Abstract
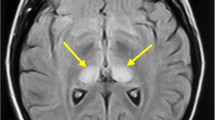
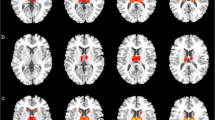
Pruritus, a common feature of animal prion diseases such as scrapie, is rarely reported in humans with Creutzfeldt–Jakob disease (CJD), and its anatomical background is not well defined. The present study was undertaken to carry out a methodical prospective search for the prevalence of pruritus in CJD patients and investigate its anatomical substrate by MRI. The study group included consecutive familial and sporadic CJD patients carrying the E200K PRNP mutation followed up in a longitudinal prospective study between the years 2005 and 2008. Pruritus was prospectively screened for and diffusion-weighted imaging (DWI) was used to correlate brain diffusion abnormalities with pruritus in CJD patients. Pruritus was present in 6/31 (19.35%) patients with familial disease (fCJD) and in none of the patients with sporadic disease (sCJD). Pruritus was a presenting symptom in one patient and evolved during the course of the disease in the other five patients. The pruritus was generalized in three patients, regional in two and localized in one patient. It was transient in one patient and continued throughout the disease in five patients. DWI showed that pruritus was significantly associated with reduced diffusion in the several areas known to be affected by CJD, but most significantly in the midbrain periaqueductal grey matter. Pruritus is relatively common in patients with familial CJD carrying the E200K mutation. Our findings point to a central origin that involves damage to the inhibitory gating mechanism for itch in the periaqueductal grey matter.

Similar content being viewed by others
References
Prusiner SB (1991) Molecular biology of prion diseases. Science 252:1515–1522
Eggenberger E (2007) Prion disease. Neurol Clin 25:833–842 viii
Knight RS, Will RG (2004) Prion diseases. J Neurol Neurosurg Psychiatry 75(Suppl 1):i36–i42
Goldfarb LG, Korczyn AD, Brown P, Chapman J, Gajdusek DC (1990) Mutation in codon 200 of scrapie amyloid precursor gene linked to Creutzfeldt–Jakob disease in Sephardic Jews of Libyan and non-Libyan origin. Lancet 336:637–638
Hsiao K, Meiner Z, Kahana E et al (1991) Mutation of the prion protein in Libyan Jews with Creutzfeldt–Jakob disease. N Engl J Med 324:1091–1097
Petrie L, Heath B, Harold D (1989) Scrapie: report of an outbreak and brief review. Can Vet J 30:321–327
Wilesmith JW, Wells GA, Cranwell MP, Ryan JB (1988) Bovine spongiform encephalopathy: epidemiological studies. Vet Rec 123:638–644
Shabtai H, Nisipeanu P, Chapman J, Korczyn AD (1996) Pruritus in Creutzfeldt-Jakob disease. Neurology 46:940–941
Chapman J, Brown P, Goldfarb LG, Arlazoroff A, Gajdusek DC, Korczyn AD (1993) Clinical heterogeneity and unusual presentations of Creutzfeldt–Jakob disease in Jewish patients with the PRNP codon 200 mutation. J Neurol Neurosurg Psychiatry 56:1109–1112
Young GS, Geschwind MD, Fischbein NJ et al (2005) Diffusion-weighted and fluid-attenuated inversion recovery imaging in Creutzfeldt–Jakob disease: high sensitivity and specificity for diagnosis. AJNR Am J Neuroradiol 26:1551–1562
Lee H, Rosenmann H, Chapman J et al (2009) Thalamo-striatal diffusion reductions precede disease onset in prion mutation carriers. Brain 132:2680–2687
World Health Organization (1998) Human transmissible spongiform encephalopathies. Weekly Epidemiological Record 73:361–365
Fulbright RK, Hoffmann C, Lee H, Pozamantir A, Chapman J, Prohovnik I (2008) MR imaging of familial Creutzfeldt–Jakob disease: a blinded and controlled study. AJNR Am J Neuroradiol 29:1638–1643
Cyngiser TA (2008) Creutzfeldt–Jakob disease: a disease overview. Am J Electroneurodiagnos Technol 48:199–208
Skinningsrud A, Stenset V, Gundersen AS, Fladby T (2008) Cerebrospinal fluid markers in Creutzfeldt–Jakob disease. Cerebrospinal Fluid Res 5:14
Cohen OS, Hoffmann C, Lee H, Chapman J, Fulbright RK, Prohovnik I (2009) MRI detection of the cerebellar syndrome in Creutzfeldt–Jakob disease. Cerebellum 8:373–381
Meiner Z, Gabizon R, Prusiner SB (1997) Familial Creutzfeldt–Jakob disease Codon 200 prion disease in Libyan Jews. Medicine (Baltimore) 76:227–237
Gabizon R, Rosenmann H, Meiner Z, Kahana I, Kahana E, Shugart Y, Ott J, Prusiner SB (1993) Mutation and polymorphism of the prion protein gene in Libyan Jews with Creutzfeldt–Jakob disease (CJD). Am J Hum Genet 53(4):828–835
Ikoma A, Rukwied R, Stander S, Steinhoff M, Miyachi Y, Schmelz M (2003) Neurophysiology of pruritus: interaction of itch and pain. Arch Dermatol 139:1475–1478
Schmelz M, Schmidt R, Weidner C, Hilliges M, Torebjork HE, Handwerker HO (2003) Chemical response pattern of different classes of C-nociceptors to pruritogens and algogens. J Neurophysiol 89:2441–2448
Andrew D, Craig AD (2001) Spinothalamic lamina I neurons selectively sensitive to histamine: a central neural pathway for itch. Nat Neurosci 4:72–77
Schoene WC, Masters CL, Gibbs CJ Jr et al (1981) Transmissible spongiform encephalopathy (Creutzfeldt–Jakob disease). Atypical clinical and pathological findings. Arch Neurol 38:473–477
Meiner Z, Halimi M, Polakiewicz RD, Prusiner SB, Gabizon R (1992) Presence of prion protein in peripheral tissues of Libyan Jews with Creutzfeldt–Jakob disease. Neurology 42:1355–1360
Thomzig A, Schulz-Schaeffer W, Wrede A et al (2007) Accumulation of pathological prion protein PrPSc in the skin of animals with experimental and natural scrapie. PLoS Pathog 3:e66
Notari S, Moleres FJ, Hunter SB et al (2010) Multiorgan detection and characterization of protease resistant prion protein in a case of variant CJD examined in the United States. PLoS One 19(5):e8765
Yosipovitch G, Samuel LS (2008) Neuropathic and psychogenic itch. Dermatol Ther 21:32–41
Groschup MH, Beekes M, McBride PA, Hardt M, Hainfellner JA, Budka H (1999) Deposition of disease-associated prion protein involves the peripheral nervous system in experimental scrapie. Acta Neuropathol 98:453–457
McBride PA, Schulz-Schaeffer WJ, Donaldson M et al (2001) Early spread of scrapie from the gastrointestinal tract to the central nervous system involves autonomic fibers of the splanchnic and vagus nerves. J Virol 75:9320–9327
Beekes M, McBride PA (2007) The spread of prions through the body in naturally acquired transmissible spongiform encephalopathies. FEBS J 274:588–605
Koeppel MC, Bramont C, Ceccaldi M, Habib M, Sayag J (1993) Paroxysmal pruritus and multiple sclerosis. Br J Dermatol 129:597–598
Massey EW (1984) unilateral neurogenic pruritus following stroke. Stroke 15:901–903
Andreev VC, Petkov I, Berova N, Mustakov G (1975) Isolated lymphogranulomatosis of the skin. Dermatol Monatsschr 161:209–214
Sullivan MJ, Drake ME Jr (1984) Unilateral pruritus and Nocardia brain abscess. Neurology 34:828–829
King CA, Huff FJ, Jorizzo JL (1982) Unilateral neurogenic pruritus: paroxysmal itching associated with central nervous system lesions. Ann Intern Med 97:222–223
Kimyai-Asadi A, Nousari HC, Kimyai-Asadi T, Milani F (1999) Poststroke pruritus. Stroke 30:692–693
Shapiro PE, Braun CW (1987) Unilateral pruritus after a stroke. Arch Dermatol 123:1527–1530
Yamamoto M, Yabuki S, Hayabara T, Otsuki S (1981) Paroxysmal itching in multiple sclerosis: a report of three cases. J Neurol Neurosurg Psychiatry 44:19–22
Mochizuki H, Tashiro M, Kano M, Sakurada Y, Itoh M, Yanai K (2003) Imaging of central itch modulation in the human brain using positron emission tomography. Pain 105:339–346
Mochizuki H, Sadato N, Saito DN et al (2007) Neural correlates of perceptual difference between itching and pain: a human fMRI study. Neuroimage 36:706–717
Herde L, Forster C, Strupf M, Handwerker HO (2007) Itch induced by a novel method leads to limbic deactivations a functional MRI study. J Neurophysiol 98:2347–2356
Valet M, Pfab F, Sprenger T et al (2008) cerebral processing of histamine-induced itch using short-term alternating temperature modulation—an FMRI study. J Invest Dermatol 128:426–433
Leknes SG, Bantick S, Willis CM, Wilkinson JD, Wise RG, Tracey I (2007) Itch and motivation to scratch: an investigation of the central and peripheral correlates of allergen- and histamine-induced itch in humans. J Neurophysiol 97:415–422
Chapman J, Cohen OS, Nitzan Z, Tsabari R, Prohovnik I (2007) The CJD rating scale: a new tool for evaluation of neurologic symptomatology in early CJD patients. Neurology 68(Suppl 1):A141
Acknowledgment
The study was supported by NIH grant NS043488.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cohen, O.S., Chapman, J., Lee, H. et al. Pruritus in familial Creutzfeldt–Jakob disease: a common symptom associated with central nervous system pathology. J Neurol 258, 89–95 (2011). https://doi.org/10.1007/s00415-010-5694-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-010-5694-1