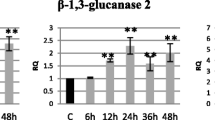
Abstract
Lignin and lignans share monolignols as common precursors and are both potentially involved in plant defence against pathogens. In this study, we investigated the effects of fungal elicitors on lignin and lignan metabolism in flax (Linum usitatissimum) cell suspensions. Cell suspension cultures of flax were treated with elicitor preparations made from mycelium extracts of Botrytis cinerea, Phoma exigua and Fusarium oxysporum F ssp lini. Elicitors induced a rapid stimulation of the monolignol pathway, as confirmed by the increase in PAL (phenylalanine ammonia-lyase, EC 4.1.3.5), CCR (cinnamoyl-CoA reductase EC 1.2.1.44) and CAD (cinnamyl alcohol dehydrogenase EC 1.1.1.195) gene expression and PAL activity. At the same time, CCR activity only increased significantly in F. oxysporum-treated cells 24 h post elicitation. On the other hand, CAD activity measured for coniferyl alcohol formation was transiently decreased but a substrate-specific activation of CAD activity was observed in F. oxysporum-treated cells when using sinapyl alcohol as substrate. The accumulation of monolignol-derived products varied according to the elicitor used. B. cinerea or P. exigua-elicited cell cultures were characterised by a reinforcement of the cell wall by a deposit of 8-O-4′-linked non-condensed lignin structures and phenolic monomers, while at the same time no stimulation of 8-8′-linked lignan or 8-5′-linked phenylcoumaran lignan accumulation was observed. Additionally, elicitation of cell cultures with F. oxysporum extracts even triggered a strong incorporation of monolignols in the non condensed labile ether-linked lignin fraction concomitantly with a decrease in lignan and phenylcoumaran lignan accumulation. Several hypotheses are proposed to explain the putative role of these compounds in the defence response of flax cells against pathogens.







Similar content being viewed by others
Abbreviations
- ANHSEC:
-
Anhydro-secoisolariciresinol
- CAD:
-
Cinnamyl alcohol dehydrogenase
- CCR:
-
Cinnamoyl-CoA reductase;
- DCG:
-
Dehydrodiconiferyl alcohol glucoside
- PAL:
-
Phenylalanine ammonia lyase
- PCBER:
-
Phenylcoumaran benzylic ether reductase
- PLR:
-
Pinoresinol lariciresinol reductase
- SAD:
-
Sinapyl alcohol dehydrogenase
- SECO:
-
Secoisolariciresinol
References
Allwood EG, Davies DR, Gerrish C, Ellis BE, Bolwell GP (1999) Phosphorylation of phenylalanine ammonia-lyase: evidence for a novel protein kinase and identification of the phosphorylated residue. FEBS Lett 457:47–52
Akin DE, Rigsby LL, Gamble GR, Morisson WH III, Kimball BA, Pinter PJ Jr, Wall GW, Garcia RL, LaMorte RL (1995) Biodegradation of plant cell walls, wall carbohydrates, and wall aromatics in wheat grown in ambient or enriched CO2 concentrations. J Sci Food Agric 67:399–406
Attoumbre J, Hano C, Mesnard F, Lamblin F, Bensaddek L, Raynaud- Le Grandic S, Laine E, Baltora-Rosset S, Fliniaux MA (2005) Identification by NMR and accumulation of a neolignan, the dehydrodiconiferyl alcohol-4-β-D-glucoside, in Linum usitatissimum cell cultures. Comptes Rendus Chimie
Baucher M, Chabbert B, Pilate G, Van Doorsselaere J, Tollier MT, Petit-Conil M, Cornu D, Monties B, Van Montagu M, Inze D, Jouanin L, Boerjan W (1996) Red xylem and higher lignin extraction by down-regulating a cinnamyl alcohol dehydrogenase in poplar. Plant Physiol 112:1479–1490
Beuerle T, Pichersky E (2002) Enzymatic synthesis and purification of aromatic Coenzyme A esters. Anal Biochem 302:305–312
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annu Rev Plant Biol 54:519–546
Borevitz JO, **a Y, Blount J, Dixon RA, Lamb C (2000) Activation tagging identifies a conserved MYB regulator of phenylpropanoid biosynthesis. Plant Cell 12:2383–2393
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brill EM, Abrahams S, Hayes CM, Jenkins CLD, Watson JM (1999) Molecular characterisation and expression of a wound-inducible cDNA encoding a novel cinnamyl-alcohol dehydrogenase enzyme in lucerne (Medicago sativa L.). Plant Mol Biol 41:279–291
Campbell MM, Ellis BE (1992) Fungal elicitor-mediated responses in pine cell cultures: cell wall-bound phenolics. Phytochemistry 31:737–742
Charlet S, Bensaddek L, Raynaud S, Gillet F, Mesnard F, Fliniaux MA (2002) An HPLC procedure for the quantification of anhydrosecoisolariciresinol. Application to the evaluation of flax lignan content. Plant Physiol Biochem 40:225–229
Davin LB, Wang HB, Crowell AL, Bedgar LD, Martin DM, Sarkanen S, Lewis NG (1997) Stereoselective bimolecular phenoxy radical coupling by an auxiliary (dirigent) protein without an active center. Science 275:362–366
De Ascensao AR, Dubery IA (2003) Soluble and wall-bound phenolics and phenolic polymers in Musa acuminata roots exposed to elicitors from Fusarium oxysporum f.sp. cubense. Phytochemistry 63:679–86
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7:1085–1097
Dixon RA, Achnine L, Kota P, Liu CJ, Reddy MSS, Wang L (2002) The phenylpropanoid pathway and plant defense–a genomics perspective. Mol Plant Pathol 3:371–390
Dunn DC, Duncan LW, Romeo JT (1998) Changes in arginine, PAL activity, and nematode behavior in salinity-stressed citrus. Phytochemistry 49:413–417
Faix O (1991) Classification of lignins from different botanical origins by FT-IR spectroscopy. Holzforschung 45:21–27
Ford JD, Huang KS, Wang HB, Davin LB, Lewis NG (2001) Biosynthetic pathway to the cancer chemopreventive secoisolariciresinol diglucoside-hydroxymethyl glutaryl ester-linked lignan oligomers in flax (Linum usitatissimum) seed. J Nat Prod 64:1388–1397
Fuss E (2003) Lignans in plant cell and organ cultures: An overview. Phytochem Rev 2:307–320
Gang DR, Kasahara H, **a ZQ, Vander Mijnsbrugge K, Bauw G, Boerjan W, Van Montagu M, Davin LB, Lewis NG (1999) Evolution of plant defense mechanisms. Relationships of phenylcoumaran benzylic ether reductases to pinoresinol-lariciresinol and isoflavone reductases. J Biol Chem 274:7516–7527
Goffner D, Campbell MM, Campargue C, Clastre M, Borderies G, Boudet A, Boudet AM (1994) Purification and characterization of cinnamoyl-coenzyme A:NADP oxidoreductase in Eucalyptus gunnii. Plant Physiol 106:625–632
Goujon T, Ferret V, Mila I, Pollet B, Ruel K, Burlat V, Joseleau J-P, Barrière Y, Lapierre C, Jouanin L (2003) Down regulation of the AtCCR1 gene in Arabidopsis thaliana: effects on phenotype, lignins and cell wall degradability. Planta 217:218–228
Hawkins SW, Boudet AM (1994) Purification and characterization of cinnamyl alcohol dehydrogenase isoforms from the periderm of Eucalyptus gunnii Hook. Plant Physiol 104:75–84
Hawkins SW, Boudet AM (2003) Defense lignin and hydroxycinnamyl alcohol dehydrogenase activities in wounded Eucalyptus gunnii. Forest Pathol 33:91–104
Iiyama L, Lam TBT, Stone BA (1990) Phenolic acid bridges between polysaccharides and lignin in wheat straw internodes. Phytochemistry 29:733–737
Kiedrowski S, Kawalleck P, Hahlbrock K, Somssich IE, Dangl JL (1992) Rapid activation of a novel plant defence gene is strictly dependent on the Arabidopsis RPM1 disease resistance locus. EMBO J 11:4677–4684
Lamblin F, Saladin G, Dehorter B, Cronier D, Grenier E, Lacoux J, Bruyant P, Laine E, Chabbert B, Girault F, Monties B, Morvan C, David H, David A (2001) Overexpression of a heterologous sam gene encoding S-adenosylmethionine synthetase in flax (Linum usitatissimum) cells: Consequences on methylation of lignin precursors and pectins. Physiol Plant 112:223–232
Lange BM, Lapierre C, Sandermann H Jr (1995) Elicitor-induced spruce stress lignin (structural similarity to early developmental lignins). Plant Physiol 108:1277–1287
Lapierre C, Monties B, Rolando C (1986) Thioacidolysis of poplar lignins: identification of monomeric syringyl products and characterization of guaiacyl-syringyl lignin fractions. Holzforschung 40:113–118
Lauvergeat V, Lacomme C, Lacombe C, Lasserre E, Roby D, Grima-Pettenati J (2001) Two cinnamoyl-CoA reductase (CCR) genes from Arabidopsis thaliana are differentially expressed during development and in response to infection with pathogenic bacteria. Phytochemistry 57:1187–1195
Lewis NG, Davin LB, Sarkanen S (1998) Lignin and lignan biosynthesis: distinctions and reconciliations. In: Lewis NG, Sarkanen S (eds) Lignin and Lignan Biosynthesis. ACS Symposium Series, Washington pp 1–27
MacRae DW, Towers GHN (1984) Biological activities of lignans. Phytochemistry 23:1207–1220
Mitchell HJ, Hall JL, Barber MS (1994) Elicitor-induced cinnamyl alcohol dehydrogenase activity in lignifying wheat (Triticum aestivum L.) leaves. Plant Physiol 104:551–556
Mitchell HJ, Hall SA, Stratford R, Hall JL, Barber MS (1999) Differential induction of cinnamyl alcohol dehydrogenase during defensive lignification in wheat (Triticum aestivum L.): characterisation of the major inducible form. Planta 208:31–37
Morreel K, Ralph J, Kim H, Lu F, Goeminne G, Ralph S, Messens E, Boerjan W (2004) Profiling of oligolignols reveals monolignol coupling conditions in lignifying poplar xylem. Plant Physiol 136:3537–3549
Murakana T, Miyata M, Ito K, Tachibana S (1998) Production of podophyllotoxin in Juniperus chinensis callus cultures treated with oligosaccharides and a biogenetic precursor. Phytochemistry 49:491–496
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Negrel J, Javelle F (1995) Induction of phenylpropanoid and tyramine metabolism in pectinase- or pronase-elicited cell suspension cultures of tobacco (Nicotiana tabacum). Physiol Plant 95:569–574
Olivain C, Trouvelot S, Binet M-N, Cordier C, Pugin A, Alabouvette C (2003) Colonization of flax roots and early physiological responses of flax cells inoculated with pathogenic and nonpathogenic strains of Fusarium oxysporum. Appl Environ Microbiol 69:5453–5462
Piquemal J, Lapierre C, Myton K, O’Connell A, Schuch W, Grima-Pettenati J, Boudet AM (1998) Down-regulation of cinnamoyl-CoA reductase induces significant changes of lignin profiles in transgenic tobacco plants. Plant J 13:71–83
Raes J, Rohde A, Christensen JH, Van de Peer Y, Boerjan W (2003) Genome-wide characterization of the lignification toolbox in Arabidopsis. Plant Physiol 133:1051–1071
Schmidt J, Petersen M (2002) Pinoresinol and matairesinol accumulation in a Forsythia x intermedia cell suspension culture. Plant Cell Tiss Org Cult 68:91–98
Somssich IE, Wernert P, Kiedrowski S, Hahlbrock K (1996) Arabidopsis thaliana defense-related protein ELI3 is an aromatic alcohol:NADP(+) oxidoreductase. Proc Natl Acad Sci USA 93:14199–14203
Stewart D, Yahiaoui N, McDougall GJ, Myton K, Marque C, Boudet AM, Haigh J (1997) Fourier-transform infrared and raman spectroscopic evidence for the incorporation of cinnamaldehydes into the lignin of transgenic tobacco (Nicotiana tabacum L.) plants with reduced expression of cinnamyl alcohol dehydrogenase. Planta 201:311–318
Sugimoto K, Takeda S, Hirochika H (2000) MYB-related transcription factor NtMYB2 induced by wounding and elicitors is a regulator of the tobacco retrotransposon Tto1 and defense-related genes. Plant Cell 12:2511–2527
Suzuki H, Reddy MSS, Naoumkina M, Aziz N, May GD, Huhman DV, Summer LW, Blount JW, Mendes P, Dixon RA (2004) Methyljasmonate and yeast elicitor induce differential transcriptional and metabolic reprogramming in cell suspension cultures of the model legume Medicago sativa. Planta 220:696–707
Tokunaga N, Sakakibara N, Umezawa T, Ito Y, Fukuda H, Sato Y (2005) Involvement of extracellular dilignols in lignification during tracheary element differentiation of isolated Zinnia mesophyll cells. Plant Cell Physiol 46:224–232
Vander Mijnsbrugge K, Beeckman H, De Rycke R, Van Montagu M, Engler G, Boerjan W (2000) Phenylcoumaran benzylic ether reductase, a prominent poplar xylem protein, is strongly associated with phenylpropanoid biosynthesis in lignifying cells. Planta 211:502–509
Van Uden W, Pras N, Vossebeld EM, Mol JNM, Malingre TM (1990) Production of 5-methoxypodophyllotoxin in cell suspension cultures of Linum flavum L. Plant Cell Tiss Org Cult 20:81–87
Vardapetyan HR, Kirakosyan AB, Oganesyan AA, Penesyan AR, Alfermann WA (2003) Effect of various elicitors on lignan biosynthesis in callus cultures of Linum austriacum. Russ J Plant Physiol 50:297–300
Wescott ND, Muir AD (2003) Flax seed lignan in disease prevention and health promoting. Phytochem Rev 2:401–417
Zeier J, Schreiber L (1999) Fourier transform infrared-spectroscopic characterisation of isolated endodermal cell walls from plant roots:chemical nature in relation to anatomical development. Planta 209:537–542
Acknowledgments
C. Hano and M. Addi contributed equally to this work. We thank J.-P. Trouvé (coopérative linière Terre de Lin, Fontaine le Dun, France) for gifts of flax seeds cv Barbara, P. exigua and F. oxysporum mycelia, and B. Dehorter (Université des Sciences et Technologies de Lille, France) for giving us B. cinerea mycelium. We thank E. Duverger (Université d’Orléans, France) for epi-fluorescence microscopy. We also wish to thank L. Gutierrez (Université de Picardie Jules Verne, France) for technical advice on RT-PCR experiments. This work was financially supported, in part, by a grant from the “Région Centre” (France) and by the “Conseil Général d’Eure et Loir” (France).
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Hano and M. Addi contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Hano, C., Addi, M., Bensaddek, L. et al. Differential accumulation of monolignol-derived compounds in elicited flax (Linum usitatissimum) cell suspension cultures. Planta 223, 975–989 (2006). https://doi.org/10.1007/s00425-005-0156-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-005-0156-1