Abstract
Background
The exploitation of routinely collected clinical health information is warranted to optimize the case detection and diagnostic workout of Alzheimer’s disease (AD). We aimed to derive an AD prediction score based on routinely collected primary care data.
Methods
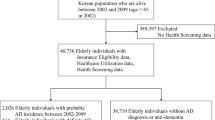
We built a cohort selecting 199,978 primary care patients 60 + part of the Health Search Database between January 2002 and 2009, followed up until 2019 to detect incident AD cases. The cohort was randomly divided into a derivation and validation sub-cohort. To identify AD and non-AD cases, we applied a clinical algorithm that involved two clinicians. According to a nested case–control design, AD cases were matched with up to 10 controls based on age, sex, calendar period, and follow-up duration. Using the derivation sub-cohort, 32 potential AD predictors (sociodemographic, clinical, drug-related, etc.) were tested in a logistic regression and selected to build a prediction model. The predictive performance of this model was tested on the validation sub-cohort by mean of explained variation, calibration, and discrimination measurements.
Results
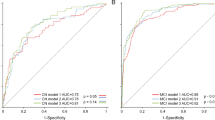
We identified 3223 AD cases. The presence of memory disorders, hallucinations, anxiety, and depression and the use of NSAIDs were associated with future AD. The combination of the predictors allowed the production of a predictive score that showed an explained variation (pseudo-R2) for AD occurrence of 13.4%, good calibration parameters, and an area under the curve of 0.73 (95% CI: 0.71–0.75). In accordance with this model, 7% of patients presented with a high-risk score for develo** AD over 15 years.
Conclusion
An automated risk score for AD based on routinely collected clinical data is a promising tool for the early case detection and timely management of patients by the general practitioners.

Similar content being viewed by others
References
World Health Organization (2015) World report on ageing and health. World Health Organization. https://apps.who.int/iris/handle/10665/186463
Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396(10258):1204–1222. https://doi.org/10.1016/S0140-6736(20)30925-9. Erratum in: Lancet. 2020 Nov 14;396(10262):1562.
Feigin VL, Vos T, Nichols E, Owolabi MO, Carroll WM, Dichgans M et al (2020) The global burden of neurological disorders: translating evidence into policy. Lancet Neurol 19:255–265. https://doi.org/10.1016/S1474-4422(19)30411-9
World Health Organization (2015) Global action against dementia. https://apps.who.int/iris/bitstream/handle/10665/179537/9789241509114_eng.pdf
Alzheimer’s Disease International (2015) The global impact of dementia: An analysis of prevalence, cost and trends. https://www.alzint.org/resource/world-alzheimer-report-2015/
Winblad B, Amouyel P, Andrieu S, Ballard C, Brayne C, Brodaty H et al (2016) Defeating Alzheimer’s disease and other dementias: a priority for European science and society. Lancet Neurol 15:455–532
Anderson RM, Hadjichrysanthou C, Evans S, Wong MM (2017) Why do so many clinical trials of therapies for Alzheimer’s disease fail? Lancet 390:2327–2329
Cummings J, Lee G, Ritter A, Sabbagh M, Zhong K (2019) Alzheimer’s disease drug development pipeline: 2019. Alzheimers Dement (N Y) 5:272–293
Livingston G, Huntley J, Sommerlad A, Ames D, Ballard C, Banerjee S et al (2020) Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 396:413–446
Dubois B, Padovani A, Scheltens P, Rossi A, Dell’Agnello G (2016) Timely diagnosis for Alzheimer’s disease: a literature review on benefits and challenges. J Alzheimers Dis 49:617–631
Hampel H, O’Bryant SE, Molinuevo JL, Zetterberg H, Masters CL, Lista S et al (2018) Blood-based biomarkers for Alzheimer disease: map** the road to the clinic. Nat Rev Neurol 14:639–652
Downs MG (1996) The role of general practice and the primary care team in dementia diagnosis and management. Int J Ger Psychiatry 11:937–942
Grande G, Tramacere I, Vetrano DL, Clerici F, Pomati S, Mariani C, Filippini G, REMIND Study Group (Rete Milanese Integrata per le Demenze) (2017) Role of anticholinergic burden in primary care patients with first cognitive complaints. Eur J Neurol. 24(7):950–955. https://doi.org/10.1111/ene.13313
Grande G, Vetrano DL, Mazzoleni F, Lovato V, Pata M, Cricelli C, Lapi F (2020) Detection and prediction of incident Alzheimer dementia over a 10-year or longer medical history: a population-based study in primary care. Dement Geriatr Cogn Disord 49(4):384–389. https://doi.org/10.1159/000509379
Ford E, Greenslade N, Paudyal P, Bremner S, Smith HE, Banerjee S, Sadhwani S, Rooney P, Oliver S, Cassell J (2018) Predicting dementia from primary care records: a systematic review and meta-analysis. PLoS One 13(3):e0194735. https://doi.org/10.1371/journal.pone.0194735
Clift AK et al (2020) Living risk prediction algorithm (QCOVID) for risk of hospital admission and mortality from coronavirus 19 in adults: national derivation and validation cohort study. BMJ 371:m3731
Ganna A, Reilly M, De Faire U, Pedersen N, Magnusson P, Ingelsson E (2012) Risk prediction measures for case-cohort and nested case-control designs: an application to cardiovascular disease. Am J Epidemiol 175(7):715–724. https://doi.org/10.1093/aje/kwr374
Van Hoorde K, Van Huffel S, Timmerman D, Bourne T, Van Calster B (2015) A spline-based tool to assess and visualize the calibration of multiclass risk predictions. J Biomed Inform 54:283–293. https://doi.org/10.1016/j.jbi.2014.12.016
Van Calster B, Nieboer D, Vergouwe Y, De Cock B, Pencina MJ, Steyerberg EW (2016) A calibration hierarchy for risk models was defined: from utopia to empirical data. J Clin Epidemiol 74:167–176. https://doi.org/10.1016/j.jclinepi.2015.12.005
Cox DR (1957) Note on Grou**. J Am Stat Assoc 52:543–547. https://doi.org/10.1080/01621459.1957.10501411
Delaney JA et al (2007) Lifestyle variables and the risk of myocardial infarction in the general practice research database. BMC Cardiovasc Disord 7:38
Kivipelto M, Ngandu T, Laatikainen T, Winblad B, Soininen H, Tuomilehto J (2006) Risk score for the prediction of dementia risk in 20 years among middle aged people: a longitudinal, population-based study. Lancet Neurol 5:735–741
Stephan BC, Kurth T, Matthews FE, Brayne C, Dufouil C (2010) Dementia risk prediction in the population: are screening models accurate? Nat Rev Neurol 6:318–326
Müller T, Payton NM, Kalpouzos G, Jessen F, Grande G, Bäckman L, Laukka EJ (2020) Cognitive, genetic, brain volume, and diffusion tensor imaging markers as early indicators of dementia. J Alzheimers Dis 77(4):1443–1453. https://doi.org/10.3233/JAD-200445
Grande G, Rizzuto D, Vetrano DL, Marseglia A, Vanacore N, Laukka JE, Welmer AK, Fratiglioni L (2020) Cognitive and physical markers of prodromal dementia: a 12-year-long population study. Alzheimers Dement 16(1):153–161
Collins GS, Altman D (2012) Predicting the 10 year risk of cardiovascular disease in the United Kingdom: independent and external validation of an updated version of QRISK2. BMJ 344:e4181
Hippisley-Cox J, Coupland C, Brindle P (2013) Derivation and validation of QStroke score for predicting risk of ischaemic stroke in primary care and comparison with other risk scores: a prospective open cohort study. BMJ 2(346):f2573. https://doi.org/10.1136/bmj.f2573
Hippisley-Cox J, Coupland C (2017) Development and validation of QDiabetes-2018 risk prediction algorithm to estimate future risk of type 2 diabetes: cohort study. BMJ 359:j5019. https://doi.org/10.1136/bmj.j5019
Walters K, Hardoon S, Petersen I et al (2016) Predicting dementia risk in primary care: development and validation of the Dementia Risk Score using routinely collected data. BMC Med 14:6
Ford E, Sheppard J, Oliver S, Rooney P, Banerjee S, Cassell JA (2021) Automated detection of patients with dementia whose symptoms have been identified in primary care but have no formal diagnosis: a retrospective case-control study using electronic primary care records. BMJ Open 11(1):e039248. https://doi.org/10.1136/bmjopen-2020-039248
Petersen RC (2004) Mild cognitive impairment as a diagnostic entity. J Intern Med 256(3):183–194. https://doi.org/10.1111/j.1365-2796.2004.01388.x
Jessen F, Kleineidam L, Wolfsgruber S, Bickel H, Brettschneider C, Fuchs A et al (2020) Prediction of dementia of Alzheimer type by different types of subjective cognitive decline. Alzheimers Dement 16:1745–1749. https://doi.org/10.1002/alz.12163
Dotson VM, Beydoun MA, Zonderman AB (2010 Jul 6) Recurrent depressive symptoms and the incidence of dementia and mild cognitive impairment. Neurology 75(1):27–34
Brommelhoff JA, Gatz M, Johansson B, McArdle JJ, Fratiglioni L, Pedersen NL (2009) Depression as a risk factor or prodromal feature for dementia? Findings in a population-based sample of Swedish twins. Psychol Aging 24(2):373–384
Singh-Manoux A, Dugravot A, Fournier A, Abell J, Ebmeier K, Kivimaki M et al (2017) Trajectories of depressive symptoms before diagnosis of dementia: a 28-year follow-up study. JAMA Psychiat 74(7):712–718
Enache D, Winblad B, Aarsland D (2011) Depression in dementia: epidemiology, mechanisms, and treatment. Curr Opin Psychiatry 24(6):461–472
Gallacher J, Bayer A, Fish M et al (2009) Does anxiety affect risk of dementia? Findings from the Caerphilly Prospective Study. Psychosom Med 71:659–666
Burton C, Campbell P, Jordan K, Strauss V, Mallen C (2013) The association of anxiety and depression with future dementia diagnosis: a case-control study in primary care. Fam Pract 30(1):25–30. https://doi.org/10.1093/fampra/cms044
Szekely CA, Green RC, Breitner JC, Ostbye T, Beiser AS, Corrada MM et al (2008) No advantage of A beta 42-lowering NSAIDs for prevention of Alzheimer dementia in six pooled cohort studies. Neurology 70(24):2291–2298
Ferrucci L, Fabbri E (2018) Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol 15:505–522
Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C et al (2019) Chronic inflammation in the etiology of disease across the life span. Nat Med 25:1822–1832
Funding
The study was sponsored by Roche S.p.A.. The corresponding author had full access to all the data in the study and has final responsibility for the decision to submit it for publication.
Author information
Authors and Affiliations
Contributions
FL contributed to the conception and design of the study and conducted the statistical analyses. All the authors contributed to the interpretation of the results. GG and DLV drafted the first version of the manuscript. All the authors critically revised the manuscript for important intellectual content. All the authors made a significant contribution to the research and the development of the manuscript and approved the final version for publication.
Corresponding author
Ethics declarations
Ethical approval
According to a by-law on the classification and implementation of observational drug-related research, as issued by the Italian Medicines Agency, the present study does not require approval by an ethics committee.
Conflict of interest
FL, EM, EB and IC provided consultancies in protocol preparation for epidemiological studies and data analyses for Roche and Abbvie; DT and CC provided clinical consultancies for Roche, Abbvie and Viatris; VL and LG are employees of Roche S.p.A., Monza, Italy; the other co-authors have no conflict of interest to diclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Grande, G., Vetrano, D.L., Marconi, E. et al. Development and internal validation of a prognostic model for 15-year risk of Alzheimer dementia in primary care patients. Neurol Sci 43, 5899–5908 (2022). https://doi.org/10.1007/s10072-022-06258-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06258-7