Abstract
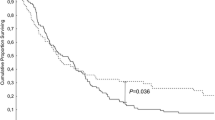
Pancreatoduodenectomy is one of the most challenging surgical specimens for pathologists. Recently, two different, standardized protocols have been proposed: the axial slicing Leeds protocol (LP) and the bi-valving Adsay protocol (AP). Comparison between standardized and non-standardized protocols (NSP) was performed with emphasis on margin involvement and lymph node yield. Pancreatoduodenectomy cases were retrospectively recruited: 46 sampled with LP, 52 cases with AP and 46 cases with NSP. Clinico-pathologic data and rates of margin/surface involvement were collected and their prognostic influence on survival was assessed. Statistical differences between NSP and AP and LP were seen for nodal yield (p = 0.0001), N+ (p = 0.0001) and lymph node ratio - LNR (p < 0.0008) but not between AP and LP. Differences in R1/R0 status were statistically significant between NSP group (R1–15%) and both the LP (R1–73.9%) and AP (R1–70%) groups (p = 0.0001) but not between LP and AP groups. At univariate survival analysis, grade (p = 0.0023) and number of involved margins (p = 0.0096) in AP and “N-category” (p = 0.0057) “resection margin status” (p = 0.0094), “stage” (p = 0.0143), and “number of involved margins” (p = 0.00398) in LP were statistically significant, while no variable was significant in the NSP group. At multivariate analysis “N category,” “resection margin status,” “stage,” “number of involved margins,” and “LNR” retained significance for the LP group. These results show that both LP and AP perform better than non-standardized sampling making standardization mandatory in pancreatoduodenectomy cut up. Both AP and LP show strengths and weaknesses, and these may impact on the choice of protocol in different institutions.


Similar content being viewed by others
References
Malvezzi M, Bertuccio P, Levi F, La Vecchia C, Negri E (2014) European cancer mortality predictions for the year 2014. Ann Oncol 25:1650–1656. https://doi.org/10.1093/annonc/mdu138
Ducreux M, Cuhna AS, Caramella C, Hollebecque A, Burtin P, Goéré D, Seufferlein T, Haustermans K, Van Laethem JL, Conroy T, Arnold D, Guidelines Committee ESMO (2015) Cancer of the pancreas: ESMO clinical practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 26:v56–v68. https://doi.org/10.1093/annonc/mdv295
Winter JM, Cameron JL, Campbell KA, Arnold MA, Chang DC, Coleman J, Hodgin MB, Sauter PK, Hruban RH, Riall TS, Schulick RD, Choti MA, Lillemoe KD, Yeo CJ (2006) 1423 pancreaticoduodenectomies for pancreatic cancer: a single-institution experience. J Gastrointest Surg 10:1199–1210
Neoptolemos JP, Stocken DD, Dunn JA, Almond J, Beger HG, Pederzoli P, Bassi C, Dervenis C, Fernandez-Cruz L, Lacaine F, Buckels J, Deakin M, Adab FA, Sutton R, Imrie C, Ihse I, Tihanyi T, Olah A, Pedrazzoli S, Spooner D, Kerr DJ, Friess H, Büchler MW, European Study Group for Pancreatic Cancer (2001) Influence of resection margins on survival for patients with pancreatic cancer treated by adjuvant chemoradiation and/or chemotherapy in the ESPAC-1 randomized controlled trial. Ann Surg 234:758–768
Paniccia A, Hosokawa P, Henderson W, Schulick RD, Edil BH, McCarter MD, Gajdos C (2015) Characteristics of 10-year survivors of pancreatic ductal adenocarcinoma. JAMA Surg 150:701–710. https://doi.org/10.1001/jamasurg.2015.0668
Schmidt CM, Powell ES, Yiannoutsos CT, Howard TJ, Wiebke EA, Wiesenauer CA, Baumgardner JA, Cummings OW, Jacobson LE, Broadie TA, Canal DF, Goulet RJ Jr, Curie EA, Cardenes H, Watkins JM, Loehrer PJ, Lillemoe KD, Madura JA (2004) Pancreaticoduodenectomy: a 20-year experience in 516 patients. Arch Surg 139:718–725
Raut CP, Tseng JF, Sun CC, Wang H, Wolff RA, Crane CH, Hwang R, Vauthey JN, Abdalla EK, Lee JE, Pisters PW, Evans DB (2007) Impact of resection status on pattern of failure and survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Ann Surg 246:52–60
Verbeke CS, Leitch D, Menon KV, McMahon MJ, Guillou PJ, Anthoney A (2006) Redefining the R1 resection in pancreatic cancer. Br J Surg 93:1232–1237
Menon KV, Gomez D, Smith AM, Anthoney A, Verbeke CS (2009) Impact of margin status on survival following pancreatoduodenectomy for cancer: the Leeds pathology protocol (LEEPP). HPB (Oxford) 11:18–24. https://doi.org/10.1111/j.1477-2574.2008.00013.x
Verbeke CS, Gladhaug IP (2016) Dissection of pancreatic resection specimens. Surg Pathol Clin 9:523–538. https://doi.org/10.1016/j.path.2016.05.001
Adsay NV, Basturk O, Saka B, Bagci P, Ozdemir D, Balci S, Sarmiento JM, Kooby DA, Staley C, Maithel SK, Everett R, Cheng JD, Thirabanjasak D, Weaver DW (2014) Whipple made simple for surgical pathologists: orientation, dissection, and sampling of pancreaticoduodenectomy specimens for a more practical and accurate evaluation of pancreatic, distal common bile duct, and ampullary tumors. Am J Surg Patho 38:480–493. https://doi.org/10.1097/PAS.0000000000000165
Soer E, Brosens L, van de Vijver M, Dijk F, van Velthuysen ML, Farina-Sarasqueta A, Morreau H, Offerhaus J, Koens L, Verheij J (2018) Dilemmas for the pathologist in the oncologic assessment of pancreatoduodenectomy specimens: an overview of different grossing approaches and the relevance of the histopathological characteristics in the oncologic assessment of pancreatoduodenectomy specimens. Virchows Arch 472:533–543. https://doi.org/10.1007/s00428-018-2321-5
Takahashi D, Kojima M, Sugimoto M, Kobayashi S, Takahashi S, Konishi M, Gotohda N, Nagino M (2018) Pathologic evaluation of surgical margins in pancreatic cancer specimens using color coding with tissue marking dyes. Pancreas 47:830–836. https://doi.org/10.1097/MPA.0000000000001106
Casadei R, Ricci C, Taffurelli G, Pacilio CA, Santini D, Di Marco M, Minni F (2018) Multicolour versus monocolour inking specimens after pancreaticoduodenectomy for periampullary cancer: a single Centre prospective randomised clinical trial. Int J Surg 51:63–70. https://doi.org/10.1016/j.ijsu.2018.01.021
Schwarz RE, Smith DD (2006) Extent of lymph node retrieval and pancreatic cancer survival: information from a large US population database. Ann Surg Oncol 13:1189–2000. https://doi.org/10.1245/s10434-006-9016-x
Slidell MB, Chang DC, Cameron JL, Wolfgang C, Herman JM, Schulick RD, Choti MA, Pawlik TM (2008) Impact of total lymph node count and lymph node ratio on staging and survival after pancreatectomy for pancreatic adenocarcinoma: a large, population-based analysis. Ann Surg Oncol 15:165–174. https://doi.org/10.1245/s10434-007-9587-1
Konstantinidis IT, Warshaw AL, Allen JN, Blaszkowsky LS, Castillo CF, Deshpande V, Hong TS, Kwak EL, Lauwers GY, Ryan DP, Wargo JA, Lillemoe KD, Ferrone CR (2013) Pancreatic ductal adenocarcinoma: is there a survival difference for R1 resections versus locally advanced unresectable tumors? What is a "true" R0 resection? Ann Surg 257:731–736. https://doi.org/10.1097/SLA.0b013e318263da2f
Campbell F, Smith RA, Whelan P, Sutton R, Raraty M, Neoptolemos JP, Ghaneh P (2009) Classification of R1 resections for pancreatic cancer: the prognostic relevance of tumour involvement within 1 mm of a resection margin. Histopathology 55:277–283. https://doi.org/10.1111/j.1365-2559.2009.03376.x
Verbeke CS (2008) Resection margins and R1 rates in pancreatic cancer - are we there yet? Histopathology 52:787–796
Staley CA, Cleary KR, Abbruzzese JL, Lee JE, Ames FC, Fenoglio CJ, Evans DB (1996) The need for standardized pathologic staging of pancreaticoduodenectomy specimens. Pancreas 12:373–380
Lüttges J, Zamboni G, Klöppel G (1999) Recommendation for the examination of pancreaticoduodenectomy specimens removed from patients with carcinoma of the exocrine pancreas. Dig Surg 16:291–296
Tomlinson JS, Jain S, Bentrem DJ, Sekeris EG, Maggard MA, Hines OJ, Reber HA, Ko CY (2007) Accuracy of staging node-negative pancreas cancer: a potential quality measure. Arch Surg 142:767–773. https://doi.org/10.1001/archsurg.142.8.767
Warschkow R, Widmann B, Beutner U, Marti L, Steffen T, Schiesser M, Schmied BM (2017) The more the better-lower rate of stage migration and better survival in patients with retrieval of 20 or more regional lymph nodes in pancreatic cancer: a population-based propensity score matched and trend SEER analysis. Pancreas 46:648–657. https://doi.org/10.1097/MPA.0000000000000784
Adsay NV, Basturk O, Altinel D, Khanani F, Coban I, Weaver DW, Kooby DA, Sarmiento JM, Staley C (2009) The number of lymph nodes identified in a simple pancreatoduodenectomy specimen: comparison of conventional vs orange-peeling approach in pathologic assessment. Mod Pathol 22:107–112. https://doi.org/10.1038/modpathol.2008.167
Pawlik TM, Gleisner AL, Cameron JL, Winter JM, Assumpcao L, Lillemoe KD, Wolfgang C, Hruban RH, Schulick RD, Yeo CJ, Choti MA (2007) Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery 141:610–618
Esposito I, Kleeff J, Bergmann F, Reiser C, Herpel E, Friess H, Schirmacher P, Büchler MW (2008) Most pancreatic cancer resections are R1 resections. Ann Surg Oncol 15:1651–1660. https://doi.org/10.1245/s10434-008-9839-8
Osipov A, Nissen N, Rutgers J, Dhall D, Naziri J, Chopra S, Li Q, Hendifar AE, Tuli R (2017) Redefining the positive margin in pancreatic cancer: impact on patterns of failure, long-term survival and adjuvant therapy. Ann Surg Oncol 24:3674–3682. https://doi.org/10.1245/s10434-017-6076-z
Gnerlich JL, Luka SR, Deshpande AD, Dubray BJ, Weir JS, Carpenter DH, Brunt EM, Strasberg SM, Hawkins WG, Linehan DC (2012) Microscopic margins and patterns of treatment failure in resected pancreatic adenocarcinoma. Arch Surg 147:753–760. https://doi.org/10.1001/archsurg.2012.1126
Zhang Y, Frampton AE, Cohen P, Kyriakides C, Bong JJ, Habib NA, Spalding DR, Ahmad R, Jiao LR (2012) Tumor infiltration in the medial resection margin predicts survival after pancreaticoduodenectomy for pancreatic ductal adenocarcinoma. J Gastrointest Surg 16:1875–1882. https://doi.org/10.1007/s11605-012-1985-4
Acknowledgments
We wish to thank Simona Pigozzi for technical help.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Federica Grillo, Jacopo Ferro, Alessandro Vanoli, Sara Delfino, Francesca Pitto, Leonardo Peñuela, Rita Bianchi, Oneda Grami, and Luca Mastracci. The first draft of the manuscript was written by Federica Grillo, Alessandro Vanoli, Leonardo Peñuela, and Luca Mastracci, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Informed consent
This is a retrospective study in which all patients signed informed consent for surgery and histologic diagnosis. All patients were anonymized, and no new histological sections were produced.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the topical collection on Quality in Pathology
Rights and permissions
About this article
Cite this article
Grillo, F., Ferro, J., Vanoli, A. et al. Comparison of pathology sampling protocols for pancreatoduodenectomy specimens. Virchows Arch 476, 735–744 (2020). https://doi.org/10.1007/s00428-019-02687-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-019-02687-6