Abstract
Purpose
The clinicopathological features of colon cancer differ between proximal and distal sites; however, the influence of tumor location on liver metastasis has not been fully examined. The aim of this study was to evaluate the differences in the features of liver metastasis between proximal and distal colon cancer.
Methods
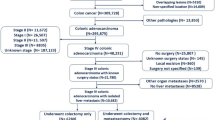
The clinicopathological data from 931 colon cancer patients who were treated surgically were examined retrospectively using a multivariate analysis.
Results
The incidence of synchronous liver metastasis was 7.1% (31/438) in proximal colon patients and 11.6% (57/493) in distal colon patients. Both univariate and multivariate analyses showed distal colon cancer to be a risk factor for synchronous liver metastasis.
Conclusion
The incidence of synchronous liver metastasis differs between proximal and distal colon cancer.
Similar content being viewed by others
References
Scheele J, Stangl R, Altendorf-Hofmann A. Hepatic metastases from colorectal carcinoma: impact of surgical resection on the natural history. Br J Surg. 1990;77:1241–6.
August DA, Sugarbaker PH, Ottow RT, Gianola FJ, Schneider PD. Hepatic resection of colorectal metastasis: influence of clinical factors and adjuvant intraperitoneal 5-fluorouracil via Tenchhoff catheter on survival. Ann Surg. 1985;201:210–8.
Takata O, Kawamura YJ, Konishi F, Sasaki J, Kai T, Miyakura Y, et al. cDNA array analysis for prediction of hepatic metastasis of colorectal carcinoma. Surg Today. 2006;36(7):608–14.
Iacopetta B. Are there two sides to colorectal cancer? Int J Cancer. 2002;101:403–8.
Bufill JA. Colorectal cancer: evidence for distinct genetic categories based on proximal or distal tumor location. Ann Intern Med. 1990;11:779–88.
Komuro K, Tada M, Tamoto E, Kawakami A, Matsunaga A, Teramoto K, et al. Right- and left-sided colorectal cancers display distinct expression profiles and the anatomical stratification allows a high accuracy prediction of lymph node metastasis. J Surg Res. 2005;124:216–24.
Ikeda Y, Mori M, Yoshizumi T, Sugimachi K. Cancer and adenomatous polyp distribution in the colorectum. Am J Gastroenterol. 1999;94:191–3.
Benedix F, Kube R, Meyer F, Schmidt U, Gastinger I, Lippert H, et al. Comparison of 17,641 patients with right- and left-sided colon cancer: differences in epidemiology, perioperative course, histology, and survival. Dis Colon Rectum. 2010;53(1):57–64.
Tsai MS, Su YH, Ho MC, Liang JT, Chen TP, Lai HS, et al. Clinicopathological features and prognosis in resectable synchronous and metachronous colorectal liver metastasis. Ann Surg Oncol. 2007;14(2):786–94.
Wigmore SJ, Madhavan K, Redhead DN, Currie EJ, Garden OJ. Distribution of colorectal liver metastases in patients referred for hepatic resection. Cancer (Phila). 2000;89(2):285–7.
Sobin LH, Witedkind CH. UICC TNM classification of malignant tumors. 5th ed. New York: Wiley-Liss; 1997. p. 66–9.
Beart RW, Melto LJ 3rd, Maruta M, Dockerty MB, Frydenberg HB, O’Fallen WM. Trends in right and left-sided colon cancer. Dis Colon Rectum. 1983;63:1888–91.
Fleshner P, Slater G, Aufses AH Jr. Age and sex distribution of patients with colorectal cancer. Dis Colon Rectum. 1989;32:107–11.
Ghahremani GG, Dowlatshahi K. Colorectal carcinomas: diagnostic implications of their changing frequency and anatomic distribution. World J Surg. 1989;13:321–4.
Jass JR. Subsite distribution and incidence of colorectal cancer in New Zealand, 1974–1983. Dis Colon Rectum. 1991;34:56–9.
Levi F, Randimbison L, La Vecchia C. Trends in subsite distribution of colorectal cancers and polyps from Vaud Cancer Registry. Cancer (Phila). 1993;72:46–50.
Ikeda Y, Koyanagi N, Mori M, Ezaki T, Toyomasu T, Minagawa S, et al. Increased incidence of proximal colon cancer in the elderly. J Clin Gastroenterol. 1996;23:105–8.
Cooper SG, Yuan Z, Landefeld SC, Johansen FJ, Rimm AA. A national-based study of incidence of colorectal cancer and age. Cancer (Phila). 1995;75:775–81.
Greenson JK, Bonner JD, Ben-Yzhak O, Cohen HI, Miselevich I, Resnick MB, et al. Phenotype of microsatellite unstable colorectal carcinomas: well-differentiated and focally mucinous tumors and the absence of dirty necrosis correlate with microsatellite instability. Am J Surg Pathol. 2003;27:563–70.
Adachi Y, Inomata M, Kakisako K, Sato K, Shiraishi N, Kitano S. Histopathologic characteristics of colorectal cancer with liver metastasis. Dis Colon Rectum. 1999;42:1053–6.
Ouchi K, Sugawara T, Ono H, Fujiya T, Kamiyama Y, Kakugawa Y, et al. Histologic features and clinical significance of venous invasion in colorectal carcinoma with hepatic metastasis. Cancer (Phila). 1996;78:2313–7.
Hughes KS, Scheele J, Sugarbaker PH. Surgery for colorectal cancer metastatic to liver. Surg Clin North Am. 1989;69:339–59.
Chu DJ, Erickson AC, Russell PM, Thompson C, Lang NP, Broadwater RJ, et al. Prognostic significance of carcinoembryonic antigen in colorectal carcinoma. Arch Surg. 1991;126:314–6.
Zheng LD, Tong QS, Weng MX, He J, Lv Q, Pu JR, et al. Enhanced expression of resistin-like molecule beta in human colon cancer and its clinical significance. Dig Dis Sci. 2009;54(2):274–81.
Mitomi H, Mori A, Kanazawa H, Nishiyama Y, Ihara A, Otani Y, et al. Venous invasion and down-regulation of p21 (WAF1/CIP1) are associated with metastasis in colorectal carcinomas. Hepatogastroenterology. 2005;52(65):1421–6.
Sugarbaker PH. Metastatic inefficiency: the scientific basis for resection of liver metastases from colorectal cancer. J Surg Oncol Suppl. 1993;3:158–60.
Pantel K, Cote RJ, Fodstad O. Detection and clinical importance of micrometastatic disease. J Natl Cancer Inst. 1999;91:1113–4.
Hostetter RB, Augustus LB, Mankarious R, Chi KF, Fan D, Toth C, et al. Carcinoembryonic antigen as a selective enhancer of colorectal cancer metastasis. J Natl Cancer Inst. 1990;82:380–5.
Phillips RK, Hittinger R, Blesovsly L, Fry JS, Fielding LP. Large bowel cancer; surgical pathology and its relationship to survival. Br J Surg. 1984;71:604–10.
Wolmark N, Fisher B, Wieand HS. The prognostic value of the modifications of Dukes’ C class of colorectal cancer. Ann Surg. 1986;203:115–22.
Galandiuk S, Wieand HS, Moertel CG, Cha SS, Fitzgibbons RJ Jr, Pemberton JH, et al. Patterns of recurrence after curative resection of carcinoma of the colon and rectum. Surg Gynecol Obstet. 1992;174:27–32.
Ikeda Y, Akagi K, Kinoshita J, Abe T, Miyazaki M, Mori M, et al. Different distribution of Dukes’ stage between proximal and distal colorectal cancer. Hepatogastroenterology. 2002;49:1535–7.
Redson M. Carcinogenesis in the GI tract: from morphology to genetics and back again. Mod Pathol. 2001;230:309–18.
Distler P, Holt PR. Are right and left-sided colon neoplasms distinct tumors? Dig Dis. 1997;15:302–11.
Thibodeau SN, Bren G, Schaid D. Microsatellite instability in cancer of the proximal colon. Science. 1993;260:816–9.
Ikeda Y, Oda S, Ohno S, Maehara Y, Sugimachi K. Features of microsatellite instability in colorectal cancer: comparison between colon and rectum. Oncology. 2001;61:168–74.
Haddad R, Ogilvie RT, Croitoru M, Muniz V, Gryfe R, Pollet A, et al. Microsatellite instability as a prognostic factor in resected colorectal cancer liver metastases. Ann Surg Oncol. 2004;11:977–82.
Ookawa K, Sakamoto M, Hirohashi S, Yoshida Y, Sugimura T, Terada M, et al. Concordant p53 and DCC alterations and allelic losses on chromosomes 13q and 14q associated with liver metastases of colorectal carcinoma. Int J Cancer. 1993;53:382–7.
Kato M, Ito Y, Kobayashi S, Isono K. Detection of DCC and Ki-ras gene alterations in colorectal carcinoma tissue as prognostic markers for liver metastatic recurrence. Cancer (Phila). 1996;77:1729–35.
Conflict of interest
Daisuke Yoshida and co-authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, D., Ikeda, Y., Waki, K. et al. Different incidence of synchronous liver metastasis between proximal and distal colon cancer. Surg Today 42, 426–430 (2012). https://doi.org/10.1007/s00595-011-0056-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-011-0056-x