Abstract
The authors describe a new coating for use in electrochemically controlled in-tube solid phase microextraction (EC-IT-SPME). It consists of a nanocomposite that was prepared from polypyrrole and deep eutectic solvent (DES) by electrochemical deposition on the inner walls of a stainless steel capillary that serves as a working electrode. The hypertension drug losartan acts as an acidic model analyte. The extraction efficiency, mechanical stability, chemical stability and lifetime of the coating were investigated. It is found to be quite stable in relatively acidic and basic media and to be re-usable >450 times without decrease in extraction efficiency. Its extraction capability in comparison to the commercial polypyrrole coating is better by a factor of 1.5. The coated steel capillary was used as the anode (anion-exchanger), and a platinum electrode was used as the cathode. By passing a sample solution through the electrode, losartan can be extracted by applying a positive potential to the flow. In the next step, losartan is electrochemically desorbed and subjected to HPLC analysis with UV detection. Under optimal conditions, losartan can be quantified with limits of detection that range from 50 to 500 ng L−1 depending on the sample matrix. Response is linear in the 0.1–500 μg L−1 concentration range. The inter- and intra-assay precisions (RSDs; in %, for n = 3) are in the range from 2.4–4.6% and from 1.9–3.9%, respectively.

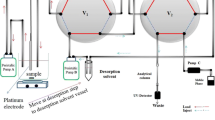
Schematic of the preparation of a nano-structured polypyrrole-deep eutectic solvent nanocomposite coating placed on the inner surface of a stainless steel capillary and used for electrochemically controlled in-tube solid phase microextraction of losartan from biological samples.




Similar content being viewed by others
References
Eisert R, Pawliszyn J (1997) Automated in-tube solid-phase microextraction coupled to high-performance liquid chromatography. Anal Chem 69:3140–3147. https://doi.org/10.1021/ac970319a
Kataoka H (2002) Automated sample preparation using in-tube solid-phase microextraction and its application - a review. Anal Bioanal Chem 373:31–45. https://doi.org/10.1007/s00216-002-1269-z
Wu J, Tragas C, Lord H, Pawliszy J (2002) Analysis of polar pesticides in water and wine samples by automated in-tube solid-phase microextraction coupled with high-performance liquid chromatography–mass spectrometry. J Chromatogr A 976:357–367. https://doi.org/10.1016/S0021-9673(02)01072-5
Queiroz MEC, Melo LP (2014) Selective capillary coating materials for in-tube solid-phase microextraction coupled to liquid chromatography to determine drugs and biomarkers in biological samples: a review. Anal Chim Acta 826:1–11. https://doi.org/10.1016/j.aca.2014.03.024
Melo LP, Queiroz RHC, Queiroz MEC (2011) Automated determination of rifampicin in plasma samples by in-tube solid-phase microextraction coupled with liquid chromatography. J Chromatogr B 879:2454–2458. https://doi.org/10.1016/j.jchromb.2011.06.041
Asiabi H, Yamini Y, Seidi S, Esrafili A, Rezaei F (2015) Electroplating of nanostructured polyaniline–polypyrrole composite coating in a stainless-steel tube for on-line in-tube solid phase microextraction. J Chromatogr A 1397:19–26. https://doi.org/10.1016/j.chroma.2015.04.015
Asiabi H, Yamini Y, Seidi S, Safari M, Shamsayei M (2016) Evaluation of in-tube solid-phase microextraction method for co-extraction of acidic, basic, and neutral drugs. RSC Adv 6:14049–14058. https://doi.org/10.1039/C5RA27825B
Asiabi H, Yamini Y, Rezaei F, Seidi S (2015) Nanostructured polypyrrole for automated and electrochemically controlled in-tube solid-phase microextraction of cationic nitrogen compounds. Microchim Acta 182:1941–1948. https://doi.org/10.1007/s00604-015-1534-4
Asiabi H, Yamini Y, Seidi S, Shamsayei M, Safari M, Rezaei F (2016) On-line electrochemically controlled in-tube solid phase microextraction of inorganic selenium followed by hydride generation atomic absorption spectrometry. Anal Chim Acta 922:37–47. https://doi.org/10.1016/j.aca.2016.04.001
Bagheri H, Aghakhani A, Akbari M, Ayazi Z (2011) Electrospun composite of polypyrrole polyamide as a micro-solid phase extraction sorbent. Anal Bioanal Chem 400:3607–3613
Zhao S, Wu M, Zhao F, Zeng B (2013) Electrochemical preparation of polyaniline–polypyrrole solid-phase microextraction coating and its application in the GC determination of several esters. Talanta 117:146–151. https://doi.org/10.1007/s00216-011-4993-4
Wu J, Pawliszyn J (2004) Solid-phase microextraction based on polypyrrole films with different counter ions. Anal Chim Acta 520:257–264. https://doi.org/10.1016/j.aca.2004.05.019
Saraji M, Rezaei B, Boroujeni MK, Bidgoli AAH (2013) Polypyrrole/sol–gel composite as a solid-phase microextraction fiber coating for the determination of organophosphorus pesticides in water and vegetable samples. J Chromatogr A 1279:20–26. https://doi.org/10.1016/j.chroma.2013.01.017
Sarafraz-Yazdi A, Rounaghi G, Razavipanah I, Vatani H, Amiri A (2014) New polypyrrole carbon nanotubes–silicon dioxide solid-phase microextraction fiber for the preconcentration and determination of benzene, toluene, ethylbenzene, and o-xylene using gas liquid chromatography. J Sep Sci 37:2605–2612. https://doi.org/10.1002/jssc.201400178
Zhu Q, Gao F, Yang Y, Zhang B, Wang W, Hu Z, Wang Q (2015) Electrochemical preparation of polyaniline capped Bi2S3nanocomposite and its application in impedimetric DNA biosensor. Sensors Actuators B Chem 207:819–826. https://doi.org/10.1016/j.snb.2014.10.120
Ho TD, Canestraro AJ, Anderson JL (2011) Ionic liquids in solid-phase microextraction: a review. Anal Chim Acta 695:18–43. https://doi.org/10.1016/j.aca.2011.03.034
Favre C, Abello L, Delabouglise D (1997) Effect of the anion on the level and aging of the conducting state of electropolymerized 3-methylthiophene thin films. Adv Mater 9:722–725. https://doi.org/10.1002/adma.19970090909
Lu W, Mattes BR (2005) Factors influencing electrochemical actuation of polyaniline fibers in ionic liquids. Synth Met 152:53–56. https://doi.org/10.1016/j.synthmet.2005.07.122
Ai Y, Zhao F, Zeng B (2015) Novel proton-type ionic liquid doped polyaniline for the headspace solid-phase microextraction of amines. Anal Chim Acta 880:60–66. https://doi.org/10.1016/j.aca.2015.04.028
Wu M, Zhang H, Zhao F, Zeng B (2014) A novel poly(3,4-ethylenedioxythiophene)-ionic liquid composite coating for the headspace solid-phase microextraction and gas chromatography determination of several alcohols in soft drinks. Anal Chim Acta 850:41–48. https://doi.org/10.1016/j.aca.2014.08.029
Zhao F, Wang M, Ma Y, Zeng B (2011) Electrochemical preparation of polyaniline–ionic liquid based solid phase microextraction fiber and its application in the determination of benzene derivatives. J Chromatogr A 1218:387–391. https://doi.org/10.1016/j.chroma.2010.12.017
Leron RB, Li M (2013) Solubility of carbon dioxide in a choline chloride–ethylene glycol based deep eutectic solvent. Thermochim Acta 551:14–19
García A, Rodríguez-Juan E, Rodríguez-Gutiérrez G, Rios JJ, Fernández-Bolaños J (2016) Extraction of phenolic compounds from virgin olive oil by deep eutectic solvents (DESs). Food Chem 197:554–561. https://doi.org/10.1016/j.foodchem.2015.10.131
Singh B, Lobo H, Shankarling G (2011) Selective n-alkylation of aromatic primary amines catalyzed by bio-catalyst or deep eutectic solvent. Catal Lett 141:178–182. https://doi.org/10.1007/s10562-010-0479-9
Leron RB, Li M (2012) High-pressure density measurements for choline chloride: urea deep eutectic solvent and its aqueous mixtures at T = (298.15 to 323.15) K and up to 50 MPa. J Chem Thermodynamics 54:293–301. https://doi.org/10.1016/j.jct.2012.05.008
Jhong H, Wong D, Wan C, Wang Y, Wei T (2009) A novel deep eutectic solvent-based ionic liquid used as electrolyte for dye-sensitized solar cells. Electrochem Commun 11:209–211. https://doi.org/10.1016/j.elecom.2008.11.001
Abbott AP, Capper G, McKenzie KJ, Ryder KS (2007) Electrodeposition of zinc–tin alloys from deep eutectic solvents based on choline chloride. J Electroanal Chem 599:288–294. https://doi.org/10.1016/j.jelechem.2006.04.024
Matuszewski BK, Constanzer ML, Chavez-Eng CM (2003) Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal Chem 75:3019–3030. https://doi.org/10.1021/ac020361s
Zhang M, Wei F, Zhang Y, Nie J, Feng Y (2006) Novel polymer monolith microextraction using a poly(methacrylic acid-ethylene glycol dimethacrylate) monolith and its application to simultaneous analysis of several angiotensin II receptor antagonists in human urine by capillary zone electrophoresis. J Chromatogr A 1102:294–301. https://doi.org/10.1016/j.chroma.2005.10.057
Nie J, Zhang M, Fan Y, Wen Y, **ang B, Feng Y (2005) Biocompatible in-tube solid-phase microextraction coupled to HPLC for the determination of angiotensin II receptor antagonists in human plasma and urine. J Chromatogr B 828:62–69. https://doi.org/10.1016/j.jchromb.2005.09.015
Rao RN, Raju SS, Vali RM (2013) Ionic-liquid based dispersive liquid–liquid microextraction followed by high performance liquid chromatographic determination of anti-hypertensives in rat serum. J Chromatogr B 931:174–180. https://doi.org/10.1016/j.aca.2009.09.044
Acknowledgements
Financial support from Tarbiat Modares University is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no conflict of interests.
Electronic supplementary material
ESM 1
(DOCX 547 kb)
Rights and permissions
About this article
Cite this article
Asiabi, H., Yamini, Y., Shamsayei, M. et al. A nanocomposite prepared from a polypyrrole deep eutectic solvent and coated onto the inner surface of a steel capillary for electrochemically controlled microextraction of acidic drugs such as losartan. Microchim Acta 185, 169 (2018). https://doi.org/10.1007/s00604-018-2684-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2684-y