Abstract
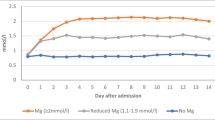
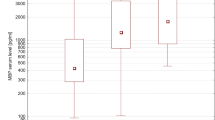
Magnesium, one of the essential trace elements, plays important roles in maintaining both normal cellular and body functions. S100 calcium-binding protein B (S100B) has been used as a marker of glial damage in several neurological disorders. Thirty patients with ruptured intracranial aneurysms treated by clip** are included. The patients were randomized within 4 days after the attack of hemorrhage. The patients were divided into two groups with 15 patients in each group. Group I received magnesium infusion within 4 days. Group II is the control group. World Federation of Neurological Surgeons, Fisher, and Glasgow outcome scores are evaluated at an intensive care unit in addition to 3 months clinical follow-up evaluation. Samplings of serum S100B were performed on admission and on postoperative days 1, 3, and 7. There is a statistically significant difference between both groups as regards the second reading of the S100B (day 1 postoperative; P < 0.05). There is no statistically significant difference between both groups as regards outcome at 3 months using clinical status and S100B values. There is a tendency in the magnesium group to have better outcomes. Further studies with more number of patients with subarachnoid hemorrhage are needed to determinate the accuracy of S100B protein as a prognostic marker and of magnesium sulfate as a neuroprotector.


Similar content being viewed by others
References
Barker FG, Ogilvy CS (1996) Efficacy of prophylactic nimodipine for delayed ischemic deficit after subarachnoid hemorrhage: a metaanalysis. J Neurosurg 84(3):405–414
Boet R, Chan MT, Poon WS, Wong GK, Wong HT, Gin T (2005) Intravenous magnesium sulfate to improve outcome after aneurysmal subarachnoid hemorrhage: interim report from a pilot study. Acta Neurochir Suppl 95:263–264
Clark JF, Pyne GJ, Choutka J, Carrozzella JA, Khoury J, Broderick JP (2001) In vitro therapy with dobutamine, isoprenaline and sodium nitroprusside protects vascular smooth muscle metabolism from subarachnoid haemorrhage induced cerebral vasospasm. Acta Neurochir (Wien) 143(7):721–728
Dabrowski W (2009) Magnesium supplementation significantly reduces serum S100beta concentrations in patients who have undergone coronary artery bypass surgery. Magnes Res 22(1):21–31
Dabrowski W (2007) Do changes in S100beta protein correlate with serum magnesium concentrations in patients undergoing extracorporeal circulation? Magnes Res 20(3):168–176
Dorhout Mees SM, van den Bergh WM, Algra A, Rinkel GJ (2007) Achieved serum magnesium concentrations and occurrence of delayed cerebral ischaemia and poor outcome in aneurysmal subarachnoid haemorrhage. J Neurol Neurosurg Psychiatry 78(7):729–731
Feigin VL, Rinkel GJ, Algra A, Vermeulen M, van Gijn J (1998) Calcium antagonists in patients with aneurysmal subarachnoid hemorrhage: a systematic review. Neurology 50(4):876–883
Feng DF, Zhu ZA, Lu YC (2004) Effects of magnesium sulfate on traumatic brain edema in rats. Chin J Traumatol 7(3):148–152
Fisher CM, Kistler JP, Davis JM (1980) Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery 6(1):1–9
Jennett B, Bond M (1975) Assessment of outcome after severe brain damage. Lancet 1(7905):480–484
Kay A, Petzold A, Kerr M, Keir G, Thompson E, Nicoll J (2003) Decreased cerebrospinal fluid apolipoprotein E after subarachnoid hemorrhage: correlation with injury severity and clinical outcome. Stroke 34(3):637–642
Keyrouz SG, Diringer MN (2007) Clinical review: prevention and therapy of vasospasm in subarachnoid hemorrhage. Crit Care 11(4):220
Kleine TO, Benes L, Zofel P (2003) Studies of the brain specificity of S100B and neuron-specific enolase (NSE) in blood serum of acute care patients. Brain Res Bull 6(3):265–279
Meloni BP, Zhu H, Knuckey NW (2006) Is magnesium neuroprotective following global and focal cerebral ischemia? A review of published studies. Magnes Res 19(2):123–137
Muroi C, Terzic A, Fortunati M, Yonekawa Y, Keller E (2008) Magnesium sulfate in the management of patients with aneurysmal subarachnoid hemorrhage: a randomized, placebo-controlled, dose-adapted trial. Surg Neurol 69(1):33–39, Discussion 9
Oertel M, Schumacher U, McArthur DL, Kastner S, Boker DK (2006) S-100B and NSE: markers of initial impact of subarachnoid haemorrhage and their relation to vasospasm and outcome. J Clin Neurosci 13(8):834–840
Prevedello DM, Cordeiro JG, de Morais AL, Saucedo NS Jr, Chen IB, Araújo JC (2006) Magnesium sulfate: role as possible attenuating factor in vasospasm morbidity. Surg Neurol 65:S1:14–S1:21
Raabe AKO, Woszczyk A, Lang J, Gerlach R, Zimmermann M, Seifert V (2004) S-100B protein as a serum marker of secondary neurological complications in neurocritical care patients. Neurol Res 26:440–445
(1988) Report of World Federation of Neurological Surgeons Committee on a Universal Subarachnoid Hemorrhage Grading Scale. J Neurosurg 68(6): 985–986
Sanchez-Peña P, Pereira AR, Sourour NA, Biondi A, Lejean L, Colonne C, Boch AL, Al Hawari M, Abdennour L, Puybasset L (2008) S100B as an additional prognostic marker in subarachnoid aneurysmal hemorrhage. Crit Care Med 36:2267–2273
Schmid-Elsaesser R, Kunz M, Zausinger S, Prueckner S, Briegel J, Steiger HJ (2006) Intravenous magnesium versus nimodipine in the treatment of patients with aneurysmal subarachnoid hemorrhage: a randomized study. Neurosurgery 58(6):1054–1065
Van den Bergh WM, Albrecht KW, Berkelbach van der Sprenkel JW, Rinkel GJ (2003) Magnesium therapy after aneurysmal subarachnoid haemorrhage a dose-finding study for long term treatment. Acta Neurochir (Wien) 145(3):195–199
Veyna RS, Seyfried D, Burke DG, Zimmerman C, Mlynarek M, Nichols V, Marrocco A, Thomas AJ, Mitsias PD, Malik GM (2002) Magnesium sulfate therapy after aneurysmal subarachnoid hemorrhage. J Neurosurg 96(3):510–514
Weiss N, Sanchez-Pena P, Roche S, Beaudeux JL, Colonne C, Coriat P, Puybasset L (2006) Prognosis value of plasma S100B protein levels after subarachnoid aneurysmal hemorrhage. Anesthesiology 104(4):658–666
Wong GK, Poon WS, Chan MT, Boet R, Gin T, Stephanie CP, Zee B (2010) Intravenous Magnesium Sulphate for Aneurysmal Subarachnoid Hemorrhage (IMASH): a randomized, double-blinded, placebo-controlled, multicenter phase III trial. Stroke 41:921–926
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
Claudius Thomé, Innsbruck, Austria
Hassan et al. have performed a randomized study on the effect of intravenous magnesium sulfate on outcome and S100B levels after aneurysmal subarachnoid hemorrhage (SAH) in a series of 30 patients. S100B was chosen as a surrogate parameter for neurological damage. Patients were included, treated by surgical clip**, and started on an established dose of magnesium or an equal volume of saline within 4 days after SAH. Unfortunately, S100B levels were analyzed in relation to the time point of surgery rather than the time point of SAH, so that it is unclear whether the significantly lower S100B value on the first postoperative day in the magnesium group is a therapeutic effect or merely due to intergroup differences in timing. Vasospasm was present in 3 of 15 treated versus 2 of 15 control patients, but a definition of vasospasm—be it clinically, angiographically, or per TCD—is lacking. There are some methodological flaws like the absence of a power calculation as the small patient numbers would have never allowed meaningful conclusions. Most of these drawbacks can be somewhat overcome by the dedication to perform a randomized study, which is considered the most appropriate means to establish scientific knowledge. One criticism, however, cannot be stressed enough: Favorable outcome of 73% in the magnesium group versus 60% in the control group reaching a P value of 0.43 simply states that there is no difference in outcome! This cannot be interpreted as “a tendency in the magnesium group to have better outcomes” as concluded in the abstract!
Overall, I like the idea of the manuscript to combine a surrogate biomarker with a randomized study design for a potentially neuroprotective agent. Nevertheless, there already is a large amount of data on magnesium sulfate in SAH from six phase II and one phase III randomized-controlled trials, which has been recently reviewed by Suarez et al. (1) The phase III trial with 327 patients revealed no difference in outcome whatsoever (2), which is in line with the results of the present study.
References
1. Suarez JI (2011) Participants in the International Multidisciplinary Consensus Conference on the Critical Care Management of Subarachnoid Hemorrhage. Magnesium sulfate administration in subarachnoid hemorrhage. Neurocrit Care 15:302–307.
2. Wong GK, Poon WS, Chan MT, Boet R, Gin T, Ng SC, Zee BC (2010) Plasma magnesium concentrations and clinical outcomes in aneurysmal subarachnoid hemorrhage patients: post hoc analysis of intravenous magnesium sulfate for aneurysmal subarachnoid hemorrhage trial. Stroke 41:1841–1844.
Rights and permissions
About this article
Cite this article
Hassan, T., Nassar, M., Elhadi, S.M. et al. Effect of magnesium sulfate therapy on patients with aneurysmal subarachnoid hemorrhage using serum S100B protein as a prognostic marker. Neurosurg Rev 35, 421–427 (2012). https://doi.org/10.1007/s10143-011-0368-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-011-0368-8