Abstract
Background/Aims
Fibroblast growth factor 23 (FGF23) and soluble α-Klotho are emerging potential biomarkers of phosphorus and vitamin D metabolism which change in concentration in early chronic kidney disease (CKD) in order to maintain normal phosphorus levels. Tubular reabsorption of phosphate (TRP) has been commonly used to assess renal tubular phosphate transport. The aim of this study was to evaluate the usefulness of TRP as a surrogate marker of parameters of CKD-mineral bone disease (CKD-MBD) in CKD.
Methods
A cross-sectional study was performed in 93 stable patients with predialysis CKD stage 1–5. In all patients, TRP, estimated glomerular filtration rate (eGFR), calcium, phosphate, intact parathyroid hormone (iPTH), 25-hydroxyvitamin D, serum FGF23 and urine soluble α-Klotho levels were measured.
Results
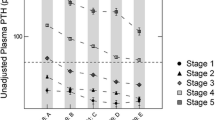
As renal function declined, TRP significantly decreased (P < 0.001; r = 0.763) and both iPTH and serum FGF23 increased (P < 0.001; r = −0.598, P < 0.001; r = −0.453, respectively). The prevalence of hyperphosphatemia, secondary hyperparathyroidism, FGF23 excess and abnormal TRP increased progressively with declining eGFR. Although TRP level changed later than FGF23, abnormal levels of both TRP and FGF23 were observed earlier than changes in iPTH and serum phosphate. Decreased TRP was found to be independently associated with decreased eGFR and increased iPTH but was not associated with urine soluble α-Klotho or serum FGF23 level in multiple linear regression analysis.
Conclusion
TRP is a simple, useful and cost-saving surrogate marker of the assessment of altered mineral metabolism in CKD patients and can be used as an alternative to serum FGF23, especially for mild to moderate renal insufficiency.


Similar content being viewed by others
References
Isakova T, Gutierrez OM, Wolf M. A blueprint for randomized trials targeting phosphorus metabolism in chronic kidney disease. Kidney Int. 2009;76(7):705–16. doi:10.1038/ki.2009.246.
Gutierrez O, Isakova T, Rhee E, Shah A, Holmes J, Collerone G, et al. Fibroblast growth factor-23 mitigates hyperphosphatemia but accentuates calcitriol deficiency in chronic kidney disease. J Am Soc Nephrol JASN. 2005;16(7):2205–15. doi:10.1681/asn.2005010052.
Martin KJ, Gonzalez EA. Prevention and control of phosphate retention/hyperphosphatemia in CKD-MBD: what is normal, when to start, and how to treat? Clin J Am Soc Nephrol. 2011;6(2):440–6. doi:10.2215/cjn.05130610.
Huang CL, Moe OW. Klotho: a novel regulator of calcium and phosphorus homeostasis. Pflugers Arch. 2011;462(2):185–93. doi:10.1007/s00424-011-0950-5.
Hu MC, Shi M, Zhang J, Quinones H, Griffith C, Kuro-o M, et al. Klotho deficiency causes vascular calcification in chronic kidney disease. J Am Soc Nephrol JASN. 2011;22(1):124–36. doi:10.1681/asn.2009121311.
Shimamura Y, Hamada K, Inoue K, Ogata K, Ishihara M, Kagawa T, et al. Serum levels of soluble secreted alpha-Klotho are decreased in the early stages of chronic kidney disease, making it a probable novel biomarker for early diagnosis. Clin Exp Nephrol. 2012;16(5):722–9. doi:10.1007/s10157-012-0621-7.
Payne RB. Renal tubular reabsorption of phosphate (TmP/GFR): indications and interpretation. Ann Clin Biochem. 1998;35(Pt 2):201–6.
Walton Rj, Fau-Bijvoet OL, Bijvoet OL. Nomogram for derivation of renal threshold phosphate concentration. Lancet. 1975;2(7929):309–10.
Isakova T, Wahl P, Vargas GS, Gutierrez OM, Scialla J, **e H, et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 2011;79(12):1370–8. doi:10.1038/ki.2011.47.
Levin A, Bakris GL, Molitch M, Smulders M, Tian J, Williams LA, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 2007;71(1):31–8. doi:10.1038/sj.ki.5002009.
Isakova T, Gutierrez OM, Smith K, Epstein M, Keating LK, Juppner H, et al. Pilot study of dietary phosphorus restriction and phosphorus binders to target fibroblast growth factor 23 in patients with chronic kidney disease. Nephrol Dial Transplant. 2011;26(2):584–91. doi:10.1093/ndt/gfq419.
Oliveira RB, Cancela AL, Graciolli FG, Dos Reis LM, Draibe SA, Cuppari L, et al. Early control of PTH and FGF23 in normophosphatemic CKD patients: a new target in CKD-MBD therapy? Clin J Am Soc Nephrol. 2010;5(2):286–91. doi:10.2215/cjn.05420709.
Forster IC, Hernando N, Biber J, Murer H. Proximal tubular handling of phosphate: a molecular perspective. Kidney Int. 2006;70(9):1548–59. doi:10.1038/sj.ki.5001813.
Slatopolsky E, Robson AM, Elkan I, Bricker NS. Control of phosphate excretion in uremic man. J Clin Invest. 1968;47(8):1865–74. doi:10.1172/jci105877.
Quarles LD. Endocrine functions of bone in mineral metabolism regulation. J Clin Invest. 2008;118(12):3820–8. doi:10.1172/jci36479.
Quarles LD. Role of FGF23 in vitamin D and phosphate metabolism: implications in chronic kidney disease. Exp Cell Res. 2012;318(9):1040–8. doi:10.1016/j.yexcr.2012.02.027.
Kuro OM. Klotho, phosphate and FGF-23 in ageing and disturbed mineral metabolism. Nat Rev Nephrol. 2013;. doi:10.1038/nrneph.2013.111.
Liu S, Tang W, Zhou J, Stubbs JR, Luo Q, Pi M, et al. Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J Am Soc Nephrol JASN. 2006;17(5):1305–15. doi:10.1681/asn.2005111185.
Shimada T, Yamazaki Y, Takahashi M, Hasegawa H, Urakawa I, Oshima T, et al. Vitamin D receptor-independent FGF23 actions in regulating phosphate and vitamin D metabolism. Am J Physiol Renal Physiol. 2005;289(5):F1088–95. doi:10.1152/ajprenal.00474.2004.
Hu MC, Kuro-o M, Moe OW. Klotho and kidney disease. J Nephrol. 2010;23(Suppl 16):S136–44.
Komaba H, Koizumi M, Tanaka H, Takahashi H, Sawada K, Kakuta T, et al. Effects of cinacalcet treatment on serum soluble Klotho levels in haemodialysis patients with secondary hyperparathyroidism. Nephrol Dial Transplant. 2012;27(5):1967–9. doi:10.1093/ndt/gfr645.
Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, Isakova T, et al. FGF23 induces left ventricular hypertrophy. J Clin Invest. 2011;121(11):4393–408. doi:10.1172/jci46122.
Akimoto T, Yoshizawa H, Watanabe Y, Numata A, Yamazaki T, Takeshima E, et al. Characteristics of urinary and serum soluble Klotho protein in patients with different degrees of chronic kidney disease. BMC Nephrol. 2012;13:155. doi:10.1186/1471-2369-13-155.
Kuwahara N, Sasaki S, Kobara M, Nakata T, Tatsumi T, Irie H, et al. HMG-CoA reductase inhibition improves anti-aging klotho protein expression and arteriosclerosis in rats with chronic inhibition of nitric oxide synthesis. Int J Cardiol. 2008;123(2):84–90. doi:10.1016/j.ijcard.2007.02.029.
Lim SC, Liu JJ, Subramaniam T, Sum CF. Elevated circulating alpha-klotho by angiotensin II receptor blocker losartan is associated with reduction of albuminuria in type 2 diabetic patients. J Renin Angiotensin Aldosterone Syst JRAAS. 2013;. doi:10.1177/1470320313475905.
Acknowledgments
The authors would like to thank Professor S. W. Kim of the Division of Urology, The Catholic University of Korea and Professor B. S. Choi of Seogang University for their valuable discussions. This study was supported by a grant of the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (C·W.P; A111055).
Conflict of interest
The authors have declared that no conflicts of interest exist.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hong, Y.A., Lim, J.H., Kim, M.Y. et al. Assessment of tubular reabsorption of phosphate as a surrogate marker for phosphate regulation in chronic kidney disease. Clin Exp Nephrol 19, 208–215 (2015). https://doi.org/10.1007/s10157-014-0962-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-014-0962-5



