Abstract
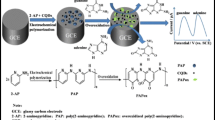
A glassy carbon electrode (GCE) was modified with graphene quantum dots (GQDs) carrying silver nanoparticles. The modified GCE displays excellent performance in the electrochemical oxidation of guanine and adenine in showing lower anodic peak overpotentials (of 0.625 and 0.929 V, respectively) and increased anodic peak currents. The effective surface area of the modified GCE is about 21.5 times larger than that of the bare GCE. The electron transfer coefficient (α) and the electron transfer number (n) were calculated to be 2 and 0.60 for guanine, and 2 and 0.64 for adenine, respectively. Linear relationships between peak current and the concentrations were obtained in the range from 0.015 to 430 μM (for guanine) and 0.015 to 390 μM (for adenine). The detection limits are 10 nM and 12 nM (at a signal-to-noise ratio of 3), respectively. The modified GCE can well distinguish between guanine and adenine in mixed solutions, has fast response, a low detection limit, good reproducibility, and high stability.

A glassy carbon novel electrode modified with graphene quantum dots and silver nanoparticles was prepared. The modified electrode displays excellent performance in the electrochemical oxidation of guanine and adenine with fast response, a low detection limit, good reproducibility and stability






Similar content being viewed by others
References
Lu CH, Yang HH, Zhu CL, Chen X, Chen GN (2009) A graphene platform for sensing biomolecules. Angew Chem Int Ed 48:4785–4787
Alwarappan S, Erdem A, Liu C, Li CZ (2009) Probing the electrochemical properties of graphene nanosheets for biosensing applications. J Phys Chem C 113:8853–8857
Zhang Y, Wu CY, Zhou XJ, Wu XC, Yang YQ, Wu HX, Guo SW, Zhang JY (2013) Graphene quantum dots/gold electrode and its application in living cell H2O2 detection. Nanoscale 5:1816–1819
Sun HJ, Wu L, Wei WL, Qu XG (2013) Recent advances in graphene quantum dots for sensing. Mater Today 16:433–442
Shen J, Zhu Y, Yang X, Li C (2012) Graphene quantum dots: emergent nanolights for bioimaging, sensors, catalysis and photovoltaic devices. Chem Commun 48:3686–3699
Li LL, Wu GH, Yang GH, Peng J, Zhao JW, Zhu JJ (2013) Focusing on luminescent graphene quantum dots: current status and future perspectives. Nanoscale 5:4015–4039
Peng J, Gao W, Gupta BK, Liu Z, Romero-Aburto R, Ge LH, Song L, Alemany LB, Zhan XB, Gao GH, Vithayathil SA, Kaipparettu BA, Marti AA, Hayashi T, Zhu JJ, Ajayan PM (2012) Graphene quantum dots derived from carbon fibers. Nano Lett 12:844–849
Cheng HH, Zhao Y, Fan YQ, **e XJ, Qu LT, Shi GQ (2012) Graphene quantum dot assembled nanotubes: A new platform for efficient Raman enhancement. ACS Nano 6:2237–2244
Tang LB, Ji RB, Cao XK, Lin JY, Jiang HX, Li XM, Teng KS, Luk CM, Zeng SJ, Hao JH, Lau SP (2012) Deep ultraviolet photoluminescence of water-soluble self-passivated graphene quantum dots. ACS Nano 6:5102–5110
Zhao J, Chen GF, Zhu L, Li GX (2011) Graphene quantum dots-based platform for the fabrication of electrochemical biosensors. Electrochem Commun 13:31–33
Gan T, Hu SS (2011) Electrochemical sensors based on graphene materials. Microchim Acta 175:1–19
Mao SX, Li WF, Long YM, Tu YF, Deng AP (2012) Sensitive electrochemical sensor of tryptophan based on Ag@C core-shell nanocomposite modified glassy carbon electrode. Anal Chim Acta 738:35–40
Rounaghi G, kakhki RM, Azizi-toupkanloo H (2012) Voltammetric determination of 4-nitrophenol using a modified carbon paste electrode based on a new synthetic crown ether/silver nanoparticles. Mater Sci Eng C 32:172–177
Wang WP, Zhou L, Wang SM, Luo Z, Hu ZD (2008) Rapid and simple determination of adenine and guanine in DNA extract by micellar electrokinetic chromatography with indirect laser-induced fluorescence detection. Talanta 74:1050–1055
Zen JM, Chang MR, Ilangovan G (1999) Simultaneous determination of guanine and adenine contents in DNA, RNA and synthetic oligonucleotides using a chemically modified electrode. Analyst 124:679–684
Yang FQ, Guan J, Li SP (2007) Fast simultaneous determination of 14 nucleosides and nucleobases in cultured cordyceps using ultra- performance liquid chromatography. Talanta 73:269–273
**ao F, Zhao FQ, Li JW, Liu LQ, Zeng BZ (2008) Characterization of hydrophobic ionic liquid-carbon nanotubes-gold nanoparticles composite film coated electrode and the simultaneous voltammetric determination of guanine and adenine. Electrochim Acta 53:7781–7788
Liu T, Zhu XB, Cui L, Ju P, Qu XJ, Ai SY (2011) Simultaneous determination of adenine and guanine utilizing PbO2-carbon nanotubes-ionic liquid composite film modified glassy carbon electrode. J Electroanal Chem 651:216–221
Liu HY, Wang GF, Chen DL, Zhang W, Li CJ, Fang B (2008) Fabrication of polythionine/NPAu/MWNTs modified electrode for simultaneous determination of adenine and guanine in DNA. Sensor Actuat B 128:414–421
Huang KJ, Niu DJ, Sun JY, Han CH, Wu ZW, Li YL, **ong XQ (2011) Novel electrochemical sensor based on functionalized graphene for simultaneous determination of adenine and guanine in DNA. Colloid Surf B-Biointerfaces 82:543–549
Marmur J, Rownd R, Schildkraut CL (1963) Progress in nucleic acid research. Academic, New York
Laviron E (1974) Adsorption, autoinhibition and autocatalysis in polarography and in linear potential sweep voltammetry. J Electroanal Chem 52:355–393
Adams RN (1969) Electrochemistry at solid electrodes. Marcel Dekker, New York
Anson FC (1964) Application of potentiostatic current integration to the study of the adsorption of cobalt(III)-(ethylenedinitrilo(tetraacetate) on mercury electrodes. Anal Chem 36:932–934
Sun W, Li YZ, Duan YY, Jiao K (2008) Direct electrocatalytic oxidation of adenine and guanine on carbon ionic liquid electrode and the simultaneous determination. Biosens Bioelectron 24:988–993
Zou LN, Li YM, Ye BX (2011) Voltammetric sensing of guanine and adenine using a glassy carbon electrode modified with a tetraoxocalix[2] arene[2]triazine Langmuir-Blodgett film. Microchim Acta 173:285–291
Wei YL, Luo LQ, Ding YP, Liu X, Chu YL (2013) A glassy carbon electrode modified with poly(eriochrome black T) for sensitive determination of adenine and guanine. Microchim Acta 180:887–893
Davision N (1972) The Biochemistry of the Nucleic Acids, 7th edn. Cox & Nyman, Norfolk
Acknowledgments
This work was supported by the HongLiu Natural Science Foundation of Lanzhou University of Technology and the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 237 kb)
Rights and permissions
About this article
Cite this article
Wang, G., Shi, G., Chen, X. et al. A glassy carbon electrode modified with graphene quantum dots and silver nanoparticles for simultaneous determination of guanine and adenine. Microchim Acta 182, 315–322 (2015). https://doi.org/10.1007/s00604-014-1335-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-014-1335-1