Abstract
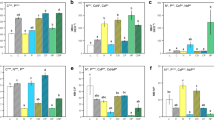
A dominant paradigm in ecology is that plants are limited by nitrogen (N) during primary succession. Whether generalizable patterns of nutrient limitation are also applicable to metabolically and phylogenetically diverse soil microbial communities, however, is not well understood. We investigated if measures of N and phosphorus (P) pools inform our understanding of the nutrient(s) most limiting to soil microbial community activities during primary succession. We evaluated soil biogeochemical properties and microbial processes using two complementary methodological approaches—a nutrient addition microcosm experiment and extracellular enzyme assays—to assess microbial nutrient limitation across three actively retreating glacial chronosequences. Microbial respiratory responses in the microcosm experiment provided evidence for N, P and N/P co-limitation at Easton Glacier, Washington, USA, Puca Glacier, Peru, and Mendenhall Glacier, Alaska, USA, respectively, and patterns of nutrient limitation generally reflected site-level differences in soil nutrient availability. The activities of three key extracellular enzymes known to vary with soil N and P availability developed in broadly similar ways among sites, increasing with succession and consistently correlating with changes in soil total N pools. Together, our findings demonstrate that during the earliest stages of soil development, microbial nutrient limitation and activity generally reflect soil nutrient supply, a result that is broadly consistent with biogeochemical theory.




Similar content being viewed by others
References
Alexander EB, Burt R (1996) Soil development on moraines of Mendenhall Glacier, southeast Alaska. 1. The moraines and soil morphology. Geoderma 72:1–17
Allison SD (2006) Soil minerals and humic acids alter enzyme stability: implications for ecosystem processes. Biogeochemistry 81:361–373
Allison VJ, Condron LM, Peltzer DA, Richardson SJ, Turner BL (2007) Changes in enzyme activities and soil microbial community composition along carbon and nutrient gradients at the Franz Josef chronosequence, New Zealand. Soil Biol Biochem 39:1770–1781
Anesio AM, Hodson AJ, Fritz A, Psenner R, Sattler B (2008) High microbial activity on glaciers: importance to the global carbon cycle. Glob Change Biol 15:955–960
Bardgett RD, Richter A, Bol R, Garnett MH, Bäumler R, Xu X, Lopez-Capel E, Manning DA, Hobbs PJ, Hartley IR, Wanek W (2007) Heterotrophic microbial communities use ancient carbon following glacial retreat. Biol Lett 3:487–490
Bååth E (2001) Estimation of fungal growth rates in soil using 14C-acetate incorporation into ergosterol. Soil Biol Biochem 33:2011–2018
Bradford MA, Fierer N, Reynolds JF (2008) Soil carbon stocks in experimental mesocosms are dependent on the rate of labile carbon, nitrogen and phosphorus inputs to soils. Funct Ecol 22:964–974
Brankatschk R, Toewe S, Kleineidam K, Schloter M, Zeyer J (2011) Abundances and potential activities of nitrogen cycling microbial communities along a chronosequence of a glacier forefield. ISME J 5:1025–1037
Castle SC, Neff JC (2009) Plant response to nutrient availability across bedrock geologies. Ecosystems 12:101–113
Castle SC, Lekberg Y, Affleck D, Cleveland CC (2016a) Soil abiotic and biotic controls on plant performance during primary succession in a glacial landscape. J Ecol 104:1555–1565
Castle SC, Nemergut DR, Grandy AS, Leff JW, Graham EB, Hood E, Schmidt SK, Wickings K, Cleveland CC (2016b) Biogeochemical drivers of microbial community convergence across actively retreating glaciers. Soil Biol Biochem 101:74–84
Chapin FS, Walker LR, Fastie CL, Sharman LC (1994) Mechanisms of primary succession following deglaciation at Glacier Bay, Alaska. Ecol Monogr 64:149–175
Cleveland CC, Townsend AR (2006) Nutrient additions to a tropical rain forest drive substantial soil carbon dioxide losses to the atmosphere. Proc Nat Acad Sci 103:10316–10321
Cleveland C, Liptzin D (2007) C:N: P stoichiometry in soil: is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 85:235–252
Cleveland CC, Reed SC, Townsend AR (2006) Nutrient regulation of organic matter decomposition in a tropical rain forest. Ecology 87:492–503
Cline LC, Zak DR (2015) Soil microbial communities are shaped by plant-driven changes in resource availability during secondary succession. Ecology 96:3374–3385
Craine JM, Morrow C, Fierer M (2007) Microbial nitrogen limitation increases decomposition. Ecology 88:2105–2113
Crews TE, Kitayama K, Fownes JH, Riley RH, Herbert DA, Mueller-Dombois D, Vitousek PM (1995) Changes in soil-phosphorus fractions and ecosystem dynamics across a long chronosequence in Hawaii. J Ecol 76:1407–1424
Darcy JL, Schmidt SK (2016) Nutrient limitation of microbial phototrophs on a debris-covered glacier. Soil Biol Biochem 95:156–163
Dick WA, Tabatabai MA (1987) Kinetics and activities of phosphatase-clay complexes. Soil Sci 143:5–15
Doane T, Horwath W (2003) Spectrophotometric determination of nitrate with a single reagent. Anal Lett 36:2713–2722
Duc L, Noll M, Meier BE, Burgmann H, Zeyer J (2009) High diversity of diazotrophs in the forefield of a receding alpine glacier. Microb Ecol 57:179–190
Elser JJ, Bracken ME, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142
Eviner VT, Chapin FS III, Vaughn CE (2000) Nutrient manipulations in terrestrial ecosystems. In: Sala OE, Jackson RB, Mooney HA, Howarth RW (eds) Methods in ecosystem science. Springer, New York, pp 291–307
Fernández-Martínez MA, Pérez-Ortega S, Pointing SB, Green TA, Pintado A, Rozzi R, Sancho LG, de los Ríos A (2017) Microbial succession dynamics along glacier forefield chronosequences in Tierra del Fuego (Chile). Polar Biol 40:1939–1957
Fierer N, Nemergut DR, Knight R, Craine JM (2010) Changes through time: integrating microorganisms into the study of succession. Res Microbiol 161:635–642
Frossard E, Condron LM, Oberson A, Sinaj S, Fardeau JC (2000) Processes governing phosphorus availability in temperate soils. J Environ Qual 29:15–23
Gallo ME, Amonette R, Lauber C, Sinsabaugh RL, Zak DR (2004) Microbial community structure and oxidative enzyme activity in Nitrogen-amended north temperate forest soils. Microb Ecol 48:218–229
German DP, Weintraub MN, Grandy AS, Lauber CL, Rinkes ZL, Allison SD (2011) Optimization of hydrolytic and oxidative enzyme methods for ecosystem studies. Soil Biol Biochem 43:1387–1397
Göransson H, Venterink HO, Bååth E (2011) Soil bacterial growth and nutrient limitation along a chronosequence from a glacier forefield. Soil Biol Biochem 43:1333–1340
Halvorson JJ, Franz EH, Smith JL, Black RA (1992) Nitrogenase activity, nitrogen fixation, and nitrogen inputs by lupines at Mount St Helens. Ecology 73:87–98
Harpole WS, Ngai JT, Cleland EE, Seabloom EW, Borer ET, Bracken ME, Elser JJ, Gruner DS, Hillebrand H, Shurin JB, Smith JE (2011) Nutrient co-limitation of primary producer communities. Ecol Lett 14:852–862
Hobbie SE, Vitousek PM (2000) Nutrient limitation of decomposition in Hawaiian forests. Ecology 81:1867–1877
Horwath W, Paul E (1994) Microbial biomass. In: Weaver RW, Angle JS, Bottomley PS (eds) Methods of soil analysis, part 2: microbiological and biochemical properties. Soil Science Society of America, Fitchburg, pp 754–760
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Jeannotte R, Sommerville DW, Hamel C, Whalen JK (2004) A microplate assay to measure soil microbial biomass phosphorus. Biol Fertil Soils 40:201–205
King AJ, Meyer AF, Schmidt SK (2008) High levels of microbial biomass and activity in unvegetated tropical and temperate alpine soils. Soil Biol Biochem 40:2605–2610
Knelman JE, Legg TM, O’Neill SP, Washenberger CL, González A, Cleveland CC, Nemergut DR (2012) Bacterial community structure and function change in association with colonizer plants during early primary succession in a glacier forefield. Soil Biol Biogeochem 46:172–180
Knelman JE, Schmidt SK, Lynch RC, Darcy JL, Castle SC, Cleveland CC, Nemergut DR (2014) Nutrient addition dramatically accelerates microbial community succession. PloS One 9: e102609. doi:10.1371/journal.pone.0102609
Laliberté E, Turner BL, Costes T, Pearse SJ, Wyrwoll K-H, Zemunik G, Lambers H (2012) Experimental assessment of nutrient limitation along a 2-million year dune chronosequence in the south-western Australia biodiversity hotspot. J Ecol 100:631–642
Lebauer DS, Treseder KK (2008) Nitrogen limitation of net primary productivity in terrestrial ecosystems is globally distributed. Ecology 89:371–379
Leff JW, Jones SE, Prober SM, Barberán A, Borer ET, Firn JL, Harpole WS, Hobbie SE, Hofmockel KS, Knops JM, McCulley RL (2015) Consistent responses of soil microbial communities to elevated nutrient inputs in grasslands across the globe. Proc Natl Acad Sci 112(35):10967–10972
Ley R, Williams M, Schmidt S (2004) Microbial population dynamics in an extreme environment: controlling factors in talus soils at 3750 m in the Colorado Rocky Mountains. Biogeochemistry 68:313–335
Matthews JA (1992) The ecology of recently-deglaciated terrain: a geoecological approach to glacier forelands and primary succession. Cambridge University Press, Cambridge
McGill WB, Cole CV (1981) Comparative aspects of cycling of organic C, N, S and P through soil organic matter. Geoderma 26:267–286
Menge DNL, Hedin LO (2009) Nitrogen fixation in different biogeochemical niches along a 120,000-year chronosequence in New Zealand. Ecology 90:2190–2201
Mulvaney RL (1994) Nitrogen—inorganic forms. In: Sparks DL (ed) Methods of soil analysis, part 3: chemical methods. Soil Science Society of America, Fitchburg, pp 1129–1131
Nemergut, DR (2004) Evolution and ecology of high altitude soil microbial communities. Ph.D. dissertation, University of Colorado, Boulder, CO
Nemergut DR, Anderson SP, Cleveland CC, Martin AP, Miller AE, Seimon A, Schmidt SK (2007) Microbial community succession in an unvegetated recently deglaciated soil. Microb Ecol 53:110–122
Neter J, Kutner MH, Nachtsheim CJ, Wasserman W (1996) Applied linear statistical models, 4th edn. Irwin McGraw-Hill, Chicago
Ohtonen R, Fritze H, Pennanen T, Jumpponen A, Trappe J (1999) Ecosystem properties and microbial community changes in primary succession on a glacier forefront. Oecologia 119:239–246
Olander LP, Vitousek PM (2000) Regulation of soil phosphatase and chitinase activity by N and P availability. Biogeochemistry 49:175–191
Parfitt RL, Ross DJ, Coomes DA, Richardson SJ, Smale MC, Dahlgren RA (2005) N and P in New Zealand soil chronosequences and relationships with foliar N and P. Biogeochemistry 75:305–328
Peltzer D, Wardle D, Allison V, Baisden W (2010) Understanding ecosystem retrogression. Ecol Monogr 80:509–529
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Ramirez KS, Craine JM, Fierer N (2012) Consistent effects of nitrogen amendments on soil microbial communities and processes across biomes. Glob Change Biol 18:1918–1927
Reed SC, Vitousek PM, Cleveland CC (2011) Are patterns in nutrient limitation belowground consistent with those aboveground: results from a 4 million year chronosequence. Biogeochemistry 106:323–336
Reynolds H, Packer A, Bever J, Clay K (2003) Grassroots ecology: plant-microbe-soil interactions as drivers of plant community structure and dynamics. Ecology 84:2281–2291
Richardson SJ, Peltzer DA, Allen RB, McGlone MS, Parfitt RL (2004) Rapid development of phosphorus limitation in termperate rainforest along the Franz Josef chronosequence. Oecologia 139:267–276
Saiya-Cork K, Sinsabaugh R, Zak D (2002) The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol Biochem 34:1309–1315
Sattin S, Cleveland C, Hood E, Reed S (2009) Functional shifts in unvegetated, perhumid, recently-deglaciated soils do not correlate with shifts in soil bacterial community composition. J Microbiol 47:673–681
Schimel JP, Schaffer SM (2012) Microbial control over carbon cycling in soil. Front Microbiol 3:1–11
Schmidt SK, Reed SC, Nemergut DR, Grandy AS, Cleveland CC, Weintraub MN, Hill AW, Costello EK, Meyer AF, Neff JC, Martin AM (2008) The earliest stages of ecosystem succession in high-elevation (5000 metres above sea level), recently deglaciated soils. Proc R Soc B Biol Sci 275:2793–2802
Schmidt SK, Nemergut DR, Sowell P, Reed SC, Cleveland CC (2011) Estimating phosphorus availability for microbial growth in an emerging landscape. Geoderma 163:135–140
Schmidt SK, Nemergut DR, Todd BT, Lynch RC, Darcy JL, Cleveland CC, King AJ (2012) A simple method for determining limiting nutrients for photosynthetic crusts. Plant Ecol Divers. doi:10.1080/175508742012738714
Selmants P, Hart S (2010) Phosphorus and soil development: does the Walker and Syers model apply to semiarid ecosystems? Ecology 91:474–484
Sigler WV, Zeyer J (2004) Colony-forming analysis of bacterial community succession in deglaciated soils indicates pioneer stress-tolerant opportunists. Microb Ecol 48:316–323
Sinsabaugh RL (1994) Enzymic analysis of microbial patterns and process. Biol Fertil Soils 17:69–74
Sinsabaugh RL, Follstad Shah JJ (2012) Ecoenzymatic stoichiometry and ecological theory. Annu Rev Ecol Evol Syst 43:313–343
Sinsabaugh RL, Antibus RK, Linkins AE, Rayburn L, Repert D, Weiland T (1993) Wood decomposition: nitrogen and phosphorus dynamics in relation to extracellular enzyme activity. Ecology 74:1586–1593
Sinsabaugh RL, Carreiro MM, Repert DA (2002) Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochemistry 60:1–24
Sinsabaugh RL, Lauber CL, Weintraub MN, Ahmed B, Allison SD, Crenshaw C, Contosta AR, Cusack D, Frey S, Gallo ME, Gartner TB (2008) Stoichiometry of soil enzyme activity at global scale. Ecol Lett 11:1252–1264
Sinsabaugh RL, Hill BH, Follstad Shah JJ (2009) Ecoenzymatic stoichiometry of microbial organic nutrient acquisition in soil and sediment. Nature 462:795–798
Stursova M, Crenshaw CL, Sinsabaugh RL (2006) Microbial responses to long-term N deposition in a semiarid grassland. Microb Ecol 51:90–98
Sullivan BW, Alvarez-Clare S, Castle SC, Porder S, Reed SC, Schreeg L, Townsend AR, Cleveland CC (2014) Assessing nutrient limitation in complex forested ecosystems: alternatives to large-scale fertilization experiments. Ecology 95:668–681
Tabor RW, Haugerud RA, Hildreth W, Brown EH (2003) Geologic map of the Mount Baker 30 × 60 minute quadrangle. Washington US Geological Survey Map I-2660, scale 1:100,000
Tscherko D, Rustemeier J, Richter A, Wanek W, Kanedler E (2003) Functional diversity of the soil microflora in primary succession across two glacier forelands in the Central Alps. Eur J Soil Sci 54:685–696
Turner BL, Laliberté E (2014) Soil development and nutrient availability along a 2 million-year coastal dune chronosequence under species-rich mediterranean shrubland in southwestern Australia. Ecosystems 100:631–642
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Vitousek PM (2004) Nutrient cycling and limitation: Hawai’i as a model system. Princeton University Press, Princeton
Vitousek P, Farrington H (1997) Nutrient limitation and soil development: experimental test of a biogeochemical theory. Biogeochemistry 37:63–75
Vitousek PM, Walker LR, Whiteaker LD, Matson PA (1993) Nutrient limitations to plant growth during primary succession in Hawaii Volcanoes National Park. Biogeochemistry 23:197–215
Waldrop MP, Balser TC, Firestone MK (2000) Linking microbial community composition to function in a tropical soil. Soil Biol Biochem 32:1837–1846
Walker L, del Moral R (2003) Primary succession and ecosystem rehabilitation. Cambridge University Press, New York
Walker T, Syers J (1976) Fate of phosphorus during pedogenesis. Geoderma 15:1–19
Wardle D, Bardgett R, Klironomos J, Setälä H, van der Putten W, Wall D (2004) Ecological linkages between aboveground and belowground biota. Science 304:1629–1633
Weatherburn MW (1967) Phenol-hypochlorite reaction for determination of ammonia. Anal Chem 39:971–974
Weintraub M, Scott-Denton L, Schmidt S, Monson R (2007) The effects of tree rhizodeposition on soil exoenzyme activity, dissolved organic carbon, and nutrient availability in a subalpine forest ecosystem. Oecologia 154:327–338
Yoshitake S, Uchida M, Koizumi H, Nakatsubo T (2007) Carbon and nitrogen limitation of soil microbial respiration in a High Arctic successional glacier foreland near Ny-Ålesund, Svalbard. Polar Res 26:22–30
Acknowledgements
We would like to thank R. Callaway, S. Dobrowski, A. Larson, Y. Lekberg, A. Marklein, M. Nasto, and three anonymous reviewers for comments on early drafts of this manuscript. Authors declare no conflict of interest. This work was supported by a National Science Foundation Grant (NSF DEB-0922306) made to CC, EH, DN, and SS.
Author information
Authors and Affiliations
Contributions
SCC and CCC conceived and designed the experiments. SCC performed the experiments and analyzed the data. SCC, BWS, JK, and CCC wrote the manuscript; other authors provided editorial advice and all authors approved of the final version of the manuscript.
Corresponding author
Additional information
Communicated by Jason P. Kaye.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Castle, S.C., Sullivan, B.W., Knelman, J. et al. Nutrient limitation of soil microbial activity during the earliest stages of ecosystem development. Oecologia 185, 513–524 (2017). https://doi.org/10.1007/s00442-017-3965-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-017-3965-6