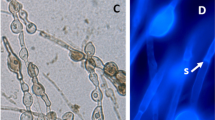
Abstract
The co-existence of two arbuscular mycorrhizal fungal (AMF) species, Glomus intraradices and Glomus claroideum, in the root systems of plants was investigated in a greenhouse experiment aimed at reconstructing interactions during an early stage of primary succession on a coal-mine spoil bank in Central Europe. Two plant species, Tripleurospermum inodorum and Calamagrostis epigejos, were inoculated either with one or both AMF species. Fungal development, determined by trypan blue and alkaline phosphatase staining as well as by PCR amplification of rRNA genes with species-specific primers, and the expression of five genes with different metabolic functions in the intraradical structures of G. intraradices were followed after 6 and 9 weeks of cultivation. The two AMF closely co-existed in the root systems of both plants possibly through similar colonisation rates and competitivity. Inoculation with the two fungi, however, did not bring any additional benefit to the host plants in comparison with single inoculation; moreover, plant growth depression observed after inoculation with G. claroideum persisted also in mixed inoculation. The expression of all the assayed G. intraradices genes was affected either by host plant or by co-inoculation with G. claroideum. The effects of both factors depended on the time of sampling, which underlines the importance of addressing this topic in time-course studies.


Similar content being viewed by others
References
Abbott LK, Robson AD (1983) Introduction of vesicular arbuscular mycorrhizal fungi into agricultural soils. Aust J Agric Res 34:741–749. doi:10.1071/AR9830741
Alkan N, Gadkar V, Yarden O, Kapulnik Y (2006) Analysis of quantitative interactions between two species of arbuscular mycorrhizal fungi, Glomus mosseae and G. intraradices, by real time PCR. Appl Environ Microbiol 72:4192–4199. doi:10.1128/AEM.02889-05
Aono T, Maldonado-Mendoza IE, Dewbre GR, Harrison MJ, Saito M (2004) Expression of alkaline phosphatase genes in arbuscular mycorrhizas. New Phytol 162:525–534. doi:10.1111/j.1469-8137.2004.01041.x
Balestrini R, Lanfranco L (2006) Fungal and plant gene expression in arbuscular mycorrhizal symbiosis. Mycorrhiza 16:509–524. doi:10.1007/s00572-006-0069-2
Boddington CL, Dodd JC (1998) A comparison of the development and metabolic activity of mycorrhizas formed by arbuscular mycorrhizal fungi from different genera on two tropical forage legumes. Mycorrhiza 8:149–157. doi:10.1007/s005720050228
Cordier C, Gianinazzi-Pearason V, Gianinazzi S (1996) An immunological approach for the study of spatial relationships between arbuscular mycorrhizal fungi in planta. In: Azcon-Aguilar C, Barea JM (eds) Mycorrhizas in integrated systems: from genes to plant development. European Commission, EUR 16758, Luxemburg, pp 189–194
Czechowski T, Bari RP, Stitt M, Scheible WR, Udvardi MK (2004) Real-time RT-PCR profiling of over 1400 Arabidopsis transcription factors: unprecedented sensitivity reveals novel root- and shoot-specific genes. Plant J 38:366–379. doi:10.1111/j.1365-313X.2004.02051.x
Delp G, Timonen S, Rosewarne GM, Barker SJ, Smith S (2003) Differential expression of Glomus intraradices genes in external mycelium and mycorrhizal roots of tomato and barley. Mycol Res 107:1083–1093. doi:10.1017/S0953756203008311
Edathil TT, Manian S, Udaiyan K (1996) Interaction of multiple VAM fungal species on root colonization, plant growth and nutrient status of tomato seedlings (Lycopersicon esculentum Mill). Agric Ecosyst Environ 59:63–68. doi:10.1016/0167-8809(96)0104-7
Franken P, Gnädinger F (1994) Analysis of parsley arbuscular endomycorrhiza-infection development and messenger-RNA levels of defense-related genes. Mol Plant Microbe Interact 7:612–620. doi:10.1094/MPMI-7-0612
Gollotte A, van Tuinen D, Atkinson D (2004) Diversity of arbuscular mycorrhizal fungi colonising roots of the grass species Agrostis capillaris and Lolium perenne in a field experiment. Mycorrhiza 14:111–117. doi:10.1007/s00572-003-0244-7
Guillemin JP, Orozco MO, Gianinazzi-Pearson V, Gianinazzi S (1995) Influence of phosphate fertilization on fungal alkaline-phosphatase and succinate-dehydrogenase activities in arbuscular mycorrhiza of soybean and pineapple. Agric Ecosyst Environ 53:63–69. doi:10.1016/0167-8809(94)00555-S
Helgason T, Merryweather JW, Denison J, Wilson P, Young JPW, Fitter AH (2002) Selectivity and functional diversity in arbuscular mycorrhizas of co-occurring fungi and plants from a temperate deciduous woodland. J Ecol 90:371–384. doi:10.1046/j.1365-2745.2001.00674.x
Hepper CM, Azcon-Aquilar C, Rosendahl S, Sen R (1988) Competition between three species of Glomus used as spatially separated introduced and indigenous mycorrhizal inocula for leek (Allium porrum L.). New Phytol 110:207–215. doi:10.1111/j.1469-8137.1988.tb00254.x
Hetrick BAD, Wilson GWT, Todd TC (1990) Differential responses of C3 and C4 grasses to mycorrhizal symbiosis, phosphorus fertilization, and soil microorganisms. Can J Bot 68:461–467. doi:10.1139/b90-061
Jacquot E, van Tuinen D, Gianinazzi S, Gianinazzi-Pearson V (2000) Monitoring species of arbuscular mycorrhizal fungi in planta and in soil by nested PCR application to the study of the impact of sewage sludge. Plant Soil 226:179–188. doi:10.1023/A:1026475925703
Jacquot-Plumey E, van Tuinen D, Chatagnier O, Gianinazzi S, Gianinazzi-Pearson V (2001) 25S rDNA-based molecular monitoring of glomalean fungi in sewage sludge-treated field plots. Environ Microbiol 3:525–531. doi:10.1111/j.1462-2920.2001.00219.x
Jansa J, Smith FA, Smith SE (2008) Are there benefits of simultaneous root colonization by different arbuscular mycorrhizal fungi? New Phytol 177:779–789. doi:10.1111/j.1469-8137.2007.02294.x
Koide RT (2000) Functional complementarity in the arbuscular mycorrhizal symbiosis. New Phytol 147:233–235. doi:10.1111/j.1469-8137.2000.00710.x
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–505. doi:10.1016/S0953-7562(89)80195-9
Munkvold L, Kjøller R, Vestberg M, Rosendahl S, Jakobsen I (2004) High functional diversity within species of arbuscular mycorrhizal fungi. New Phytol 164:357–364. doi:10.1111/j.1469-8137.2004.01169.x
Pearson JN, Abbott LK, Jasper DA (1993) Mediation of competition between 2 colonizing VA mycorrhizal fungi by the host plant. New Phytol 123:93–98. doi:10.1111/j.1469-8137.1993.tb04534.x
Pivato B, Mazurier S, Lemanceau P, Siblot S, Berta G, Mougel C, van Tuinen D (2007) Medicago species affect the community composition of arbuscular mycorrhizal fungi associated with roots. New Phytol 176:197–210. doi:10.1111/j.1469-8137.2007.02151.x
Püschel D, Rydlová J, Vosátka M (2007) Mycorrhiza influences plant community structure in succession on spoil banks. Basic Appl Ecol 8:510–520. doi:10.1016/j.baae.2006.09.002
Rhody D, Stommel M, Roeder C, Mann P, Franken P (2003) Differential RNA accumulation of two beta-tubulin genes in arbuscular mycorrhizal fungi. Mycorrhiza 13:137–142. doi:10.1007/s00572-002-0209-2
Seddas PMA, Arias CM, Arnould C, van Tuinen D, Godfroy O, Benhassou HA, Gouzy J, Morandi D, Dessaint F, Gianinazzi-Pearson V (2009) Symbiosis-related plant genes modulate molecular responses in an arbuscular mycorrhizal fungus during early root interactions. Mol Plant Microbe Interact 22:341–351. doi:10.1094/MPMI-22-3-0341
Smith SE, Read DJ (1997) Mycorrhizal symbiosis. Academic, London
Tisserant B, Gianinazzi-Pearson V, Gianinazzi S, Gollotte A (1993) In planta histochemical staining of fungal alkaline phosphatase activity for analysis of efficient arbuscular mycorrhizal infections. Mycol Res 97:245–250. doi:10.1016/S0953-7562(09)80248-7
Tisserant B, Gianinazzi S, Gianinazzi-Pearson V (1996) Relationships between lateral root order, arbuscular mycorrhiza development, and the physiological state of the symbiotic fungus in Platanus acerifolia. Can J Bot 74:1947–1955. doi:10.1139/b96-233
Trouvelot A, Kough JL, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un systeme radiculaire. Recherche de methodes d’estimation ayant une signification fonctionnelle. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and genetical aspects of mycorrhizae. INRA, Paris, pp 217–221
van der Heijden MGA, Boller T, Wiemken A, Sanders IR (1998) Different arbuscular mycorrhizal fungal species are potential determinants of plant community structure. Ecology 79:2082–2091. doi:10.1890/0012-9658(1998)079[2082:DAMFSA]2.0.CO;2
van Tuinen D, Jacquot E, Zhao B, Gollotte A, Gianinazzi-Pearson V (1998a) Characterization of root colonization profiles by a microcosm community of arbuscular mycorrhizal fungi using 25S rDNA-targeted nested PCR. Mol Ecol 7:879–887. doi:10.1046/j.1365-294x.1998.00410.x
van Tuinen D, Zhao B, Gianinazzi-Pearson V (1998b) PCR in studies of AM fungi: from primers to application. In: Varma AK (ed) Mycorrhiza Manual. Springer, Heidelberg, pp 387–399
Vierheilig H (2004) Further root colonization by arbuscular mycorrhizal fungi in already mycorrhizal plants is suppressed after a critical level of root colonization. J Plant Physiol 161:339–341. doi:10.1078/0176-1617-01097
Violi HA, Treseder KK, Menge JA, Wright SF, Lovatt CJ (2007) Density dependence and interspecific interactions between arbuscular mycorrhizal fungi mediated plant growth, glomalin production, and sporulation. Can J Bot 85:63–75. doi:1139/B06-151
Weidmann S, Sanchez L, Descombin J, Chatagnier O, Gianinazzi S, Gianinazzi-Pearson V (2004) Fungal elicitation of signal transduction-related plant genes precedes mycorrhiza establishment and requires the dmi3 gene in Medicago truncatula. Mol Plant–Microbe Interact 17:1385–1393. doi:094/MPMI.2004.17.12.1385
Wilson JM (1984) Competition for infection between vesicular arbuscular mycorrhizal fungi. New Phytol 97:427–435. doi:111/j.1469-8137.1984.tb03608.x
Wilson JM, Trinick MJ (1983) Infection development and interaction between vesicular-arbuscular mycorrhizal fungi. New Phytol 93:543–553. doi:10.1111/j.1469-8137.1983.tb02705.x
Zhu YG, Smith SE, Barrit AR, Smith FA (2001) Phosphorus (P) efficiencies and mycorrhizal responsiveness of old and modern wheat cultivars. Plant Soil 237:249–255. doi:10.1023/A:1013343811110
Acknowledgements
The authors are grateful to Dr. Judith Fehrer for the design of discriminating primers for the quantification of AMF in roots and valuable advice to this part of the work. We would also like to thank to Dr. Jana Rydlová and Dr. David Püschel for providing plant seeds and to Barbora Slouková for technical support. Financial support for this study was provided by the Ministry of Education, Youth and Sports of the Czech Republic (Grant 1 M0571 and 2005-06-059) and by the Grant Agency of the Academy of Sciences of the Czech Republic (Grant AV0Z60050516).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Janoušková, M., Seddas, P., Mrnka, L. et al. Development and activity of Glomus intraradices as affected by co-existence with Glomus claroideum in one root system. Mycorrhiza 19, 393–402 (2009). https://doi.org/10.1007/s00572-009-0243-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-009-0243-4