Abstract
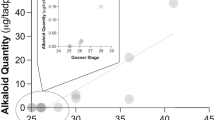
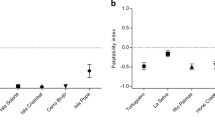
Within and among populations, alkaloid defenses of the strawberry poison frog (Oophaga pumilio) vary spatially, temporally, and with life history stage. Natural variation in defense has been implicated as a critical factor in determining the level of protection afforded against predators and pathogens. Oophaga pumilio tadpoles sequester alkaloids from nutritive eggs and are, thus, entirely dependent on their mothers for their defense. However, it remains unclear how tadpole alkaloid composition relates to that of its mother and how variation in maternally provisioned defenses might result in varying levels of protection against predators. Here, we demonstrate that natural variation in the alkaloid composition of a mother frog is reflected as variation in her tadpole’s alkaloid composition. Tadpoles, like mother frogs, varied in their alkaloid composition but always contained the identical alkaloids found in their mother. Alkaloid quantity in tadpoles was highly correlated with alkaloid quantity in their mothers. Additionally, alkaloid quantity was the best predictor of tadpole palatability, wherein tadpoles with higher alkaloid quantities were less palatable. Mother frogs with greater quantities of alkaloids are, thus, providing better protection for their offspring by provisioning chemical defenses during one of the most vulnerable periods of life.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Alonso-Mejía A, Brower LP (1994) From model to mimic: age-dependent unpalatability in monarch butterflies. Experientia 50:176–181. https://doi.org/10.1007/BF01984960
Basham EW, Saporito RA, González-Pinzón M, Romero-Marcucci A, Scheffers BR (2020) Chemical defenses shift with the seasonal vertical migration of a Panamanian poison frog. Biotropica 53:28–37. https://doi.org/10.1111/btp.12842
Bolton SK, Dickerson K, Saporito RA (2017) Variable alkaloid defenses in the dendrobatid poison frog Oophaga pumilio are perceived as differences in palatability to arthropods. J Chem Ecol 43:273–289. https://doi.org/10.1007/s10886-017-0827-y
Bowers MD, Williams EH (1995) Variable chemical defence in the checkerspot butterfly Euphydryas gillettii (Lepidoptera: NymphaIidae). Ecolog Entomol 20:208–212
Brown KS (1987) Chemistry at the Solanaceae/Ithomiinae interface. Ann Mo Bot Gard 74:359–397. https://doi.org/10.2307/2399406
Brown JL, Morales V, Summers K (2010) A key ecological trait drove the evolution of biparental care and monogamy in an amphibian. Am Nat 175:436–446. https://doi.org/10.1086/650727
Daly JW, Garraffo HM, Spande TF, Jaramillo C, Rand AS (1994) Dietary source for skin alkaloids of poison frogs (Dendrobatidae)? J Chem Ecol 20:943–955. https://doi.org/10.1007/BF02059589
Daly JW, Spande TF, Garraffo HM (2005) Alkaloids from amphibian skin: a tabulation of over eight-hundred compounds. J Nat Prod 68:1556–1575. https://doi.org/10.1021/np0580560
DeMarchi JA, Britton A, O’Donnell K, Saporito RA (2018) Behavioural preference for low levels of UV-B radiation in two neotropical frog species from Costa Rica. J Trop Ecol 34:336–340. https://doi.org/10.1017/S0266467418000287
Donnelly MA (1989a) Reproductive phenology and age structure of Dendrobates pumilio in northeastern Costa Rica. J Herpetol 23:362–367. https://doi.org/10.2307/1564047
Donnelly MA (1989b) Effects of reproductive resource supplementation on space-use patterns in Dendrobates pumilio. Oecologia 81:212–218. https://doi.org/10.1007/BF00379808
Dugas MB (2018) Simple observations with complex implications: what we have learned and can learn about parental care from a frog that feeds its young. Zool Anz 273:192–202. https://doi.org/10.1016/j.jcz.2017.11.012
Dugas MB, Wamelink CN, Killius AM, Richards-Zawacki CL (2016) Parental care is beneficial for offspring, costly for mothers, and limited by family size in an egg-feeding frog. Behav Ecol 27:476–483. https://doi.org/10.1093/beheco/arv173
Dugas MB, Strickler SA, Stynoski JL (2017) Tadpole begging reveals high quality. J Evol Biol 30:1024–1033. https://doi.org/10.5061/dryad.sf1s3
Dumbacher JP, Wako A, Derrickson SR, Samuelson A, Spande TF, Daly JW (2004) Melyrid beetles (Choresine): a putative source for the batrachotoxin alkaloids found in poison-dart frogs and toxic passerine birds. Proc Natl Acad Sci 101:15857–15860. https://doi.org/10.1073/pnas.0407197101
Dumbacher JP, Menon GK, Daly JW (2009) Skin as a toxin storage organ in the endemic New Guinean genus Pitohui. Auk 126:520–530. https://doi.org/10.1525/auk.2009.08230
Dyer LA, Dodson CD, Gentry G (2003) A bioassay for insect deterrent compounds found in plant and animal tissues. Phytochem Anal 14:381–388. https://doi.org/10.1002/pca.734
Eisner T, Eisner M, Rossini C, Iyengar VK, Roach BL, Benedikt E, Meinwald J (2000) Chemical defense against predation in an insect egg. Proc Natl Acad Sci 97:1634–1639. https://doi.org/10.1073/pnas.030532797
Fischer EK, Roland AB, Moskowitz NA, Vidoudez C, Ranaivorazo N, Tapia EE, Trauger SA, Vences M, Coloma LA, O’Connell LA (2019) Mechanisms of convergent egg provisioning in poison frogs. Curr Biol 29:4145–4151. https://doi.org/10.1016/j.cub.2019.10.032
Fordyce JA, Marion ZH, Shapiro AM (2005) Phenological variation in chemical defense of the pipevine swallowtail, Battus philenor. J Chem Ecol 31:2835–2846. https://doi.org/10.1007/s10886-005-8397-9
Gade MR, Hill M, Saporito RA (2016) Color assortative mating in a mainland population of the poison frog Oophaga pumilio. Ethology 122:851–858. https://doi.org/10.1111/eth.12533
González A, Hare JF, Eisner T (1999) Chemical egg defense in Photuris firefly ‘femmes fatales.’ Chemoecology 9:177–185. https://doi.org/10.1007/s000490050051
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190
Gunzburger MS, Travis J (2005) Critical literature review of the evidence for unpalatability of amphibian eggs and larvae. J Herpetol 39:547–571. https://doi.org/10.1670/1-05A.1
Haase A, Pröhl H (2002) Female activity patterns and aggressiveness in the strawberry poison frog Dendrobates pumilio (Anura: Dendrobatidae). Amphibia-Reptilia 23:129–140. https://doi.org/10.1163/156853802760061778
Hanifin CT, Brodie ED III, Brodie ED Jr (2003) Tetrodotoxin levels in eggs of the rough-skin newt, Taricha granulosa, are correlated with female toxicity. J Chem Ecol 29:1729–1739. https://doi.org/10.1023/A:1024885824823
Hantak MM, Paluh DJ, Saporito RA (2016) Bufadienolide and alkaloid-based chemical defences in two different species of neotropical anurans are equally effective against the same arthropod predators. J Trop Ecol 32:165–169. https://doi.org/10.1017/S0266467416000055
Hayes RA, Crossland MR, Hagman M, Capon RJ, Shine R (2009) Ontogenetic variation in the chemical defenses of cane toads (Bufo marinus): toxin profiles and effects on predators. J Chem Ecol 35:391–399. https://doi.org/10.1007/s10886-009-9608-6
Hovey KJ, Seiter EM, Johnson EE, Saporito RA (2018) Sequestered alkaloid defenses in the dendrobatid poison frog Oophaga pumilio provide variable protection from microbial pathogens. J Chem Ecol 44:312–325. https://doi.org/10.1007/s10886-018-0930-8
Hutchinson DA, Mori A, Savitzky AH, Burghardt GM, Wu X, Meinwald J, Schroeder FC (2007) Dietary sequestration of defensive steroids in nuchal glands of the Asian snake Rhabdophis tigrinus. Proc Natl Acad Sci 104:2265–2270. https://doi.org/10.1073/pnas.0610785104
Hutchinson DA, Savitzky AH, Mori A, Meinwald J, Schroeder FC (2008) Maternal provisioning of sequestered defensive steroids by the Asian snake Rhabdophis tigrinus. Chemoecology 18:181–190. https://doi.org/10.1007/s00049-008-0404-5
Hutchinson DA, Savitzky AH, Burghardt GM, Nguyen C, Meinwald J, Schroeder FC, Mori A (2013) Chemical defense of an Asian snake reflects local availability of toxic prey and hatchling diet: variation in chemical defense of Rhabdophis tigrinus. J Zool 289:270–278. https://doi.org/10.1111/jzo.12004
Inoue T, Nakata R, Savitzky AH, Yoshinaga N, Mori A, Mori N (2021) New insights into dietary toxin metabolism: diversity in the ability of the natricine snake Rhabdophis tigrinus to convert toad-derived bufadienolides. J Chem Ecol 47:915–925. https://doi.org/10.1007/s10886-021-01287-6
Jeckel AM, Saporito RA, Grant T (2015) The relationship between poison frog chemical defenses and age, body size, and sex. Front Zool 12:27. https://doi.org/10.1186/s12983-015-0120-2
Jeckel AM, Kocheff S, Saporito RA, Grant T (2019) Geographically separated orange and blue populations of the Amazonian poison frog Adelphobates galactonotus (Anura, Dendrobatidae) do not differ in alkaloid composition or palatability. Chemoecology 29:225–234. https://doi.org/10.1007/s00049-019-00291-3
Jeckel AM, Bolton SK, Waters KR, Antoniazzi MM, Jared C, Matsumura K, Nishikawa K, Morimoto Y, Grant T, Saporito RA (2022) Dose-dependent alkaloid sequestration and N-methylation of decahydroquinoline in poison frogs. J Exp Zool 2022:1–10. https://doi.org/10.1002/jez.2587
Jovanovic O, Vences M, Safarek G, Rabemananjara FCE, Dolch R (2009) Predation upon Mantella aurantiaca in the Torotorofotsy wetlands, central-eastern Madagascar. Herpetology Notes 2:95–97
Lachaud J (1990) Foraging activity and diet in some neotropical ponerine ants. Ectatomma ruidum roger (Hymenoptera, Formicidae). Folia Entomológica Mexicana 78:241–256
Lawrence JP, Rojas B, Fouquet A, Mappes J, Blanchette A, Saporito RA, Bosque RJ, Courtois EA, Noonan B (2019) Weak warning signals can exist in the absence of gene flow. Proc Natl Acad Sci 116:19037–19045. https://doi.org/10.1073/pnas.1901872116
Lenger DR, Berkey JK, Dugas MB (2014) Predation on the toxic Oophaga pumilio (Anura: Dendrobatidae) by Rhadinaea decorata (Squamata: Collubridae). Herpetology Notes 7:83–84
Malcolm SB, Brower LP (1989) Evolutionary and ecological implications of cardenolide sequestration in the monarch butterfly. Experientia 45:284–295. https://doi.org/10.1007/BF01951814
Malcolm S, Rothschild M (1983) A danaid Mullerian mimic, Euploea core amymone (Cramer) lacking cardenolides in the pupal and adult stages. Biol J Lin Soc 19:27–33. https://doi.org/10.1111/j.1095-8312.1983.tb00774.x
Maple M (2002) Maternal effects on offspring fitness in Dendrobates Pumilio, the strawberry poison frog. In: Din M (ed) PhD Dissertation, University of Kentucky, Lexington. Kentucky, USA
McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS (1994) La Selva: ecology and natural history of a neotropical rainforest, 1st edn. The University of Chicago Press, Chicago and London
Mebs D (2001) Toxicity in animals. Trends in evolution? Toxicon 39:87–96. https://doi.org/10.1016/S0041-0101(00)00155-0
Mina AE, Ponti AK, Woodcraft NL, Johnson EE, Saporito RA (2015) Variation in alkaloid-based microbial defenses of the dendrobatid poison frog Oophaga pumilio. Chemoecology 25:169–178. https://doi.org/10.1007/s00049-015-0186-5
Moranz R, Brower LP (1998) Geographic and temporal variation of cardenolide-based chemical defenses of queen butterfly (Danaus gilippus) in northern Florida. J Chem Ecol 24:905–932. https://doi.org/10.1023/A:1022329702632
Murray EM, Bolton SK, Berg T, Saporito RA (2016) Arthropod predation in a dendrobatid poison frog: does frog life stage matter? Zoology 119:169–174. https://doi.org/10.1016/j.zool.2016.01.002
Nishida R (2002) Sequestration of defensive substances from plants by Lepidoptera. Annu Rev Entomol 47:57–92. https://doi.org/10.1146/annurev.ento.47.091201.145121
Nishida R, Fukami H (1989) Ecological adaptation of an Aristolochiaceae-feeding swallowtail butterfly, Atrophaneura alcinous, to aristolochic acids. J Chem Ecol 15:2549–2563. https://doi.org/10.1007/BF01014731
Opitz SEW, Müller C (2009) Plant chemistry and insect sequestration. Chemoecology 19:117–154. https://doi.org/10.1007/s00049-009-0018-6
Pasteels JM, Dobler S, Rowell-Rahier M, Ehmke A, Hartmann T (1995) Distribution of autogenous and host-derived chemical defenses in Oreina leaf beetles (Coleoptera: Chrysomelidae). J Chem Ecol 21:1163–1179. https://doi.org/10.1007/BF02228318
Pavelka LA, Kim YH, Mosher HS (1977) Tetrodotoxin and tetrodotoxin-like compounds from the eggs of the Costa Rican frog Atelopus chiriquiensis. Toxicon 15:135–139. https://doi.org/10.1016/0041-0101(77)90032-0
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Santos JC, Cannatella DC (2011) Phenotypic integration emerges from aposematism and scale in poison frogs. Proc Natl Acad Sci 108:6175–6180. https://doi.org/10.1073/pnas.1010952108
Saporito RA, Garraffo HM, Donnelly MA, Edwards AL, Longino JT, Daly JW (2004) Formicine ants: an arthropod source for the pumiliotoxin alkaloids of dendrobatid poison frogs. Proc Natl Acad Sci 101:8045–8050. https://doi.org/10.1073/pnas.0402365101
Saporito RA, Donnelly MA, Jain P, Garraffo HM, Spande TF, Daly JW (2007a) Spatial and temporal patterns of alkaloid variation in the poison frog Oophaga pumilio in Costa Rica and Panama over 30 years. Toxicon 50:757–778. https://doi.org/10.1016/j.toxicon.2007.06.022
Saporito RA, Donnelly MA, Norton RA, Garraffo HM, Spande TF, Daly JW (2007b) Oribatid mites as a major dietary source for alkaloids in poison frogs. Proc Natl Acad Sci 104:8885–8890. https://doi.org/10.1073/pnas.0702851104
Saporito RA, Zuercher R, Roberts M, Gerrow KG, Donnelly MA (2007c) Experimental evidence for aposematism in the poison frog Oophaga pumilio. Copeia 4:1006–1011
Saporito RA, Spande TF, Garraffo HM, Donnelly MA (2009) Arthropod alkaloids in poison frogs: a review of the ‘dietary hypothesis.’ Heterocycles 79:277–297
Saporito RA, Donnelly MA, Madden AA, Garraffo HM, Spande TF (2010) Sex-related differences in alkaloid chemical defenses of the dendrobatid frog Oophaga pumilio from Cayo Nancy, Bocas Del Toro, Panama. J Nat Prod 73:317–321. https://doi.org/10.1021/np900702d
Saporito RA, Donnelly MA, Spande TF, Garraffo HM (2012) A review of chemical ecology in poison frogs. Chemoecology 22:159–168. https://doi.org/10.1007/s00049-011-0088-0
Saporito RA, Norton RA, Garraffo HM, Spande TF (2015) Taxonomic distribution of defensive alkaloids in Nearctic oribatid mites (Acari, Oribatida). Exp Appl Acarol 67:317–333. https://doi.org/10.1007/s10493-015-9962-8
Saporito RA, Russell MW, Richards-Zawacki CL, Dugas MB (2019) Experimental evidence for maternal provisioning of alkaloid defenses in a dendrobatid frog. Toxicon 161:40–43. https://doi.org/10.1016/j.toxicon.2019.02.008
Savitzky AH, Mori A, Hutchinson DA, Saporito RA, Burghardt GM, Lillywhite HB, Meinwald J (2012) Sequestered defensive toxins in tetrapod vertebrates: principles, patterns, and prospects for future studies. Chemoecology 22:141–158. https://doi.org/10.1007/s00049-012-0112-z
Sellmeigher B, van den Burg MP (2020) Tadpole predation in the chemically defended Oophaga pumilio (Anura: Dendrobatidae) by Oxybelis aeneus (Squamata: Colubridae). Herpetol Not 13:301–303
Solano M, Vega A, Saporito RA (2017) Phyllobates lugubris (Lovely poison frog) Predation by Coniophanes fissidens (Yellowbelly snake). Herpetol Review. 48:831
Speed MP, Ruxton GD, Mappes J, Sherratt TN (2012) Why are defensive toxins so variable? An evolutionary perspective. Biol Rev 87:874–884. https://doi.org/10.1111/j.1469-185X.2012.00228.x
Stuckert AMM, Summers K (2022) Investigating signal modalities of aposematism in a poison frog. J Evol Biol 2022:1–7. https://doi.org/10.1111/jeb.14111
Stynoski JL (2009) Discrimination of offspring by indirect recognition in an egg-feeding dendrobatid frog, Oophaga pumilio. Anim Behav 78:1351–1356. https://doi.org/10.1016/j.anbehav.2009.09.002
Stynoski JL, O’Connell LA (2017) Developmental morphology of granular skin glands in pre-metamorphic egg-eating poison frogs. Zoomorphology 136:219–224. https://doi.org/10.1007/s00435-017-0344-0
Stynoski JL, Porras-Brenes K (2022) Meta-analysis of tadpole taste tests: consumption of anuran prey across development and predator strategies. Oecologia 199:845–857. https://doi.org/10.1007/s00442-022-05221-9
Stynoski JL, Torres-Mendoza Y, Sasa-Marin M, Saporito RA (2014a) Evidence of maternal provisioning of alkaloid-based chemical defenses in the strawberry poison frog Oophaga pumilio. Ecology 95:587–593
Stynoski JL, Shelton G, Stynoski P (2014b) Maternally derived chemical defenses are an effective deterrent against some predators of poison frog tadpoles (Oophaga pumilio). Biol Let 10:20140187
Triponez Y, Naisbit RE, Jean-Denis JB, Rahier M, Alvarez N (2007) Genetic and environmental sources of variation in the autogenous chemical defense of a leaf beetle. J Chem Ecol 33:2011–2024. https://doi.org/10.1007/s10886-007-9351-9
Villanueva E, Brooks OL, Bolton SK, Savastano N, Schulte L, Saporito RA (2022) Maternal provisioning of alkaloid defenses in dendrobatid poison frogs. J Chem Ecol 23:1–10
Weldon PJ, Kramer M, Gordon S, Spande TF, Daly JW (2006) A common pumiliotoxin from poison frogs exhibits enantioselective toxicity against mosquitoes. Proc Natl Acad Sci 103:17818–17821. https://doi.org/10.1073/pnas.0608646103
Whitfield SM, Bell KE, Philippi T, Sasa M, Bolanos F, Chaves G, Savage JM, Donnelly MA (2007) Amphibian and reptile declines over 35 years at la selva, costa rica. Proc Natl Acad Sci 104:8352–8356. https://doi.org/10.1073/pnas.0611256104
Williams BL, Hanifin CT, Brodie ED, Caldwell RL (2011) Ontogeny of tetrodotoxin levels in blue-ringed octopuses: maternal investment and apparent independent production in offspring of Hapalochlaena lunulata. J Chem Ecol 37:10–17
Acknowledgements
We thank the John Carroll University Tropical Biology class of 2019 for their help in deploying tadpole-rearing cups, especially A. Jones, M. Hatlovic, and K. Waters, as well as O. Medina-Baez, A. Perrino, J. Ryan, L. Phillip, and C. Thomas. We thank M.B. Dugas for assistance with statistical analyses. We thank M. Nichols for assistance in maintaining the GC–MS and J. Your for help with building tadpole cups. We thank the La Selva Research Station and Costa Rican government for allowing us to conduct this research, and Enrique Alonso Castro Fonseca and Orlando Vargas Ramírez for logistical support.
Funding
Funding support was provided by John Carroll University.
Author information
Authors and Affiliations
Contributions
OLB and RAS conceived of the project and designed the experiments. OLB, RAS, and JJJ contributed to data collection. OLB and RAS analyzed the data. OLB and RAS wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed. Research protocols were approved by the John Carroll University Institutional Animal Care and Use Committee (IACUC approval 1700). Costa Rican research permits “SINAC-ACC-PI-R-068–2019” and “R-037–2019-OT-CONAGEBIO" were granted by the Sistema Nacional de Áreas de Conservacion (SINAC) and the Comision Nacional para la Gestion de la Biodiversidad (CONAGEBIO), Ministerio de Ambiente y Energía, respectively. The Convention on International Trade in Endangered Species of Wild Flora and Flora (CITES) export permits “2019-CR4841/SJ#5863” and “2019-CR4636/SJ#5650” were granted by the Sistema Nacional de Áreas de Conservacion (SINAC) of the Costa Rican government.
Additional information
Communicated by Donald Miles.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brooks, O.L., James, J.J. & Saporito, R.A. Maternal chemical defenses predict offspring defenses in a dendrobatid poison frog. Oecologia 201, 385–396 (2023). https://doi.org/10.1007/s00442-023-05314-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-023-05314-z