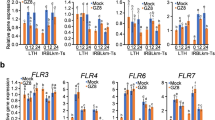
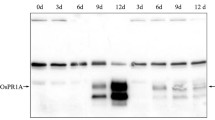
Abstract
The required for Mla12 resistance (RAR1) protein is essential for the plant immune response. In rice, a model monocot species, the function of Oryza sativa RAR1 (OsRAR1) has been little explored. In our current study, we characterized the response of a rice osrar1 T-DNA insertion mutant to infection by Magnaporthe oryzae, the causal agent of rice blast disease. osrar1 mutants displayed reduced resistance compared with wild type rice when inoculated with the normally virulent M. oryzae isolate PO6-6, indicating that OsRAR1 is required for an immune response to this pathogen. We also investigated the function of OsRAR1 in the resistance mechanism mediated by the immune receptor genes Pib and Pi5 that encode nucleotide binding-leucine rich repeat (NB-LRR) proteins. We inoculated progeny from Pib/osrar1 and Pi5/osrar1 heterozygous plants with the avirulent M. oryzae isolates, race 007 and PO6-6, respectively. We found that only Pib-mediated resistance was compromised by the osrar1 mutation and that the introduction of the OsRAR1 cDNA into Pib/osrar1 rescued Pib-mediated resistance. These results indicate that OsRAR1 is required for Pib-mediated resistance but not Pi5-mediated resistance to M. oryzae.
Similar content being viewed by others
References
An, G., Jeong, D.H., Jung, K.H., and Lee, S. (2005a). Reverse genetic approaches for functional genomics of rice. Plant Mol. Biol. 59, 111–123.
An, G., Lee, S., Kim, S.H., and Kim, S.R. (2005b). Molecular genetics using T-DNA in rice. Plant Cell Physiol. 46, 14–22.
Austin, M.J., Muskett, P., Kahn, K., Feys, B.J., Jones, J.D., and Parker, J.E. (2002). Regulatory role of SGT1 in early R gene-mediated plant defenses. Science 295, 2077–2080.
Azevedo, C., Sadanandom, A., Kitagawa, K., Freialdenhoven, A., Shirasu, K., and Schulze-Lefert, P. (2002). The RAR1 interactor SGT1, an essential component of R gene-triggered disease resistance. Science 295, 2073–2076.
Bhattarai, K.K., Li, Q., Liu, Y., Dinesh-Kumar, S.P., and Kaloshian, I. (2007). The Mi-1-mediated pest resistance requires Hsp90 and Sgt1. Plant Physiol. 144, 312–323.
Bieri, S., Mauch, S., Shen, Q.H., Peart, J., Devoto, A., Casais, C., Ceron, F., Schulze, S., Steinbiss, H.H., Shirasu, K., et al. (2004). RAR1 positively controls steady state levels of barley MLA resistance proteins and enables sufficient MLA6 accumulation for effective resistance. Plant Cell 16, 3480–3495.
Bittel, P., and Robatzek, S. (2007). Microbe-associated molecular patterns (MAMPs) probe plant immunity. Curr. Opin. Plant Biol. 10, 335–341.
Boller, T., and He, S.Y. (2009). Innate immunity in plants: an arms race between pattern recognition receptors in plants and effectors in microbial pathogens. Science 324, 742–744.
Chandra-Shekara, A.C., Navarre, D., Kachroo, A., Kang, H.G., Klessig, D., and Kachroo, P. (2004). Signaling requirements and role of salicylic acid in HRT- and rrt-mediated resistance to turnip crinkle virus in Arabidopsis. Plant J. 40, 647–659.
Chen, D.H., and Ronald, P.C. (1999). A rapid DNA minipreparation method suitable for AFLP and other PCR applications. Plant Mol Biol. Rep. 17, 53–57.
Chen, D.H., Zeigler, R.S., and Ahn, S.W. (1996). Phenotypic characterization of the rice blast resistance gene Pi-2(t). Plant Disease 80, 52–56.
Chen, X., Shang, J., Chen, D., Lei, C., Zou, Y., Zhai, W., Liu, G., Xu, J., Ling, Z., Cao, G., et al. (2006). A B-lectin receptor kinase gene conferring rice blast resistance. Plant J. 46, 794–804.
Cho, J.I., Ryoo, N., Ko, S., Lee, S.K., Lee, J., Jung, K.H., Lee, Y.H., Bhoo, S.H., Winderickx, J., An, G., et al. (2006). Structure, expression, and functional analysis of the hexokinase gene family in rice (Oryza sativa L.). Planta 224, 598–611.
Dardick, C., and Ronald, P.C. (2006). Plant and animal pathogen recognition receptors signal through non-RD kinases. PLoS Pathog. 2, e2.
de la Fuente van Bentem, S., Vossen, J.H., de Vries, K.J., van Wees, S., Tameling, W.I., Dekker, H.L., de Koster, C.G., Haring, M.A., Takken, F.L., and Cornelissen, B.J. (2005). Heat shock protein 90 and its co-chaperone protein phosphatase 5 interact with distinct regions of the tomato I-2 disease resistance protein. Plant J. 43, 284–298.
Eom, J.S., Cho, J.I., Reinders, A., Lee, S.W., Yoo, Y., Tuan, P.Q., Choi, S.B., Bang, G., Park, Y.I., Cho, M.H., et al. (2011). Impaired function of the tonoplast-localized sucrose transporter in rice, OsSUT2, limits the transport of vacuolar reserve sucrose and affects plant growth. Plant Physiol. 157, 109–119.
Holt, B.F.III, Belkhadir, Y., and Dangl, J.L. (2005). Antagonistic control of disease resistance protein stability in the plant immune system. Science 309, 929–932.
Hubert, D.A., Tornero, P., Belkhadir, Y., Krishna, P., Takahashi, A., Shirasu, K., and Dangl, J.L. (2003). Cytosolic HSP90 associates with and modulates the Arabidopsis RPM1 disease resistance protein. EMBO J. 22, 5679–5689.
Jain, M., Nijhawan, A., Tyagi, A.K., and Khurana, J.P. (2006). Validation of housekee** genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem. Biophys. Res. Commun. 345, 646–651.
Jarosch, B., Collins, N.C., Zellerhoff, N., and Schaffrath, U. (2005). RAR1, ROR1, and the actin cytoskeleton contribute to basal resistance to Magnaporthe grisea in barley. Mol. Plant Microbe Interact. 18, 397–404.
Jeong, D.H., An, S., Park, S., Kang, H.G., Park, G.G., Kim, S.R., Sim, J., Kim, Y.O., Kim, M.K., Kim, S.R., et al. (2006). Generation of a flanking sequence-tag database for activation-tagging lines in japonica rice. Plant J. 45, 123–132.
Jones, J.D., and Dangl, J.L. (2006). The plant immune system. Nature 444, 323–329.
Kadota, Y., Shirasu, K., and Guerois, R. (2010). NLR sensors meet at the SGT1-HSP90 crossroad. Trends Biochem. Sci. 35, 199–207.
Lee, S.W., Han, S.W., Bartley, L.E., and Ronald, P.C. (2006). Unique characteristics of Xanthomonas oryzae pv. oryzae AvrXa21 and implications for plant innate immunity. Proc. Natl. Acad. Sci. USA 103, 18395–18400.
Lee, S.W., Han, S.W., Sririyanum, M., Park, C.J., Seo, Y.S., and Ronald, P.C. (2009a). A type I-secreted, sulfated peptide triggers XA21-mediated innate immunity. Science 326, 850–853.
Lee, S.K., Song, M.Y., Seo, Y.S., Kim, H.K., Ko, S., Cao, P.J., Suh, J.P., Yi, G., Roh, J.H., Lee, S., et al. (2009b). Rice Pi5-mediated resistance to Magnaporthe oryzae requires the presence of two coiled-coil-nucleotide-binding-leucine-rich repeat genes. Genetics 181, 1627–1638.
Leister, R.T., Dahlbeck, D., Day, B., Li, Y., Chesnokova, O., and Staskawicz, B.J. (2005). Molecular genetic evidence for the role of SGT1 in the intramolecular complementation of Bs2 protein activity in Nicotiana benthamiana. Plant Cell 17, 1268–1278.
Liu, Y., Burch-Smith, T., Schiff, M., Feng, S., and Dinesh-Kumar, S.P. (2004). Molecular chaperone Hsp90 associates with resistance protein N and its signaling proteins SGT1 and Rar1 to modulate an innate immune response in plants. J. Biol. Chem. 279, 2101–2108.
Liu, J., Wang, X., Mitchell, T., Hu, Y., Liu, X., Dai, L., and Wang, G.L. (2010). Recent progress and understanding of the molecular mechanisms of the rice-Magnaporthe oryzae interaction. Mol. Plant Pathol. 11, 419–427.
Lu, R., Malcuit, I., Moffett, P., Ruiz, M.T., Peart, J., Wu, A.J., Rathjen, J.P., Bendahmane, A., Day, L., and Baulcombe, D.C. (2003). High throughput virus-induced gene silencing implicates heat shock protein 90 in plant disease resistance. EMBO J. 22, 5690–5699.
Martin, G.B., Bogdanove, A.J., and Sessa, G. (2003). Understanding the functions of plant disease resistance proteins. Annu. Rev. Plant Biol. 54, 23–61.
Muskett, P.R., Kahn, K., Austin, M.J., Moisan, L.J., Sadanandom, A., Shirasu, K., Jones, J.D., and Parker, J.E. (2002). Arabidopsis RAR1 exerts rate-limiting control of R gene-mediated defenses against multiple pathogens. Plant Cell 14, 979–992.
Noda, T., Nagao, H., Pham, V.D., P.V., Dinh, H.D., and Lai, V.E. (1999). Distribution of pathogenic races of rice blast fungus in Vietnam. Annu. Pytopathol. Soc. Jpn. 65, 526–530.
Okuyama, Y., Kanzaki, H., Abe, A., Yoshida, K., Tamiru, M., Saitoh, H., Fujibe, T., Matsumura, H., Shenton, M., Galam, D.C., et al. (2011). A multifaceted genomics approach allows the isolation of the rice Pia-blast resistance gene consisting of two adjacent NBLRR protein genes. Plant J. 66, 467–479.
Ou, S.H. (1985). Fungus diseases — foliage diseases. In Rice Diseases, (Kew, England: Commonwealth Mycological Institute), pp. 109–201.
Peart, J.R., Lu, R., Sadanandom, A., Malcuit, I., Moffett, P., Brice, D.C., Schauser, L., Jaggard, D.A., **ao, S., Coleman, M.J., et al. (2002). Ubiquitin ligase-associated protein SGT1 is required for host and nonhost disease resistance in plants. Proc. Natl. Acad. Sci. USA 99, 10865–10869.
Qi, M., and Yang, Y. (2002). Quantification of Magnaporthe grisea during infection of rice plants using real-time polymerase chain reaction and northern blot/phosphoimaging analyses. Phytopathology 92, 870–876.
Scofield, S.R., Huang, L., Brandt, A.S., and Gill, B.S. (2005). Development of a virus-induced gene-silencing system for hexaploid wheat and its use in functional analysis of the Lr21-mediated leaf rust resistance pathway. Plant Physiol. 138, 2165–2173.
Seo, Y.S., Lee, S.K., Song, M.Y., Suh, J.P., Hahn, T.R., Ronald, P.C., and Jeon, J.S. (2008). The HSP90.SGT1-RAR1 molecular chaperone complex: a core modulator in plant immunity. J. Plant Biol. 51, 1–10.
Seo, Y.S., Chern, M., Bartley, L.E., Han, M., Jung, K.H., Lee, I., Walia, H., Xu, X., Cao, P., Bai, W., et al. (2011). Towards establishment of a rice stress response interactome. PLoS Genet. 7, e1002020.
Shirasu, K. (2009). The HSP90-SGT1 chaperone complex for NLR immune sensors. Annu. Rev. Plant Biol. 60, 139–164.
Shirasu, K., and Schulze-Lefert, P. (2003). Complex formation, promiscuity and multi-functionality: protein interactions in diseaseresistance pathways. Trends Plant Sci. 8, 252–258.
Shirasu, K., Lahaye, T., Tan, M.W., Zhou, F., Azevedo, C., and Schulze-Lefert, P. (1999). A novel class of eukaryotic zincbinding proteins is required for disease resistance signaling in barley and development in C. elegans. Cell 99, 355–366.
Takahashi, A., Kawasaki, T., Henmi, K., Shil, K., Kodama, O., Satoh, H., and Shimamoto, K. (1999). Lesion mimic mutants of rice with alterations in early signaling events of defense. Plant J. 17, 535–545.
Takahashi, A., Casais, C., Ichimura, K., and Shirasu, K. (2003). HSP90 interacts with RAR1 and SGT1 and is essential for RPS2-mediated disease resistance in Arabidopsis. Proc. Natl. Acad. Sci. USA 100, 11777–11782.
Thomma, B.P., Nurnberger, T., and Joosten, M.H. (2011). Of PAMPs and effectors: the blurred PTI-ETI dichotomy. Plant Cell 23, 4–15.
Tornero, P., Merritt, P., Sadanandom, A., Shirasu, K., Innes, R.W., and Dangl, J.L. (2002). RAR1 and NDR1 contribute quantitatively to disease resistance in Arabidopsis, and their relative contributions are dependent on the R gene assayed. Plant Cell 14, 1005–1015.
Tsunematsu, H., Yanoria, M.J.T., Ebron, L.A., Hayashi, N., Ando, I. Kato, H., Imbe, T., and Khush, G.S. (2000). Development of monogenic lines of rice for blast resistance. Breed. Sci. 50, 229–234.
Wang, Z.X., Yano, M., Yamanouchi, U., Iwamoto, M., Monna, L., Hayasaka, H., Katayose, Y., and Sasaki, T. (1999). The Pib gene for rice blast resistance belongs to the nucleotide binding and leucine-rich repeat class of plant disease resistance genes. Plant J. 19, 55–64.
Wang, Y., Gao, M., Li, Q., Wang, L., Wang, J., Jeon, J.S., Qu, N., Zhang, Y., and He, Z. (2008). OsRAR1 and OsSGT1 physically interact and function in rice basal disease resistance. Mol. Plant Microbe Interact. 21, 294–303.
Yi, G., Lee, S.K., Hong, Y.K., Cho, Y.C., Nam, M.H., Kim, S.C., Han, S.S., Wang, G.L., Hahn, T.R., Ronald, P.C., et al. (2004). Use of Pi5(t) markers in marker-assisted selection to screen for cultivars with resistance to Magnaporthe grisea. Theor. Appl. Genet. 109, 978–985.
Zipfel, C., Robatzek, S., Navarro, L., Oakeley, E.J., Jones, J.D., Felix, G., and Boller, T. (2004). Bacterial disease resistance in Arabidopsis through flagellin perception. Nature 428, 764–767.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Song, MY., Kim, CY., Han, M. et al. Differential requirement of Oryza sativa RAR1 in immune receptor-mediated resistance of rice to Magnaporthe oryzae . Mol Cells 35, 327–334 (2013). https://doi.org/10.1007/s10059-013-2317-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-2317-6