Abstract
The study of depriving cells from background ionizing radiation for the past decades has provided valuable insights into its role in cellular homeostasis control. To explore the existence of such response in eukaryotic cells, we grew Chinese hamster (Cricetulus griseus) V79 cells for 23 days using three different dose rates: 0.91 (below background), 35 (surface control) and 72 nGy h−1 (underground KCl-amended control). We did not observe a significant difference in cell number during the course of the experiment. However, cells grown at below background showed significantly lower viability compared to those grown at both control levels after 5 days of incubation and lasted, intermittently, for up to 21 days. We also observed a clear differentiation between the underground and the surface controls that could be explained by the variety of radiation sources present during cell growth under unshielded conditions. To explore the molecular mechanisms for these responses we performed transcriptome analysis on samples collected on days 2 and 5, but only samples from day 5 resulted in significant regulation. Gene enrichment analysis revealed two control-dependent general transcriptional responses. When compared the underground-KCl control, below-background cells showed the upregulation of processes intended for the response to drugs, metals and mechanical stimuli. In comparison, the response relative to the surface control was characterized by the upregulation of responses to organic substances and abiotic stimuli involved in the regulation of signaling, as well as to cell proliferation and homeostatic control of the number of cell processes.




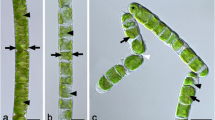
source background ionizing radiation control (c, d)
Similar content being viewed by others
References
Barzilai A, Rotman G, Shiloh Y (2002) ATM deficiency and oxidative stress: a new dimension of defective response to DNA damage. DNA Repair 1:3–25
Best A, Gorres J, Junker M, Kratz-Ludwig K, Laubenstein M, Long A, Nisi S, Smith K, Wiescher M (2016) Low energy neutron background in deep underground laboratories. Nucl Instr Meth a 812:1–6
Caltagirone A, Weiss G, Pantopoulos K (2001) Modulaton of cellular iron metabolism by hydrogen peroxide. J Biol Chem 276:19738–19745
Carbone MC, Pinto M, Antonelli F, Amicarelli F, Balata M, Belli M et al (2009) The cosmic silence experiment: on the putative adaptive role of environmental ionizing radiation. Radiat Environ Biophys 48:189–196
Castillo H, Smith G (2017) Below background ionizing radiation as an environmental cue for bacteria. Front Microbiol 8:1–7
Castillo H, Schoderbek D, Dulal S, Escobar G, Wood J, Nelson R et al (2015) Stress induction in the bacteria Shewanella oneidensis and Deinococcus radiodurans in response to below-background ionizing radiation. Int J Radiat Biol 91:749–756
Castillo H, Li X, Schilkey F, Smith GB (2018) Transcriptome analysis reveals a stress response of Shewanella oneidensis deprived of background levels of ionizing radiation. PLoS ONE 13(5):e0196472
Castillo H, Li X, Smith G (2021) Deinococcus radiodurans UWO298 dependence on background radiation for optimal growth. Front Genet 12:644292. https://doi.org/10.3389/fgene.2021.644292
Chiou HC, Hayes R (2004) Creating and baseline radiological standard for the waste isolation pilot plant underground. Isotopes Environ Health Stud 40:213–220
Choi TG, Lee J, Ha J, Kim SS (2011) Apoptosis signal-regulating kinase 1 is an intracellular inducer of p38 MAPK-mediated myogenic signaling in cardiac myoblasts. Biochem Biophys Acta 1813:1412–1421
Esch E-I, Bowles TJ, Hime A. Pichlmaier A, Reifarth R, Wollnik H. (2005). The cosmic ray muon flux at WIPP. Nucl. Instr. Meth. A. 538, 516-525
Fratini E, Carbone C, Capece D, Esposito G, Simone G, Tabocchini MA, et al. (2015) Low-radiation environment affects the development of protection mechanisms in V79 cells. Radiat Environ Biophys 54: 183±194.
International Commission on Radiological Protection (ICRP) (2007) Recommendations of the international commission on radiological protection. Elsevier, New York
Liu, J., Ma, T., Gao, M. et al. (2020). Proteomics provides insights into the inhibition of Chinese hamster V79 cell proliferation in the deep underground environment. Sci Rep 10, 14921 (2020). https://doi.org/10.1038/s41598-020-71154-z
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 25:402–408
Orino K, Lehman L, Tsuji Y, Ayaki H, Torti SV, Torti FM (2001) Ferritin and the response to oxidative stress. Biochem J 357:241–247
Planel H, Soleilhavoup JP, Tizador R, Richoilley G, Conter A, Croute F, et al. (1987) Influence on cell proliferation of background radiation or exposure to very low, chronic γ radiation Health Phys. 52: 571–578.
Riley PA (1994) Free radicals in biology: oxidative stress and the effects of ionizing radiation. Int J Radiat Biol 65:27–33
Satta L, Antonelli F, Belli M, Sapora O, Simone G, Sorrentino E et al (2002) Influence of a low background radiation environment on biochemical and biological responses in V79 cells. Radiat Environ Biophys 41:217–224
Thorne MC (2003) Background radiation: natural and man-made. J Radiol Prot 23:29–42
Acknowledgements
The authors thank Shawn Otto, Brian Dozier and Douglas Weaver from Los Alamos National Laboratory for their support to do work at the Waste Isolation Pilot Plant WIPP) underground.
Funding
These results are based upon work supported by the U.S. Department of Energy (DOE) Office of Environmental Management under award number DE-EM0002423. This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government or any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe on privately owned rights. Reference herein to any specific commercial product, process or service by trade names, trademark, manufacturer or otherwise does not necessarily constitute or imply its endorsement. The views and the opinions of the authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
Author information
Authors and Affiliations
Contributions
GS and HC designed and executed the field portion of the experiment; HC and JW executed the laboratory experiments for the transcriptome analysis and its validation; GS and HC analyzed the data; HC wrote the manuscript with editing assistance from GS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no known conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Castillo, H., Winder, J. & Smith, G. Chinese hamster V79 cells’ dependence on background ionizing radiation for optimal growth. Radiat Environ Biophys 61, 49–57 (2022). https://doi.org/10.1007/s00411-021-00951-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-021-00951-5