Abstract
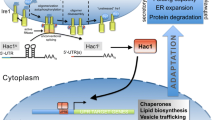
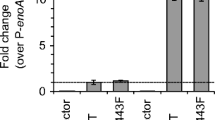
A signal transduction pathway called the unfolded protein response is activated when increased levels of misfolded proteins or incorrectly assembled subunits accumulate in the endoplasmic reticulum (ER). The expression of several genes for ER-resident foldases and chaperones, as well as genes encoding proteins that are involved in functions associated with the secretory process, are induced by this pathway. This paper describes the cloning and characterisation of genes for two components of the pathway, ire1 and ptc2, from the filamentous fungus Trichoderma reesei ( Hypocrea jecorina). The data presented demonstrates that the T. reesei genes can complement Saccharomyces cerevisiae mutants that are deficient in the corresponding homologues. The T. reesei IREI protein has intrinsic kinase activity, as revealed by an in vitro autophosphorylation assay. Overexpression of ire1 in a T. reesei strain that expresses a foreign protein (laccase 1 from Phlebia radiata), results in up-regulation of the UPR pathway, as indicated by the increased expression levels of the known UPR target genes bip1 and pdi1. Splicing of the mRNA encoding the transcription factor HAC1 is also observed. Other genes encoding proteins from different parts of the secretory pathway also respond to ire1 overexpression.





Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Meyers EW, Lipman DJ (1990) Basic Local Alignment Search Tool*1. J Mol Biol 215:403–410
Bailey MJ, Linko M (1990) Production of β-galactosidase by Aspergillus oryzae in submerged bioreactor cultivation. J Biotechnol 16:57–66
Bertolotti A, Zhang Y, Hendershot LM, Harding HP, Ron D (2000) Dynamic interaction of BiP and ER stress transducers in the unfolded- protein response. Nat Cell Biol 2:326–332
Calfon M, Zeng H, Urano F, Till JH, Hubbard SR, Harding HP, Clark SG, Ron D (2002) IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 415:92–96
Chang HJ, Jones EW, Henry SA (2002) Role of the Unfolded Protein Response pathway in regulation of INO1 and in the sec14 bypass mechanism in Saccharomyces cerevisiae. Genetics 162:29–43
Cox JS, Walter P (1996) A novel mechanism for regulating activity of a transcription factor that controls the unfolded protein response. Cell 87:391–404
Cox JS, Shamu CE, Walter P (1993) Transcriptional induction of genes encoding endoplasmic reticulum resident proteins requires a transmembrane protein kinase. Cell 73:1197–1206
Dorner AJ, Wasley LC, Kaufman RJ (1989) Increased synthesis of secreted proteins induces expression of glucose- regulated proteins in butyrate-treated Chinese hamster ovary cells. J Biol Chem 264:20602–20607
Falquet L, Pagni M, Bucher P, Hulo N, Sigrist CJA, Hofmann K, Bairoch A (2002) The PROSITE database, its status in 2002. Nucleic Acids Res 30:235–238
Haze K, Yoshida H, Yanagi H, Yura T, Mori K (1999) Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol Biol Cell 10:3787–3799
Kawahara T, Yanagi H, Yura T, Mori K (1998) Unconventional splicing of HAC1/ERN4 mRNA required for the unfolded protein response. Sequence-specific and non-sequential cleavage of the splice sites. J Biol Chem 273:1802–1807
Lee K, Tirasophon W, Shen X, Michalak M, Prywes R, Okada T, Yoshida H, Mori K, Kaufman RJ (2002) IRE1-mediated unconventional mRNA splicing and S2P-mediated ATF6 cleavage merge to regulate XBP1 in signaling the unfolded protein response. Genes Dev 16:452–466
Martinez IM, Chrispeels MJ (2003) Genomic analysis of the Unfolded Protein Response in Arabidopsis shows its connection to important cellular processes. Plant Cell 15:561–576
Montenecourt BS, Eveleigh DE (1979) Selective screening methods for the isolation of high yielding mutants of Trichoderma reesei. Adv Chem Ser 181:289–301
Mori K, Ma W, Gething MJ, Sambrook J (1993) A transmembrane protein with a cdc2+/CDC28-related kinase activity is required for signaling from the ER to the nucleus. Cell 74:743–756
Mori K, Ogawa N, Kawahara T, Yanagi H, Yura T (2000) mRNA splicing-mediated C-terminal replacement of transcription factor Hac1p is required for efficient activation of the unfolded protein response. Proc Natl Acad Sci USA 97:4660–4665
Mulder HJ, Saloheimo M, Penttilä M, Madrid SM (2004) The transcription factor HACA mediates the unfolded protein response in Aspergillus niger, and up-regulates its own transcription. Mol Genet Genomics 271:130–140
Nikawa J, Yamashita S (1992) IRE1 encodes a putative protein kinase containing a membrane-spanning domain and is required for inositol phototrophy in Saccharomyces cerevisiae. Mol Microbiol 6:1441–1446
Niku-Paavola ML, Karhunen E, Salola P, Raunio V (1988) Ligninolytic enzymes of the white-rot fungus Phlebia radiata. Biochem J 254:877–884
Nyfeler B, Nufer O, Matsui T, Mori K, Hauri HP (2003) The cargo receptor ERGIC-53 is a target of the unfolded protein response. Biochem Biophys Res Comm 304:599-604.
Patil C, Walter P (2001) Intracellular signaling from the endoplasmic reticulum to the nucleus: the unfolded protein response in yeast and mammals. Curr Opin Cell Biol 13:349–355
Penttilä M, Nevalainen H, Rättö M, Salminen E, Knowles J (1987) A versatile transformation system for the cellulolytic filamentous fungus Trichoderma reesei. Gene 61:155–164
Punt PJ, van Gemeren IA, Drint-Kuijvenhoven J, Hessing JG, van Muijlwijk-Harteveld GM, Beijersbergen A, Verrips CT, van den Hondel CA (1998) Analysis of the role of the gene bipA, encoding the major endoplasmic reticulum chaperone protein in the secretion of homologous and heterologous proteins in black Aspergilli. Appl Microbiol Biotechnol 50:447–454
Robinson AS, Hines V, Wittrup KD (1994) Protein disulfide isomerase overexpression increases secretion of foreign proteins in Saccharomyces cerevisiae. Biotechnology (NY) 12:381–384
Saloheimo M, Niku-Paavola ML, Knowles JK (1991) Isolation and structural analysis of the laccase gene from the lignin-degrading fungus Phlebia radiata. J Gen Microbiol 137:1537–1544
Saloheimo A, Henrissat B, Hoffren AM, Teleman O, Penttilä M (1994) A novel, small endoglucanase gene, egl5 , from Trichoderma reesei isolated by expression in yeast. Mol Microbiol 13:219–228
Saloheimo M, Lund M, Penttilä ME (1999) The protein disulphide isomerase gene of the fungus Trichoderma reesei is induced by endoplasmic reticulum stress and regulated by the carbon source. Mol Gen Genet 262:35–45
Saloheimo M, Valkonen M, Penttilä M (2003) Activation mechanisms of the HACI-mediated unfolded protein response in filamentous fungi. Mol Microbiol 47:1149–1161
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual (2nd edn). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Sherman F (1991) Getting started with yeast. Met Enzymol 194:3–21
Sidrauski C, Walter P (1997) The transmembrane kinase Ire1p is a site-specific endonuclease that initiates mRNA splicing in the unfolded protein response. Cell 90:1031–1039
Sidrauski C, Cox JS, Walter P (1996) tRNA ligase is required for regulated mRNA splicing in the unfolded protein response. Cell 87:405–413
Smith DB, Johnson KS (1988) Single-step purification of polypeptides expressed in Escherichia coli as fusions with glutathione S-transferase. Gene 67:31–40
Stålbrand H, Saloheimo A, Vehmaanperä J, Henrissat B, Penttilä M (1995) Cloning and expression in Saccharomyces cerevisiae of a Trichoderma reesei β-mannanase gene containing a cellulose binding domain. Appl Environ Microbiol 61:1090–1097
Stroobants AK, Hettema EH, van den Berg M, Tabak HF (1999) Enlargement of the endoplasmic reticulum membrane in Saccharomyces cerevisiae is not necessarily linked to the unfolded protein response via Ire1p. FEBS Lett 453:210–214
Travers KJ, Patil CK, Wodicka L, Lockhart DJ, Weissman JS, Walter P (2000) Functional and genomic analyses reveal an essential coordination between the unfolded protein response and ER-associated degradation. Cell 101:249–258
Valkonen M, Penttilä M, Saloheimo M (2003a) Effects of inactivation and constitutive expression of the Unfolded-Protein Response pathway on protein production in the yeast Saccharomyces cerevisiae. Appl Environ Microbiol 69:2065–2072
Valkonen M, Ward M, Wang H, Penttilä M, Saloheimo M (2003b) Improvement of foreign protein production in Aspergillus niger var. awamori by constitutive induction of the Unfolded-Protein Response. Appl Environ Microbiol 69:6979–6986
Van Tilbeurgh H, Claeyssens M, de Bruyne CK (1982) The use of 4-methylumbelliferyl and other chromophoric glycosides in the study of cellulolytic enzymes. FEBS Lett 149:152–156
Welihinda AA, Kaufman RJ (1996) The unfolded protein response pathway in Saccharomyces cerevisiae. Oligomerization and trans-phosphorylation of Ire1p (Ern1p) are required for kinase activation. J Biol Chem 271:18181–18187
Welihinda AA, Tirasophon W, Green SR, Kaufman RJ (1998) Protein serine/threonine phosphatase Ptc2p negatively regulates the unfolded-protein response by dephosphorylating Ire1p kinase. Mol Cell Biol 18:1967–1977
Yoshida H, Haze K, Yanagi H, Yura T, Mori K (1998) Identification of the cis -acting endoplasmic reticulum Stress Response Element responsible for transcriptional induction of mammalian glucose-regulated proteins. Involvement of basic leucine zipper transcription factors. J Biol Chem 273:33741–33749
Acknowledgements
The authors thank Riitta Nurmi for excellent technical assistance, Dr. Randal Kaufman for S. cerevisiae strains and Drs Michael Ward and Huaming Wang for fruitful discussions. The work was supported by Genencor International Inc. and the Finnish Technology Agency (Tekes) and partly by the Academy of Finland in the VTT Industrial Biotechnology Finnish Centre of Excellence Program 2000–2005, Project 64330.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Cerdá-Olmedo
Rights and permissions
About this article
Cite this article
Valkonen, M., Penttilä, M. & Saloheimo, M. The ire1 and ptc2 genes involved in the unfolded protein response pathway in the filamentous fungus Trichoderma reesei. Mol Genet Genomics 272, 443–451 (2004). https://doi.org/10.1007/s00438-004-1070-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-004-1070-0