Abstract
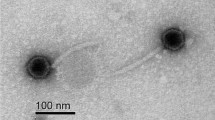
Bacteriophages have been intensively-studied and applied in a variety of practical applications such as biocontrol of food-borne pathogens. Especially, endolysin of bacteriophage is highly specific to host strains that can hydrolyze cell wall. To get the control agent for foodpoisoning Bacillus cereus, cloning and nucleotide sequence of putative endolysin from temperate phage 250 (Tp250) of emetic B. cereus 250 were investigated. Tp250 was induced by mitomycin C and belonged to Siphoviridae family. Tp250, a circular dsDNA genome of 56,505 bp, consisted of various structural and functional genes for survival or propagation. ORFs from Tp250 were involved in replication, head morphogenesis, head-to-tail joining, tail morphogenesis, lysogenic module, host lysis, and other functional genes such as related metabolisms. For cloning of putative endolysin, Tp 250 DNA was extracted and pLYS250 was constructed. The pLYS250 had 752 ORF encoding a putative endolysin with 250 amino acids, which was expressed by around 28 kDa in Escherichia coli. Therefore, Tp250 and the endolysin might be applied as one of agents to reduce B.cereus pathogen in foods directly or through over-expression and purification.
Similar content being viewed by others
References
EFSA. http://efsa.eu/EFSA/efsa. Accessed on March 5, 2009.
KFDA. http://kfda.go.kr. Accessed on June 12, 2008.
Barbara L, Anthony CBP, Grahame WG. Microbiological safety and quality of food. Vol II, pp. 1030–1056. In: Bacillus cereus. Springer-Verlag, UK (1999)
Lee SY, Costello M, Kang DH. Efficacy of chlorine dioxide gas as a sanitizer of lettuce leaves. J. Food Protect. 67: 1371–1476 (2004)
Takeuchi K, Frank JF. Penetration of Escherichia coli O157:H7 into lettuce tissues as affected by inoculum size and temperature and the effect of chlorine treatment on cell viability. J. Food Protect. 63: 434–440 (2000)
O’Toole GA, Kaplan HB, Kolter R. Biofilm formation as microbial development. Annu. Rev. Microbiol. 54: 49–79 (2000)
Burnett SL, Beuchat LR. Human pathogens associated with raw produce and unpasteurized juices, and difficulties in decontamination. J. Ind. Microbiol. Biot. 27: 104–110 (2001)
Brussow A, Kutter E. Phage Ecology. Boca Raton CRC Press, USA. pp. 129–163 (2005)
Garcia P, Rodriguez L, Rodriguez A, Martinez B. Food biopreservation: Promising strategies using bacteriocins, bacteriophages, and endolysins. Trends Food Sci. Tech. 21: 373–382 (2010)
Modi R, Hirvi Y, Hill A, Griffiths MW. Effect of phage on survival of Salmonella enteritidis during manufacture and storage of Cheddar cheese made from raw and pasteurized milk. J. Food Protect. 64: 927–933 (2001)
Whichard JM, Sriranganathan N, Pierson FW. Suppression for Salmonella growth by wild-type and large-plaque variants of bacteriophage Felix O1 in liquid culture and on chicken frankfurters. J. Food Protect. 66: 220–225 (2003)
Atterbury RJ, Dillon E, Swift C, Connerton PL, Frost JA, Dodd CE. Correlation of Campylobacter bacteriophage with reduced presence of hosts in broiler chicken ceca. Appl. Environ. Microb. 71: 4885–4887 (2005)
Martinez B, Obeso JM, Rodriguez A, Garcia P. Nisin-bacteriohage crossresistance in Staphylococcus aureus. Int. J. Food Microbiol. 122: 253–258 (2008)
Kim KP, Klumpp J, Loessner MJ. Enterobacter sakazakii bacteriophages can prevent bacterial growth in reconstituted infant formula. Int. J. Food Microbiol. 115: 195–203 (2007)
Lossener MJ. Bacteriophage endolysins-current state of research and allocations. Curr. Opin. Microbiol. 8: 480–487 (2005)
Young RY, Wang IN, Roof WD. Phages will out: Strategies of host cell lysis. Trends Microbiol. 8: 120–128 (2000)
Fischetti VA. Bacteriophage lysins as effective antibacterials. Curr. Opi. Microbiol. 11: 393–400 (2008)
Beattie SH, Williams AG. Detection of toxigenic strains of Bacillus cereus and other Bacillus spp. with an improved cytotoxicity assay. Lett. Appl. Microbiol. 28: 221–225 (1999)
Ankolekar C, Rahmati T, Labbe RG. Detection of toxigenic Bacillus cereus and Bacillus thuringiensis spores in U.S. rice. Int. J. Food Microbiol. 128: 460–466 (2009)
Kim SH, Kim JS, Choi JP, Park JH. Prevalence and frequency of food-borne pathogens on unprocessed agricultural and marine products. Korean J. Food Sci. Technol. 38: 594–598 (2006)
Cronin UP, Wilkinson MG. The growth, physiology, and potential of Bacillus cereus in cooked rice during storage temperature abuse. Food Control 20: 822–282 (2009)
Manfioletti G, Schneider C. A new and fast method for preparing high quality lambda DNA suitable for sequencing. Nucleic Acid Res. 16: 2873–2884 (1988)
Ferretti JJ, McShan WM, Ajdic D, Savic DJ, Savic G, Lyon K, Primeaux C, Sezate S, Suvorov AN, Kenton S, Lai HS, Lin SP, Qian Y, Jia HG, Najar FZ, Ren Q, Zhu H, Song L, White J, Yuan X, Clifton SW, Roe BA, Laughlin R. Complete genome sequence of an M1 strain of Streptococcus pyogenes. P. Natl. Acad. Sci. USA 98: 4658–4663 (2001)
Vollmer W, Joris B, Charlier P, Foster S. Bacterial peptidoglycan (murein) hydrolases. FEMS Microbiol. Rev. 32: 259–286 (2008)
Dziarski R, Gupta D. The peptidoglycan recognition proteins (PGRPs). Genome Biol. 7: 232.1–232.13 (2006)
Young R. Bacteriophage lysis: Mchanism and regulation. Microbiol. Rev. 56: 430–481 (1992)
McGrath S, van Sinderen D. Bacteriophage: Genetics and Molecular Biology. Caister Academic Press. NewYork, NY, USA. pp. 43–60 (2007)
Loessner MJ, Maier SK, Daubek-Puza H, Wendlinger G, Scherer S. Three Bacillus cereus bacteriophage endolysins are unrelated but reveal high homology to cell wall hydrolases from different bacilli. J. Bacteriol. 179: 2845–2851 (1997)
Regamey A, Karamata D. The N-acetylmuramoyl-l-alanine amidase encoded by the Bacillus subtilis 168 prophage SP beta. Microbiology 144: 885–893 (1998)
Kikkawa HS, Ueda T, Suzuki S, Yasuda J. Characterization of the catalytic activity of the gamma-phage lysin, PlyG, specific for Bacillus anthracis. FEMS Microbiol. Lett. 286: 236–240 (2008)
Verheust C, Fornelos N, Mahillon J. The Bacillus thuringiensis phage GIL01 encodes two enzymes with peptidoglycan hydrolase activity. FEMS Microbiol. Lett. 237: 289–295 (2004)
Low LY, Yang C, Perego M, Osterman A, Liddington RC. Structure and lytic activity of a Bacillus anthracis prophage endolysin. J. Biol. Chem. 280: 35433–35439
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, YD., Park, JH. Genomic sequence of temperate phage 250 isolated from emetic B. cereus and cloning of putative endolysin. Food Sci Biotechnol 19, 1643–1648 (2010). https://doi.org/10.1007/s10068-010-0232-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-010-0232-6