Abstract
The authors describe an electrochemical aptasensor for thrombin that is based on the use of a glassy carbon electrode (GCE) modified with polydopamine that is loaded with silver nanoparticles (PDA/AgNPs). The use of AgNPs improves the conductivity of the film and increases the surface area of the GCE. PDA was deposited on the GCE via self-polymerization, and the thrombin binding aptamer was grafted onto the PDA-modified GCE by a single step reaction. Residual electrode surface was blocked with 6-mercapto-1-hexanol. On exposure to thrombin, the electrochemical impedance of the modified electrode increases gradually. Response is linear in the 0.1 pM to 5.0 nM thrombin concentration range, and the limit of detection is as low as 36 fM. The method is selective and capable of detecting thrombin in diluted human serum. In our perception, such a GCE modified with AgNP in a PDA matrix may be applied to many other analytes for which appropriate aptamers are available.

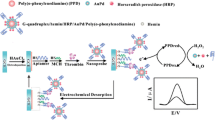
Schematic of an electrochemical aptasensor for sensitive and selective thrombin detection based on the use of a self-polymerized polydopamine film loaded with silver nanoparticles.






Similar content being viewed by others
References
Shen G, Zhang H, Yang C, Yang Q, Tang Y (2017) Thrombin ultrasensitive detection based on chiral supramolecular assembly signal-amplified strategy induced by thrombin-binding aptamer. Anal Chem 89(1):548–551
Xu W, Xue S, Yi H, **g P, Chai Y, Yuan R (2015) A sensitive electrochemical aptasensor based on the co-catalysis of hemin/G-quadruplex, platinum nanoparticles and flower-like MnO2 nanosphere functionalized multi-walled carbon nanotubes. Chem Commun 51(8):1472–1474
Zhao D, Peng Y, Xu L, Zhou W, Wang Q, Guo L (2015) Liquid-crystal biosensor based on nickel-nanosphere-induced homeotropic alignment for the amplified detection of thrombin. ACS Appl Mater Interfaces 7(42):23418–23422
Chen Z, Tan L, Hu L, Zhang Y, Wang S, Lv F (2015) Real colorimetric thrombin Aptasensor by masking surfaces of catalytically active gold nanoparticles. ACS Appl Mater Interfaces 8(1):102–108
Li S, Zhang D, Zhang Q, Lu Y, Li N, Chen Q, Liu Q (2016) Electrophoresis-enhanced localized surface plasmon resonance sensing based on nanocup array for thrombin detection. Sens Actuators B: Chem 232:219–225
Ma M, Zheng X (2015) Preparation of brightly fluorescent silica nanoparticles modified with lucigenin and chitosan, and their application to an aptamer-based sandwich assay for thrombin. Microchim Acta 182(13–14):2193–2199
Wang J, Jiang X, Han H (2016) Turn-on near-infrared electrochemiluminescence sensing of thrombin based on resonance energy transfer between CdTe/CdS coresmall/shellthick quantum dots and gold nanorods. Biosens Bioelectron 82:26–31
Tang D, Tang J, Li Q, Su B, Chen G (2011) Ultrasensitive aptamer-based multiplexed electrochemical detection by coupling distinguishable signal tags with catalytic recycling of DNase I. Anal Chem 83(19):7255–7259
Huang Y, Chen J, Zhao S, Shi M, Chen ZF, Liang H (2013) Label-free colorimetric aptasensor based on nicking enzyme assisted signal amplification and DNAzyme amplification for highly sensitive detection of protein. Anal Chem 85(9):4423–4430
Fenzl C, Nayak P, Hirsch T, Wolfbeis OS, Alshareef HN, Baeumner AJ (2017) Laser-scribed graphene electrodes for aptamer-based biosensing. ACS Sens 2(5):616–620
Liu B, Cui Y, Tang D, Yang H, Chen G (2012) Au(III)-assisted core-shell iron oxide@poly(o-phenylenediamine) nanostructures for ultrasensitive electrochemical aptasensors based on DNase I-catalyzed target recycling. Chem Commun 48(20):2624–2626
Chen S, Liu P, Su K, Li X, Qin Z, Xu W, Chen J, Li C, Qiu J (2018) Electrochemical aptasensor for thrombin using co-catalysis of hemin/G-quadruplex DNAzyme and octahedral Cu2O-au nanocomposites for signal amplification. Biosens Bioelectron 99:338–345
Wang L, Ma R, Jiang L, Jia L, Jia W, Wang H (2017) A novel "signal-on/off" sensing platform for selective detection of thrombin based on target-induced ratiometric electrochemical biosensing and bio-bar-coded nanoprobe amplification strategy. Biosens Bioelectron 92:390–395
Yang J, Dou B, Yuan R, **ang Y (2017) Aptamer/protein proximity binding-triggered molecular machine for amplified electrochemical sensing of thrombin. Anal Chem 89(9):5138–5143
Wang X, Duan** S, Tong Y, Zhong Y, Chen Z (2017) A voltammetric aptamer-based thrombin biosensor exploiting signal amplification via synergetic catalysis by DNAzyme and enzyme decorated AuPd nanoparticles on a poly(o-phenylenediamine) support. Microchim Acta 184:1791–1977
Ocana C, del Valle M (2016) Three different signal amplification strategies for the impedimetric sandwich detection of thrombin. Anal Chim Acta 912:117–124
Lee H, Dellatore SM, Miller WM, Messersmith PB (2007) Mussel-inspired surface chemistry for multifunctional coatings. Science 318(5849):426–430
Cui J, Ju Y, Liang K, Ejima H, Lörcher S, Gause KT, Richardson JJ, Caruso F (2014) Nanoscale engineering of low-fouling surfaces through polydopamine immobilisation of zwitterionic peptides. Soft Matter 10(15):2656–2663
Ye Q, Zhou F, Liu W (2011) Bioinspired catecholic chemistry for surface modification. Chem Soc Rev 40(7):4244–4258
Li P, Cai X, Wang D, Chen S, Yuan J, Li L, Shen J (2013) Hemocompatibility and anti-biofouling property improvement of poly(ethylene terephthalate) via self-polymerization of dopamine and covalent graft of zwitterionic cysteine. Colloids Surf B 110:327–332
Liu Y, Ai K, Lu L (2014) Polydopamine and its derivative materials: synthesis and promising applications in energy, environmental, and biomedical fields. Chem Rev 114:5057–5115
Proks V, Brus J, Pop-Georgievski O, Večerníková E, Wisniewski W, Kotek J, Urbanová M, Rypáček F (2013) Thermal-induced transformation of Polydopamine structures: an efficient route for the stabilization of the Polydopamine surfaces. Macromol Chem Phys 214(4):499–507
Sileika TS, Kim HD, Maniak P, Messersmith PB (2011) Antibacterial performance of polydopamine-modified polymer surfaces containing passive and active components. ACS Appl Mater Interfaces 3(12):4602–4610
Cui M, Song Z, Wu Y, Guo B, Fan X, Luo X (2016) A highly sensitive biosensor for tumor maker alpha fetoprotein based on poly (ethylene glycol) doped conducting polymer PEDOT. Biosens Bioelectron 79:736–741
Cui M, Wang Y, Wang H, Wu Y, Luo X (2017) A label-free electrochemical DNA biosensor for breast cancer marker BRCA1 based on self-assembled antifouling peptide monolayer. Sens Actuators B: Chem 244:742–749
Wang X, Sun D, Tong Y, Zhong Y, Chen Z (2017) A voltammetric aptamer-based thrombin biosensor exploiting signal amplification via synergetic catalysis by DNAzyme and enzyme decorated AuPd nanoparticles on a poly(o-phenylenediamine) support. Microchimic Acta 184(6):1791–1799
Hao L, Zhao Q (2016) Microplate based assay for thrombin detection using an RNA aptamer as affinity ligand and cleavage of a chromogenic or a fluorogenic peptide substrate. Microchimic Acta 183(6):1891–1898
Zhang L, Li L (2016) Colorimetric thrombin assay using aptamer-functionalized gold nanoparticles acting as a peroxidase mimetic. Microchimic Acta 183(1):485–490
Li Y, Li Y, Xu N, Pan J, Chen T, Chen Y, Gao W (2017) Dual-signal amplification strategy for electrochemiluminescence sandwich biosensor for detection of thrombin. Sens Actuators B: Chem 240:742–748
Heydari-Bafrooei E, Amini M, Ardakani MH (2016) An electrochemical aptasensor based on TiO2/MWCNT and a novel synthesized Schiff base nanocomposite for the ultrasensitive detection of thrombin. Biosens Bioelectron 85:828–836
Wang Y, Zhang Y, Yan T, Fan D, Du B, Ma H, Wei Q (2016) Ultrasensitive electrochemical aptasensor for the detection of thrombin based on dual signal amplification strategy of au@GS and DNA-CoPdNPs conjugates. Biosens Bioelectron 80:640–646
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOC 414 kb)
Rights and permissions
About this article
Cite this article
Xu, Q., Wang, G., Zhang, M. et al. Aptamer based label free thrombin assay based on the use of silver nanoparticles incorporated into self-polymerized dopamine. Microchim Acta 185, 253 (2018). https://doi.org/10.1007/s00604-018-2787-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2787-5