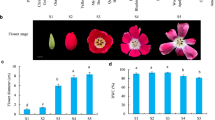
Abstract
Flowering, which plays a crucial role in the growth and development of flowering plants, is a crucial point from vegetative growth to reproductive growth. The goal of this study was to examine the differences between the transcriptomes of the Chinese cabbage mutant pdm and the corresponding wild-type line ‘FT’. We performed transcriptome analysis on mRNA isolated from flower buds of pdm and ‘FT’ using Illumina RNA sequencing (RNA-Seq) data. A total of 117 differentially expressed genes (DEGs) were detected. Among the DEGs, we identified a number of genes involved in floral development and flowering, including an F-box protein gene, EARLY FLOWERING 4 (ELF4), and transcription factors BIGPETAL (BPE) and MYB21 (v-myb avian myeloblastosis viral oncogene homolog); differential expression of these genes could potentially explain the difference in the flowers between pdm and ‘FT’. In addition, the expression patterns of 20 DEGs, including 12 floral development and flowering-related genes and eight randomly selected genes, were validated by qRT-PCR, and the results were highly concordant with the RNA-Seq results. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes pathway enrichment analyses were performed to better understand the functions of these DEGs. We also identified a large number of single nucleotide polymorphism and insertion/deletion markers, which will be a rich resource for future marker development and breeding research in Chinese cabbage. Also, our analysis revealed numerous novel transcripts and alternative splicing events. The transcriptome analysis provides valuable information for furthering our understanding of the molecular mechanisms that regulate the flowering process, and establishes a solid foundation for future genetic and functional genomic studies in Chinese cabbage.







Similar content being viewed by others
References
Aceto S, Gaudio L (2011) The mads and the beauty: genes involved in the development of orchid flowers. Curr Genomics 12:342–356
Alagna F, D’Agostino N, Torchia L, Servili M, Rao R, Pietrella M, Giuliano G, Chiusano ML, Baldoni L, Perrotta G (2009) Comparative 454 pyrosequencing of transcripts from two olive genotypes during fruit development. BMC Genomics 10:399
Amasino RM, Michaels SD (2010) The timing of flowering. Plant Physiol 154:516–520
Ansorge WJ (2009) Next-generation DNA sequencing techniques. New Biotechnol 25:195–203
Araki T (2001) Transition from vegetative to reproductive phase. Curr Opin Plant Biol 4:63–68
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet 25:25–29
Barakat A, DiLoreto DS, Zhang Y, Smith C, Baier K, Powell WA, Wheeler N, Sederoff R, Carlson JE (2009) Comparison of the transcriptomes of American chestnut (Castanea dentata) and Chinese chestnut (Castanea mollissima) in response to the chestnut blight infection. BMC Plant Biol 9:51
Blencowe BJ (2006) Alternative splicing: new insights from global analyses. Cell 126:37–47
Boss PK, Bastow RM, Mylne JS, Dean C (2004) Multiple pathways in the decision to flower: enabling, promoting, and resetting. Plant Cell 16:S18–S31
Bowman JL, Smyth DR, Meyerowitz EM (1989) Genes directing flower development in Arabidopsis. Plant Cell 1:37–52
Brioudes F, Joly C, Szécsi J, Varaud E, Leroux J, Bellvert F, Bertrand C, Bendahmane M (2009) Jasmonate controls late development stages of petal growth in Arabidopsis thaliana. Plant J 60:1070–1080
Browse J (2005) Jasmonate: an oxylipin signal with many roles in plants. Vitam Horm 72:431–456
Campbell MA, Haas BJ, Hamilton JP, Mount SM, Buell CR (2006) Comprehensive analysis of alternative splicing in rice and comparative analyses with Arabidopsis. BMC Genomics 7:327
Chagné D, Gasic K, Crowhurst RN, Han YP, Bassett HC, Bowatte DR, Lawrence TJ, Rikkerink EHA, Gardiner SE, Korban SS (2008) Development of a set of SNP markers present in expressed genes of the apple. Genomics 92:353–358
Cheng H, Song SS, **ao LT, Soo HM, Cheng ZW, **e DX, Peng JR (2009) Gibberellin acts through jasmonate to control the expression of MYB21, MYB24, and MYB57 to promote stamen filament growth in Arabidopsis. PLoS Genet 5:e1000440
Coen ES, Meyerowitz EM (1991) The war of the whorls: genetic interactions controlling flower development. Nature 353:31–37
Corbesier L, Coupland G (2006) The quest for florigen: a review of recent progress. J Exp Bot 57:3395–3403
Dassanayake M, Haas JS, Bohnert HJ, Cheeseman JM (2009) Shedding light on an extremophile lifestyle through transcriptomics. New Phytol 183:764–775
Denoeud F, Aury JM, Da Silva C, Noel B, Rogier O, Delledonne M, Morgante M, Valle G, Wincker P, Scarpelli C, Jaillon O, Artiguenave F (2009) Annotating genomes with massive-scale RNA sequencing. Genome Biol 9:R175
Devoto A, Turner JG (2003) Regulation of jasmonate-mediated plant responses in Arabidopsis. Ann Bot 92:329–337
Doyle MR, Davis SJ, Bastow RM, McWatters HG, Kozma-Bognar L, Nagy F, Millar AJ, Amasino RM (2002) The ELF4 gene controls circadian rhythms and flowering time in Arabidopsis thaliana. Nature 419:74–77
Fouquet R, Martin F, Fajardo DS, Gault CM, Gómez E, Tseung CW, Policht T, Hueros G, Settles AM (2011) Maize rough endosperm3 encodes an RNA splicing factor required for endosperm cell differentiation and has a nonautonomous effect on embryo development. Plant Cell 23:4280–4297
Gott JM, Emeson RB (2000) Functions and mechanisms of RNA editing. Annu Rev Genet 34:499–531
Herrero E, Davis SJ (2012) Time for a nuclear meeting: protein trafficking and chromatin dynamics intersect in the plant circadian system. Mol Plant 5:554–565
Huang SN, Liu ZY, Li DY, Yao RP, Meng Q, Feng H (2014) Screening of Chinese cabbage mutants produced by 60Co γ-ray mutagenesis of isolated microspore cultures. Plant Breed 133:480–488
Ingram GC, Doyle S, Carpenter R, Schultz EA, Simon R, Coen ES (1997) Dual role for fimbriata in regulating floral homeotic genes and cell division in Antirrhinum. EMBO J 16:6521–6534
James AB, Syed NH, Bordage S, Marshall J, Nimmo GA, Jenkins GI, Herzyk P, Brown JW, Nimmo HG (2012) Alternative splicing mediates responses of the Arabidopsis circadian clock to temperature changes. Plant Cell 24:961–981
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484
Kolmos E, Nowak M, Werner M, Fischer K, Schwarz G, Mathews S, Schoof H, Nagy F, Bujnicki JM, Davis SJ (2009) Integrating ELF4 into the circadian system through combined structural and functional studies. HFSP J 3:350–366
Komeda Y (2004) Genetic regulation of time to flower in Arabidosis thaliana. Annu Rev Plant Biol 55:521–535
Kriechbaumer V, Wang P, Hawes C, Abell BM (2012) Alternative splicing of the auxin biosynthesis gene YUCCA4 determines its subcellular compartmentation. Plant J 70:292–302
Krizek BA, Fletcher JC (2005) Molecular mechanisms of flower development: an armchair guide. Nat Rev Genet 6:688–698
Lee I, Wolfe DS, Nilsson O, Weigel D (1997) A LEAFY co-regulator encoded by UNUSUAL FLORAL ORGANS. Curr Biol 7:95–104
Levin JZ, Meyerowitz EM (1995) UFO: an Arabidopsis gene involved in both floral meristem and floral organ development. Plant Cell 7:529–548
Li P, Ponnala L, Gandotra N, Wang L, Si Y, Tausta SL, Kebrom TH, Provart N, Patel R, Myers CR, Reidel EJ, Turgeon R, Liu P, Sun Q, Nelson T, Brutnell TP (2010) The developmental dynamics of the maize leaf transcriptome. Nat Genet 42:1060–1067
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25:402–408
Lu TT, Lu GJ, Fan DL, Zhu CR, Li W, Zhao Q, Feng Q, Zhao Y, Guo YL, Li WJ, Huang XH, Han B (2010) Function annotation of the rice transcriptome at single-nucleotide resolution by RNA-Seq. Genome Res 20:1238–1249
Macknight R, Duroux M, Laurie R, Dijkwel P, Simpson G, Dean C (2002) Functional significance of the alternative transcript processing of the Arabidopsis floral promoter FCA. Plant Cell 14:877–888
Maher CA, Kumar-Sinha C, Cao X, Kalyana-Sundaram S, Han B, **g X, Sam L, Barrette T, Palanisamy N, Chinnaiyan AM (2009) Transcriptome sequencing to detect gene fusions in cancer. Nature 458:97–101
Martin C, Bhatt K, Baumann K, ** HL, Zachgo S, Roberts K, Schwarz-sommer Z, Glover B, Perez-Rodrigues M (2002) The mechanics of cell fate determination in petals. Philos Trans R Soc Lond B Biol Sci 357:809–813
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303
Moon J, Lee H, Kim M, Lee I (2005) Analysis of flowering pathway integrators in Arabidopsis. Plant Cell Physiol 46:292–299
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Map** and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5:621–628
Novaes E, Drost DR, Farmerie WG, Pappas GJ, Grattapaglia D, Sederoff RR, Kirst M (2008) High-throughput gene and SNP discovery in Eucalyptus grandis, an uncharacterized genome. BMC Genom 9:312
Peng J (2009) Gibberellin and jasmonate crosstalk during stamen development. J Integr Plant Biol 51:1064–1070
Picardi E, Horner DS, Chiara M, Schiavon R, Valle G, Pesole G (2010) Large-scale detection and analysis of RNA editing in grape mtDNA by RNA deep-sequencing. Nucleic Acids Res 38:4755–4767
Ramchiary N, Nguyen VD, Li XN, Hong CP, Dhandapani V, Choi SR, Yu G, Piao ZY, Lim YP (2011) Genic microsatellite markers in Brassica rapa: development, characterization, map**, and their utility in other cultivated and wild Brassica relatives. DNA Res 18:305–320
Rosloski SM, Singh A, Jali SS, Balasubramanian S, Weigel D, Grbic V (2013) Functional analysis of splice variant expression of MADS AFFECTING FLOWERING 2 of Arabidopsis thaliana. Plant Mol Biol 81:57–69
Shi CY, Yang H, Wei CL, Yu O, Zhang ZZ, Jiang CJ, Sun J, Li YY, Chen Q, **a T, Wan XC (2011) Deep sequencing of the Camellia sinensis transcriptome revealed candidate genes for major metabolic pathways of tea-specific compounds. BMC Genomics 12:131
Stamm S, Ben-Ari S, Rafalska I, Tang Y, Zhang Z, Toiber D, Thanaraj TA, Soreq H (2005) Function of alternative splicing. Gene 344:1–20
Steinhauser S, Beckert S, Capesius I, Malek O, Knoop V (1999) Plant mitochondrial RNA editing. J Mol Evol 48:303–312
Strickler SR, Bombarely A, Mueller LA (2012) Designing a transcriptome nextgeneration sequencing project for a nonmodel plant species. Am J Bot 99:257–266
Szécsi J, Joly C, Bordji K, Varaud E, Cock JM, Dumas C, Bendahmane M (2006) BIGPETALp, a bHLH transcription factor is involved in the control of Arabidopsis petal size. EMBO J 25:3912–3920
Tao X, Gu YH, Wang HY, Zheng W, Li X, Zhao CW, Zhang YZ (2012) Digital gene expression analysis based on integrated de novo transcriptome assembly of sweet potato [Ipomoea batatas (L.) Lam.]. PLoS One 7:e36234
Theissen G (2001) Development of floral organ identity: stories from the MADS house. Curr Opin Plant Biol 4:75–85
Theissen G, Becker A, Di Rosa A, Kanno A, Kim JT, Münster T, Winter KU, Saedler H (2000) A short history of MADS-box genes in plants. Plant Mol Biol 42:115–149
Trapnell C, Pachter L, Salzberg SL (2009) TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25:1105–1111
Wang BB, Brendel V (2006) Genomewide comparative analysis of alternative splicing in plants. Proc Natl Acad Sci 103:7175–7180
Wang ET, Sandberg R, Luo SJ, Khrebtukova I, Zhang L, Mayr C, Kingsmore SF, Schroth GP, Burge CB (2008) Alternative isoform regulation in human tissue transcriptomes. Nature 456:470–476
Wang W, Wang Y, Zhang Q, Qi Y, Guo D (2009a) Global characterization of Artemisia annua glandular trichome transcriptome using 454 pyrosequencing. BMC Genom 10:465
Wang Z, Gerstein M, Snyder M (2009b) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10:57–63
Wang L, Feng Z, Wang X, Wang X, Zhang X (2010) DEGseq: an R package for identifying differentially expressed genes from RNAseq data. Bioinformatics 26:136–138
Wang XW, Wang HZ, Wang J, Sun RF, Wu J, Liu SY, Bai YQ, Mun JH, Bancroft I, Cheng F, Huang S, Li X, Hua W, Wang J, Wang X, Freeling M, Pires JC, Paterson AH, Chalhoub B, Wang B, Hayward A, Sharpe AG, Park BS, Weisshaar B, Liu B, Li B, Liu B, Tong C, Song C, Duran C, Peng C, Geng C, Koh C, Lin C, Edwards D, Mu D, Shen D, Soumpourou E, Li F, Fraser F, Conant G, Lassalle G, King GJ, Bonnema G, Tang H, Wang H, Belcram H, Zhou H, Hirakawa H, Abe H, Guo H, Wang H, ** H, Parkin IA, Batley J, Kim JS, Just J, Li J, Xu J, Deng J, Kim JA, Li J, Yu J, Meng J, Wang J, Min J, Poulain J, Wang J, Hatakeyama K, Wu K, Wang L, Fang L, Trick M, Links MG, Zhao M, ** M, Ramchiary N, Drou N, Berkman PJ, Cai Q, Huang Q, Li R, Tabata S, Cheng S, Zhang S, Zhang S, Huang S, Sato S, Sun S, Kwon SJ, Choi SR, Lee TH, Fan W, Zhao X, Tan X, Xu X, Wang Y, Qiu Y, Yin Y, Li Y, Du Y, Liao Y, Lim Y, Narusaka Y, Wang YP, Wang ZY, Li ZY, Wang ZW, **ong ZY, Zhang ZH (2011) The genome of the mesopolyploid crop species Brassica rapa. Nat Genet 43:1035–1039
Weigel D, Meyerowitz EM (1994) The ABCs of floral homeotic genes. Cell 78:203–209
Yang CP, Wang YC, Liu GF, Jiang J (2004) Expression of some genes in Tamarix androssowii plant under NaHCO3 stress. J of plant Physiol and Mol Biol 30:229–233
Yong WD, Zhong K, Xu ZH, Tan KH, Zhu ZQ (2000) Gene regulation study of flowering time determination in higher plants. Chin Sci Bull 45:455–466
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-Seq: accounting for selection bias. Genome Biol 11:R14
Zahn LM, Ma X, Altman NS, Zhang Q, Wall PK, Tian D, Gibas CJ, Gharaibeh R, Leebens-Mack JH, Depamphilis CW, Ma H (2010) Comparative transcriptomics among floral organs of the basal eudicot Eschscholzia californica as reference for floral evolutionary developmental studies. Genome Biol 11:R101
Zenoni S, Ferrarini A, Giacomelli E, Xumerle L, Fasoli M, Malerba G, Bellin D, Pezzotti M, Delledonne M (2010) Characterization of transcriptional complexity during berry development in Vitis vinifera using RNA-Seq. Plant Physiol 152:1787–1795
Zhang CK, Lang P, Dane F, Ebel RC, Singh NK, Locy RD, Dozier WA (2005) Cold acclimation induced genes of trifoliate orange (Poncirus trifoliata). Plant Cell Rep 23:764–769
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (31272157) and the Postgraduate Innovation Cultivation Project of Shenyang Agricultural University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
S. Huang and Z. Liu contributed equally to this work and share the first authorship
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2015_1041_MOESM6_ESM.xls
Table S4. Summary of SNPs detected in the EP and NEP libraries (XLS) The meaning of numbers in the columns labeled ‘EP’ and ‘NEP’ were as follows, ‘1’: SNP (genotype was consistent with the column labeled ‘ALT’), ‘0’: None (genotype was consistent with the column labeled ‘REF’), ‘.’: Uncertained (lack of data support) (XLS 4082 kb)
438_2015_1041_MOESM7_ESM.xls
Table S5. Summary of Indels detected in the EP and NEP libraries (XLS) The meaning of numbers in the columns labeled ‘EP’ and ‘NEP’ were as follows, ‘1’: Indel (genotype was consistent with the column labeled ‘ALT’), ‘0’: None (genotype was consistent with the column labeled ‘REF’), ‘.’: Uncertained (lack of data support) (XLS 358 kb)
Rights and permissions
About this article
Cite this article
Huang, S., Liu, Z., Yao, R. et al. Comparative transcriptome analysis of the petal degeneration mutant pdm in Chinese cabbage (Brassica campestris ssp. pekinensis) using RNA-Seq. Mol Genet Genomics 290, 1833–1847 (2015). https://doi.org/10.1007/s00438-015-1041-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-015-1041-7