Bandosz TJ (2009) MOF-graphite oxide composites: combining the uniqueness of graphene layers and metal-organic frameworks. Adv Mater 21(46):4753–4757. https://doi.org/10.1002/adma.200901581
Pintar A, Batista J, Muševič I (2004) Palladium-copper and palladium-tin catalysts in the liquid phase nitrate hydrogenation in a batch-recycle reactor. Appl Catal B 52(1):49–60. https://doi.org/10.1016/j.apcatb.2004.02.019
Popczun EJ, McKone JR, Read CG, Biacchi AJ, Wiltrout AM, Lewis NS, Schaak RE (2013) Nanostructured nickel phosphide as an electrocatalyst for the hydrogen evolution reaction. J Am Chem Soc 135(25):9267–9270. https://doi.org/10.1021/ja403440e
Prashantha Kumar TKM, Mandlimath TR, Sangeetha P, Revathi SK, Ashok Kumar SK (2018) Nanoscale materials as sorbents for nitrate and phosphate removal from water. Environ Chem Lett 16(2):389–400. https://doi.org/10.1007/s10311-017-0682-7
Qin J, Wu K, Chen L, Wang X, Zhao Q, Liu B, Ye Z (2022) Achieving high selectivity for nitrate electrochemical reduction to ammonia over MOF-supported RuxOy clusters. J Mater Chem A 10(8):3963–3969. https://doi.org/10.1039/D1TA09441F
Ren T, Ren K, Wang M, Liu M, Wang Z, Wang H, Li X, Wang L, Xu Y (2021) Concave-convex surface oxide layers over copper nanowires boost electrochemical nitrate-to-ammonia conversion. Chem Eng J 426:130759. https://doi.org/10.1016/j.cej.2021.130759
Roberts EJ, Read CG, Lewis NS, Brutchey RL (2018) Phase directing ability of an ionic liquid solvent for the synthesis of HER-active Ni2P nanocrystals. ACS Appl Energy Mater 1(5):1823–1827. https://doi.org/10.1021/acsaem.8b00213
Sato T, Ki S, Fujishiro Y, Yoshioka T, Okuwaki A (1999) Photochemical reduction of nitrate to ammonia using layered hydrous titanate/cadmium sulphide nanocomposites. J Chem Technol Biotechnol 67(4):345–349. https://doi.org/10.1002/(sici)1097-4660(199612)67:4%3c345::Aid-jctb586%3e3.0.Co;2-#
Shipman MA, Symes MD (2017) Recent progress towards the electrosynthesis of ammonia from sustainable resources. Catal Today 286:57–68. https://doi.org/10.1016/j.cattod.2016.05.008
Stern L-A, Feng L, Song F, Hu X (2015) Ni2P as a Janus catalyst for water splitting: the oxygen evolution activity of Ni2P nanoparticles. Energy Environ Sci 8(8):2347–2351. https://doi.org/10.1039/c5ee01155h
Sun T, Zhang G, Xu D, Lian X, Li H, Chen W, Su C (2019) Defect chemistry in 2D materials for electrocatalysis. Mater Today Energy 12:215–238. https://doi.org/10.1016/j.mtener.2019.01.004
Valera-Medina A, **ao H, Owen-Jones M, David WIF, Bowen PJ (2018) Ammonia for power. Prog Energy Combust Sci 69:63–102. https://doi.org/10.1016/j.pecs.2018.07.001
Velusamy K, Periyasamy S, Kumar PS, Vo D-VN, Sindhu J, Sneka D, Subhashini B (2021) Advanced techniques to remove phosphates and nitrates from waters: a review. Environ Chem Lett 19(4):3165–3180. https://doi.org/10.1007/s10311-021-01239-2
Wan X, Guo W, Dong X, Wu H, Sun X, Chu M, Han S, Zhai J, **a W, Jia S, He M, Han B (2022) Boosting nitrate electroreduction to ammonia on NbOx via constructing oxygen vacancies. Green Chem 24(3):1090–1095. https://doi.org/10.1039/D1GC04483D
Wang C, Ye F, Shen J, Xue K-H, Zhu Y, Li C (2022a) In situ loading of Cu2O active sites on island-like copper for efficient electrochemical reduction of nitrate to ammonia. ACS Appl Mater Interfaces 14(5):6680–6688. https://doi.org/10.1021/acsami.1c21691
Wang F, Ding Q, Bai Y, Bai H, Wang S, Fan W (2022b) Fabrication of an amorphous metal oxide/p-BiVO4 photocathode: understanding the role of entropy for reducing nitrate to ammonia. Inorgan Chem Front 9(4):805–813. https://doi.org/10.1039/d1qi01472b
Wang H, Mao Q, Ren T, Zhou T, Deng K, Wang Z, Li X, Xu Y, Wang L (2021a) Synergism of interfaces and defects: Cu/oxygen vacancy-rich Cu-Mn3O4 heterostructured ultrathin nanosheet arrays for selective nitrate electroreduction to ammonia. ACS Appl Mater Interfaces 13(37):44733–44741. https://doi.org/10.1021/acsami.1c11249
Wang J, Cai C, Wang Y, Yang X, Wu D, Zhu Y, Li M, Gu M, Shao M (2021b) Electrocatalytic reduction of nitrate to ammonia on low-cost ultrathin CoOx nanosheets. ACS Catal 11(24):15135–15140. https://doi.org/10.1021/acscatal.1c03918
Wang J, Feng T, Chen J, Ramalingam V, Li Z, Kabtamu DM, He J-H, Fang X (2021) Electrocatalytic nitrate/nitrite reduction to ammonia synthesis using metal nanocatalysts and bio-inspired metalloenzymes. Nano Energy 86:106088. https://doi.org/10.1016/j.nanoen.2021c.106088
Wang P, Zhao H, Sun H, Yu H, Chen S, Quan X (2014) Porous metal–organic framework MIL-100(Fe) as an efficient catalyst for the selective catalytic reduction of NOx with NH3. RSC Adv 4(90):48912–48919. https://doi.org/10.1039/c4ra07028c
Wang Q, Astruc D (2020) State of the Art and Prospects in Metal-organic framework (MOF)-based and MOF-derived nanocatalysis. Chem Rev 120(2):1438–1511. https://doi.org/10.1021/acs.chemrev.9b00223
Wang Y, Li H, Zhou W, Zhang X, Zhang B, Yu Y (2022) Structurally disordered RuO2 nanosheets with rich oxygen vacancies for enhanced nitrate electroreduction to ammonia. Angewandte Chem Int Edition. https://doi.org/10.1002/anie.2022c02604
Wang Y, Wang C, Li M, Yu Y, Zhang B (2021d) Nitrate electroreduction: mechanism insight, in situ characterization, performance evaluation, and challenges. Chem Soc Rev 50(12):6720–6733. https://doi.org/10.1039/D1CS00116G
Wang Y, Xu A, Wang Z, Huang L, Li J, Li F, Wicks J, Luo M, Nam D-H, Tan C-S, Ding Y, Wu J, Lum Y, Dinh C-T, Sinton D, Zheng G, Sargent EH (2020a) Enhanced nitrate-to-ammonia activity on copper–nickel alloys via tuning of intermediate adsorption. J Am Chem Soc 142(12):5702–5708. https://doi.org/10.1021/jacs.9b13347
Wang Y, Zhang L, Niu Y, Fang D, Wang J, Su Q, Wang C (2021e) Boosting NH3 production from nitrate electroreduction via electronic structure engineering of Fe3C nanoflakes. Green Chem 23(19):7594–7608. https://doi.org/10.1039/D1GC01913A
Wang Y, Zhou W, Jia R, Yu Y, Zhang B (2020) Unveiling the activity origin of a copper-based electrocatalyst for selective nitrate reduction to ammonia. Angewandte Chem Int Edition 59(13):5350–5354. https://doi.org/10.1002/anie.201915992
Wang Z, Chen C, Liu H, Hrynshpan D, Savitskaya T, Chen J, Chen J (2019) Effects of carbon nanotube on denitrification performance of Alcaligenes sp. TB: promotion of electron generation, transportation and consumption. Ecotoxicol Environ Saf 183:109507. https://doi.org/10.1016/j.ecoenv.2019.109507
Wang Z, Chen C, Liu H, Hrynshpan D, Savitskaya T, Chen J, Chen J (2020) Enhanced denitrification performance of Alcaligenes sp. TB by Pd stimulating to produce membrane adaptation mechanism coupled with nanoscale zero-valent iron. Sci Total Environ 708:135063. https://doi.org/10.1016/j.scitotenv.2019.135063
Wang Z, Dai L, Yao J, Guo T, Hrynsphan D, Tatsiana S, Chen J (2021f) Enhanced adsorption and reduction performance of nitrate by Fe-Pd-Fe3O4 embedded multi-walled carbon nanotubes. Chemosphere 281:130718. https://doi.org/10.1016/j.chemosphere.2021.130718
Wang Z, Fu W, Hu L, Zhao M, Guo T, Hrynsphan D, Tatsiana S, Chen J (2021g) Improvement of electron transfer efficiency during denitrification process by Fe-Pd/multi-walled carbon nanotubes: Possessed redox characteristics and secreted endogenous electron mediator. Sci Total Environ 781:146686. https://doi.org/10.1016/j.scitotenv.2021.146686
Wang Z, Ortiz EM, Goldsmith BR, Singh N (2021h) Comparing electrocatalytic and thermocatalytic conversion of nitrate on platinum–ruthenium alloys. Catal Sci Technol 11(21):7098–7109. https://doi.org/10.1039/D1CY01075A
Wang Z, Young SD, Goldsmith BR, Singh N (2021) Increasing electrocatalytic nitrate reduction activity by controlling adsorption through PtRu alloying. J Catal 395:143–154. https://doi.org/10.1016/j.jcat.2020.12.031
Wen G, Liang J, Liu Q, Li T, An X, Zhang F, Alshehri AA, Alzahrani KA, Luo Y, Kong Q, Sun X (2022a) Ambient ammonia production via electrocatalytic nitrite reduction catalyzed by a CoP nanoarray. Nano Res 15(2):972–977. https://doi.org/10.1007/s12274-021-3583-9
Wen G, Liang J, Zhang L, Li T, Liu Q, An X, Shi X, Liu Y, Gao S, Asiri AM, Luo Y, Kong Q, Sun X (2022b) Ni2P nanosheet array for high-efficiency electrohydrogenation of nitrite to ammonia at ambient conditions. J Colloid Interface Sci 606(Pt 2):1055–1063. https://doi.org/10.1016/j.jcis.2021.08.050
Wu J, Li JH, Yu YX (2021a) Theoretical exploration of electrochemical nitrate reduction reaction activities on transition-metal-doped h-BP. J Phys Chem Lett 12(16):3968–3975. https://doi.org/10.1021/acs.jpclett.1c00855
Wu J, Yu Y-X (2021) Highly selective electroreduction of nitrate to ammonia on a Ru-doped tetragonal Co2P monolayer with low-limiting overpotential. Catal Sci Technol 11(21):7160–7170. https://doi.org/10.1039/d1cy01217g
Wu Z-Y, Karamad M, Yong X, Huang Q, Cullen DA, Zhu P, **a C, **ao Q, Shakouri M, Chen F-Y, Kim JY, **a Y, Heck K, Hu Y, Wong MS, Li Q, Gates I, Siahrostami S, Wang H (2021b) Electrochemical ammonia synthesis via nitrate reduction on Fe single atom catalyst. Nat Commun 12(1):2870. https://doi.org/10.1038/s41467-021-23115-x
**ang X, Guo L, Wu X, Ma X, **a Y (2012) Urea formation from carbon dioxide and ammonia at atmospheric pressure. Environ Chem Lett 10(3):295–300. https://doi.org/10.1007/s10311-012-0366-2
Xu S, Kwon H-Y, Ashley DC, Chen C-H, Jakubikova E, Smith JM (2019) Intramolecular hydrogen bonding facilitates electrocatalytic reduction of nitrite in aqueous solutions. Inorg Chem 58(14):9443–9451. https://doi.org/10.1021/acs.inorgchem.9b01274
Xu W, Yu C, Chen J, Liu Z (2022) Electrochemical hydrogenation of biomass-based furfural in aqueous media by Cu catalyst supported on N-doped hierarchically porous carbon. Appl Catal B Environ 305:121062. https://doi.org/10.1016/j.apcatb.2022a.121062
Xu Y, Ren K, Ren T, Wang M, Liu M, Wang Z, Li X, Wang L, Wang H (2021) Cooperativity of Cu and Pd active sites in CuPd aerogels enhances nitrate electroreduction to ammonia. Chem Commun 57(61):7525–7528. https://doi.org/10.1039/D1CC02105B
Xu Y, Ren K, Ren T, Wang M, Wang Z, Li X, Wang L, Wang H (2022) Ultralow-content Pd in-situ incorporation mediated hierarchical defects in corner-etched Cu2O octahedra for enhanced electrocatalytic nitrate reduction to ammonia. Appl Catal B Environ 306:121094. https://doi.org/10.1016/j.apcatb.2022b.121094
Xu Y, Wu R, Zhang J, Shi Y, Zhang B (2013) Anion-exchange synthesis of nanoporous FeP nanosheets as electrocatalysts for hydrogen evolution reaction. Chem Commun (camb) 49(59):6656–6658. https://doi.org/10.1039/c3cc43107j
Yao Q, Chen J, **ao S, Zhang Y, Zhou X (2021) Selective electrocatalytic reduction of nitrate to ammonia with nickel phosphide. ACS Appl Mater Interfaces 13(26):30458–30467. https://doi.org/10.1021/acsami.0c22338
Ye S, Chen Z, Zhang G, Chen W, Peng C, Yang X, Zheng L, Li Y, Ren X, Cao H, Xue D, Qiu J, Zhang Q, Liu J (2022) Elucidating the activity, mechanism and application of selective electrosynthesis of ammonia from nitrate on cobalt phosphide. Energy Environ Sci 15(2):760–770. https://doi.org/10.1039/d1ee03097c
Yin H, Chen Z, **ong S, Chen J, Wang C, Wang R, Kuwahara Y, Luo J, Yamashita H, Peng Y, Li J (2021) Alloying effect-induced electron polarization drives nitrate electroreduction to ammonia. Chem Catal 1(5):1088–1103. https://doi.org/10.1016/j.checat.2021.08.014
Yin Z, Liu J, Jiang L, Chu J, Yang T, Kong A (2022) Semi-enclosed Cu nanoparticles with porous nitrogen-doped carbon shells for efficient and tolerant nitrate electroreduction in neutral condition. Electrochim Acta 404:139585. https://doi.org/10.1016/j.electacta.2021.139585
Yu Y, Wang C, Yu Y, Wang Y, Zhang B (2020) Promoting selective electroreduction of nitrates to ammonia over electron-deficient Co modulated by rectifying Schottky contacts. SCIENCE CHINA Chem 63(10):1469–1476. https://doi.org/10.1007/s11426-020-9795-x
Zhang H, Wang G, Wang C, Liu Y, Yang Y, Wang C, Jiang W, Fu L, Xu J (2022a) CoP nanowires on carbon cloth for electrocatalytic NOx− reduction to ammonia. J Electroanal Chem 910:116171. https://doi.org/10.1016/j.jelechem.2022.116171
Zhang X, Shen B, Shen F, Zhang X, Si M, Yuan P (2017) The behavior of the manganese-cerium loaded metal-organic framework in elemental mercury and NO removal from flue gas. Chem Eng J 326:551–560. https://doi.org/10.1016/j.cej.2017.05.128
Zhang X, Wang Y, Liu C, Yu Y, Lu S, Zhang B (2021) Recent advances in non-noble metal electrocatalysts for nitrate reduction. Chem Eng J 403:126269. https://doi.org/10.1016/j.cej.2020.126269
Zhang Y, Chen X, Wang W, Yin L, Crittenden JC (2022) Electrocatalytic nitrate reduction to ammonia on defective Au1Cu (111) single-atom alloys. Appl Catal B Environ 310:121346. https://doi.org/10.1016/j.apcatb.2022b.121346
Acknowledgements
The authors acknowledge the financial support from National Research Foundation of Korea (NRF), (2022R1A2C2010686, 2019H1D3A1A01071209, 2021R1I1A1A01060380), and Korea Basic Science Institute (National research Facilities and Equipment Center) grant funded by the Ministry of Education. (No. 2019R1A6C1010042, 2021R1A6C103A427). This work is also supported by CAPES-PRINT (Grant 88881.311799/2018-01) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Finance Code 001). E.S.F.C is grateful to CAPES for the individual fellowship granted in support of their research. The author, Arun Prasad Murthy acknowledges VIT for providing VIT SEED GRANT (No. SG20210143) for carrying out this research work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Theerthagiri, J., Park, J., Das, H.T. et al. Electrocatalytic conversion of nitrate waste into ammonia: a review. Environ Chem Lett 20, 2929–2949 (2022). https://doi.org/10.1007/s10311-022-01469-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-022-01469-y