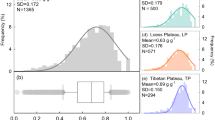
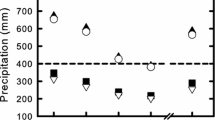
Abstract
The ability to appropriately modify physiological and morphological traits in response to temporal variation should increase fitness. We used recombinant hybrid plants generated by crossing taxa in the Piriqueta caroliniana complex to assess the effects of individual leaf traits and trait plasticities on growth in a temporally variable environment. Recombinant hybrids were used to provide a wide range of trait expression and to allow an assessment of the independent effects of individual traits across a range of genetic backgrounds. Hybrid genotypes were replicated through vegetative propagation and planted in common gardens at Archbold Biological Station in Venus, Florida, where they were monitored for growth, leaf morphological characters, and integrated water use efficiency (WUE) (C isotope ratio; δ13C) for two successive seasons. Under wet conditions only leaf area had significant effects on plant growth, but as conditions became drier, growth rates were greatest in plants with narrow leaves and higher trichome densities. Plants with higher WUE exhibited increased growth during the dry season but not during the wet season. WUE during the dry season was increased for plants with smaller, narrower leaves that had higher trichome densities and increased reflectance. Examination of alternative path models revealed that during the dry season leaf traits had significant effects on plant growth only through their direct effects on WUE, as estimated from δ13C. Over the entire growing season, plants with a greater ability to produce smaller and narrower leaves with higher trichome densities in response to reduced water availability had the greatest growth rate. These findings suggest that plants making appropriate changes to leaf morphology as conditions became dry had increased WUE, and that the ability to adjust leaf phenotypes in response to environmental variation is a mechanism by which plants increase fitness.


Similar content being viewed by others
References
Ackerly DD, et al. (2000) The evolution of plant ecophysiological traits: recent advances and future directions. Bioscience 50:979–995
Alpert P, Simms EL (2002) The relative advantages of plasticity and fixity in different environments: when is it good for a plant to adjust? Evol Ecol 16:285–297
Angilletta MJJ, Oufiero CE, Leache AD (2006) Direct and indirect effects of environmental temperature on the evolution of reproductive strategies: an information-theoretic approach. Am Nat 168:E123–E135
Arbuckle JL (2005) AMOS SPSS, 6.0 edn. Amos Development, Spring House, Pa.
Benz BR, Rhode JM, Cruzan MB (2007) Root anatomical and physiological responses to flooding in the Piriqueta caroliniana complex. Am J Bot 94:542–550
Budyko MI (1974) Climate and life, 1st edn. Academic Press, New York
Burnham KP, Anderson DR (1998) Model selection and multimodal inference—a oractical information-theoretic approach, 2nd edn. Springer, New York
Caruso CM, Maherali H, Sherrard M (2006) Plasticity of physiology in Lobelia: testing for adaptation and constraint. Evolution 60:980–990
Chu Y, Yu FH, Dong M (2006) Clonal plasticity in response to reciprocal patchiness of light and nutrients in the stoloniferous herb Glechoma longituba L. J Integr Plant Biol 48:400–408
Close DC, Beadle CL (2003) The ecophysiology of foliar anthocyanin. Bot Rev 69:149–161
Close DC, Beadle CL, Brown PH, Holz GK (2000) Cold-induced photoinhibition affects establishment of Eucalyptus nitens (Deane and Maiden) Maiden and Eucalyptus globulus Labill. Trees 15:32–41
Cruzan MB (2005) Patterns of introgression across an expanding hybrid zone: analyzing historical patterns of gene flow using non-equilibrium approaches. New Phytol 167:267–278
Cruzan MB, Rhode JM (2004) Experimental analysis of adaptive landscape topographies. In: Cronk QCB, Whitton J, Ree RH, Taylor IEP (eds) Plant adaptation: molecular genetics and ecology. National Research Council of Canada, Vancouver, pp 61–69
de Kroon H, Huber H, Stuefer JF, van Groenendael JM (2005) A modular concept of phenotypic plasticity in plants. New Phytol 166:73–82
Donovan LA, Ehleringer JR (1992) Contrasting water-use patterns among size and life-history classes of a semiarid shrub. Funct Ecol 6:482–488
Donovan LA, Ehleringer JR (1994) Potential for selection on plants for water-use efficiency as estimated by carbon-isotope discrimination. Am J Bot 81:927–935
Dudley SA (1996) Differing selection on plant physiological traits in response to environmental water availability: a test of adaptive hypotheses. Evolution 50:92–102
Dudley SA (1996) The response to differing selection on plant physiological traits: evidence for local adaptation. Evolution 50:103–110
Ehleringer J (1976) Leaf pubescence: effects on absorptance and photosynthesis in a desert shrub. Science 192:376–377
Ehleringer J (1982) The influence of water stress and temperature on leaf pubescence development in Encelia farinosa. Am J Bot 69:670–675
Ehleringer J, Cooper TA (1988) Correlations between carbon isotope ratio and microhabitat in desert plants. Oecologia 76:562–566
Ehleringer J, Mooney HA (1978) Leaf pubescence: effects on absorptance and photosynthesis in a desert shrub. Oecologia 37:183–200
Ehleringer J, Werk KS (1986) Modifications of solar-radiation absorption patterns and implications for carbon gain at the leaf level. In: Givnish TJ (ed) On the economy of plant form and function. Cambridge University Press, Cambridge, pp 57–81
Farquhar GD, Ehleringer J, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 40:503–537
Farquhar GD, O’Leary MH, Berry JA (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust J Plant Physiol 9:121–137
Fischer RA, Turner NC (1978) Plant productivity in the arid and semiarid zones. Annu Rev Plant Physiol 29:277–317
Gates DM (1980) Biophysical ecology. Springer, New York
Gibson AC (1998) Photosynthetic organs of desert plants. Bioscience 48:911–920
Givnish TJ (1987) Comparative studies of leaf form: assessing the relative roles of selective pressures and phylogenetic constrains. New Phytol 106:131–160
Gomulkiewicz R, Kirkpatrick M (1992) Quantitative genetics and the evolution of reaction norms. Evolution 46:390–411
Gould KS (2004) Nature’s swiss army knife: the diverse protective roles of anthocyanins in leaves. J Biomed Biotechnol 5:314–320
Hamerlynck EP, Huxman TE, McAuliffe JR, Smith SD (2004) Carbon isotope discrimination and foliar nutrient status Larrea tridentata (creosote bush) in contrasting Mojave Desert soils. Oecologia 138:210–215
Harper JL (1977) Population biology of plants. Academic Press, London
Heschel MS, Donohue K, Hausmann N, Schmitt J (2002) Population differentiation and natural selection for water-use efficiency in Impatiens capensis (Balsaminaceae). Int J Plant Sci 163:907–912
Heschel MS, Riginos C (2005) Mechanisms of selection for drought stress tolerance and avoidance in Impatiens capensis (Balsaminacea). Am J Bot 92:37–44
Huber H, Lukacs S, Watson MA (1999) Spatial structure of stoloniferous herbs: an interplay between structural blue-print, ontogeny and phenotypic plasticity. Plant Ecol 141:107–115
Lambers H, Lambers H, Pons T (1998) Plant physiological ecology, 1st edn. Springer, New York
Lande R, Arnold S (1983) The measurement of selection on correlated characters. Evolution 37:1210–1226
Lee DW, Lowry JB (1980) Young-leaf anthocyanin and solar ultraviolet. Biotropica 12:75–76
Levins R (1968) Evolution in changing environments. Princeton University Press, Princeton, N.J.
Lewis WH, Stripling HL, Ross RG (1962) Chromosome numbers for some angiosperms of the souteastern United States and Mexico. Rhodora 64:147–161
Ludwig F et al (2004) Selection on leaf ecophysiological traits in a desert hybrid Helianthus species and early-generation hybrids. Evolution 58:2682–2692
Malmstrom VH (1969) A new approach to the classification of climate. J Geogr 68:351–357
Martin LJ, Cruzan MB (1999) Patterns of hybridization in the Piriqueta caroliniana complex in central Florida: evidence for an expanding hybrid zone. Evolution 53:1037–1049
Martinez-Garza C, Howe HF (2005) Developmental strategy or immediate responses in leaf traits of tropical tree species? Int J Plant Sci 166:41–48
Maskas SD, Cruzan MB (2000) Patterns of intraspecific diversification in the Piriqueta caroliniana complex in eastern North America and the Bahamas. Evolution 54:815–827
Mendez M, Jones DG, Manetas Y (1999) Enhanced UV-B radiation under field conditions increases anthocyanin and reduces the risk of photoinhibition but does not affect growth in the carnivorous plant **uicula vulgaris. New Phytol 144:275–282
Miner BG, Vonesh JR (2004) Effects of fine grain environmental variability on morphological plasticity. Ecol Lett 7:794–801
Nobel P (2005) Physiochemical and environmental Plant physiology, 3rd edn. Academic Press, San Diego, Calif.
Parkhurst DF, Loucks OJ (1972) Optimal leaf size in relation to the environment. J Ecol 60:505–537
Picotte J (2006) Leaf morphological responses to environmental change in the Piriqueta caroliniana complex. Department of Biology. Portland State University, Portland, Oreg.
Pigliucci M (2001) Phenotypic plasticity: beyond nature and nurture. John Hopkins University Press, London
Reboud X, Bell G (1997) Experimental evolution in Chlamydomonas. III. Evolution of a specialist and generalist types in environments that vary in space and time. Heredity 78:507–514
Rhode JM, Cruzan MB (2005) Contributions of heterosis and epistasis to hybrid fitness. Am Nat 166:E124–E139
Rosenthal DM, Ludwig F, Donovan LA (2005) Plant responses to an edaphic gradient across an active sand dune/desert boundary in the Great Basin Desert. Int J Plant Sci 166:247–255
SAS (2002) SAS/STAT user’s guide, version 6, 4th ed. SAS Institute, Cary, N.C.
Schlichting CD, Pigliucci M (1995) Gene-regulation, quantitative genetics and the evolution of reaction norms. Evol Ecol 9:154–168
Schuepp PH (1993) Tansley review no. 59 Leaf boundary-layers. New Phytol 125:477–507
Schwinning S, Sala OE, Loik ME, Ehleringer JR (2004) Thresholds, memory, and seasonality: understanding pulse dynamics in arid/semi-arid ecosystems. Oecologia 141:191–193
Sims DA, Gamon JA (2002) Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens Environ 81:337–354
Smith WK, Nobel PS (1977) Influences of seasonal changes in leaf morphology on water-use efficiency for three desert broadleaf shrubs. Ecology 58:1033–1043
Sultan SE (1987) Evolutionary implications of phenotypic plasticity in plants. Evol Biol 21:127–178
Sultan SE (2005) An emerging focus on plant ecological development. New Phytol 166:1–5
Thornthwaite CW (1948) An approach to toward a rational classification of climate. Geogr Rev 38:55–94
van Kleunen M, Fischer M (2005) Constraints on the evolution of adaptive phenotypic plasticity in plants. New Phytol 166:49–60
Via S, Lande R (1985) Genotype-environment interaction and the evolution of phenotypic plasticity. Evolution 39:505–522
Watson MA, Casper BB (1984) Morphogenic constraints on patterns of carbon distribution in plants. Annu Rev Ecol Syst 15:233–258
White J (1979) The plant as a metapopulation. Annu Rev Ecol Syst 10:109–145
Winn AA (1996a) Adaptation to fine-grained environmental variation: an analysis of within-individual leaf variation in an annual plant. Evolution 50:1111–1118
Winn AA (1996b) The contributions of programmed developmental change and phenotypic plasticity to within-individual variation in leaf traits in Dicerandra linearifolia. J Evol Biol 9:737–752
Winn AA (1999) Is seasonal variation in leaf traits adaptive for the annual plant Dicerandra linearifolia? J Evol Biol 12:306–313
Acknowledgements
We would like to thank K. Anton, B. Benz, H. de Glanville, A. Henderson, and A. Ramakrishnan for assistance with field and lab work. Earlier versions of this manuscript were improved by comments from H. Chang, S. Eppley, and two anonymous reviewers. Thanks also to E. Menges, M. Rickey, C. Weekley, and other members of the Plant Lab at Archbold Biological Station. This project was funded by Sigma ** and Lea-Forbes Fund (PSU) grants to J. J. P., and by NSF grant DEB-0080437 to M. B. C.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Manuel Lerdau.
Rights and permissions
About this article
Cite this article
Picotte, J.J., Rosenthal, D.M., Rhode, J.M. et al. Plastic responses to temporal variation in moisture availability: consequences for water use efficiency and plant performance. Oecologia 153, 821–832 (2007). https://doi.org/10.1007/s00442-007-0794-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0794-z