Abstract
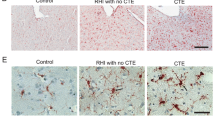
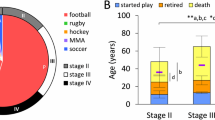
Chronic traumatic encephalopathy (CTE) is a tauopathy associated with repetitive head impacts (RHI) that has been neuropathologically diagnosed in American football players and other contact sport athletes. In 2013, McKee and colleagues proposed a staging scheme for characterizing the severity of the hyperphosphorylated tau (p-tau) pathology, the McKee CTE staging scheme. The staging scheme defined four pathological stages of CTE, stages I(mild)–IV(severe), based on the density and regional deposition of p-tau. The objective of this study was to test the utility of the McKee CTE staging scheme, and provide a detailed examination of the regional distribution of p-tau in CTE. We examined the relationship between the McKee CTE staging scheme and semi-quantitative and quantitative assessments of regional p-tau pathology, age at death, dementia, and years of American football play among 366 male brain donors neuropathologically diagnosed with CTE (mean age 61.86, SD 18.90). Spearman’s rho correlations showed that higher CTE stage was associated with higher scores on all semi-quantitative and quantitative assessments of p-tau severity and density (p’s < 0.001). The severity and distribution of CTE p-tau followed an age-dependent progression: older age was associated with increased odds for having a higher CTE stage (p < 0.001). CTE stage was independently associated with increased odds for dementia (p < 0.001). K-medoids cluster analysis of the semi-quantitative scales of p-tau across 14 regions identified 5 clusters of p-tau that conformed to increasing CTE stage (stage IV had 2 slightly different clusters), age at death, dementia, and years of American football play. There was a predilection for p-tau pathology in five regions: dorsolateral frontal cortex (DLF), superior temporal cortex, entorhinal cortex, amygdala, and locus coeruleus (LC), with CTE in the youngest brain donors and lowest CTE stage restricted to DLF and LC. These findings support the usefulness of the McKee CTE staging scheme and demonstrate the regional distribution of p-tau in CTE.






Similar content being viewed by others
References
Alosco ML, Mez J, Tripodis Y, Kiernan PT, Abdolmohammadi B, Murphy L et al (2018) Age of first exposure to tackle football and chronic traumatic encephalopathy. Ann Neurol 83:886–901. https://doi.org/10.1002/ana.25245
Alosco ML, Stein TD, Tripodis Y, Chua AS, Kowall NW, Huber BR et al (2019) Association of white matter rarefaction, arteriolosclerosis, and tau with dementia in chronic traumatic encephalopathy. JAMA Neurol. https://doi.org/10.1001/jamaneurol.2019.2244
Armstrong RA, McKee AC, Stein TD, Alvarez VE, Cairns NJ (2019) Cortical degeneration in chronic traumatic encephalopathy and Alzheimer's disease neuropathologic change. Neurol Sci 40:529–533. https://doi.org/10.1007/s10072-018-3686-6
Bieniek KF, Blessing MM, Heckman MG, Diehl NN, Serie AM, Paolini MA 2nd et al (2020) Association between contact sports participation and chronic traumatic encephalopathy: a retrospective cohort study. Brain Pathol 30:63–74. https://doi.org/10.1111/bpa.12757
Bieniek KF, Ross OA, Cormier KA, Walton RL, Soto-Ortolaza A, Johnston AE et al (2015) Chronic traumatic encephalopathy pathology in a neurodegenerative disorders brain bank. Acta Neuropathol 130:877–889. https://doi.org/10.1007/s00401-015-1502-4
Bigio EH (2008) Update on recent molecular and genetic advances in frontotemporal lobar degeneration. J Neuropathol Exp Neurol 67:635–648. https://doi.org/10.1097/NEN.0b013e31817d751c
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259. https://doi.org/10.1007/BF00308809
Braak H, Braak E (1995) Staging of Alzheimer's disease-related neurofibrillary changes. Neurobiol Aging 16:271–278. https://doi.org/10.1016/0197-4580(95)00021-6
Braak H, Del Tredici K (2011) The pathological process underlying Alzheimer's disease in individuals under thirty. Acta Neuropathol 121:171–181. https://doi.org/10.1007/s00401-010-0789-4
Brownell B, Oppenheimer DR, Hughes JT (1970) The central nervous system in motor neurone disease. J Neurol Neurosurg Psychiatry 33:338–357. https://doi.org/10.1136/jnnp.33.3.338
Cairns NJ, Neumann M, Bigio EH, Holm IE, Troost D, Hatanpaa KJ et al (2007) TDP-43 in familial and sporadic frontotemporal lobar degeneration with ubiquitin inclusions. Am J Pathol 171:227–240. https://doi.org/10.2353/ajpath.2007.070182
Cherry JD, Kim SH, Stein TD, Pothast MJ, Nicks R, Meng G et al (2020) Evolution of neuronal and glial tau isoforms in chronic traumatic encephalopathy. Brain Pathol. https://doi.org/10.1111/bpa.12867
Cherry JD, Tripodis Y, Alvarez VE, Huber B, Kiernan PT, Daneshvar DH et al (2016) Microglial neuroinflammation contributes to tau accumulation in chronic traumatic encephalopathy. Acta Neuropathol Commun 4:112. https://doi.org/10.1186/s40478-016-0382-8
Corsellis JA, Bruton CJ, Freeman-Browne D (1973) The aftermath of boxing. Psychol Med 3:270–303. https://doi.org/10.1017/s0033291700049588
de Miranda AS, Zhang CJ, Katsumoto A, Teixeira AL (2017) Hippocampal adult neurogenesis: does the immune system matter? J Neurol Sci 372:482–495. https://doi.org/10.1016/j.jns.2016.10.052
Dickson DW (2009) Neuropathology of non-Alzheimer degenerative disorders. Int J Clin Exp Pathol 3:1–23
Ehrenberg AJ, Suemoto CK, Franca Resende EP, Petersen C, Leite REP, Rodriguez RD et al (2018) Neuropathologic correlates of psychiatric symptoms in Alzheimer's disease. J Alzheimers Dis 66:115–126. https://doi.org/10.3233/JAD-180688
Erdo F, Denes L, de Lange E (2017) Age-associated physiological and pathological changes at the blood-brain barrier: a review. J Cereb Blood Flow Metab 37:4–24. https://doi.org/10.1177/0271678X16679420
Eser RA, Ehrenberg AJ, Petersen C, Dunlop S, Mejia MB, Suemoto CK et al (2018) Selective vulnerability of brainstem nuclei in distinct tauopathies: a postmortem study. J Neuropathol Exp Neurol 77:149–161. https://doi.org/10.1093/jnen/nlx113
Falcon B, Zivanov J, Zhang W, Murzin AG, Garringer HJ, Vidal R et al (2019) Novel tau filament fold in chronic traumatic encephalopathy encloses hydrophobic molecules. Nature 568:420–423. https://doi.org/10.1038/s41586-019-1026-5
Forrest SL, Kril JJ, Wagner S, Honigschnabl S, Reiner A, Fischer P et al (2019) Chronic traumatic encephalopathy (CTE) is absent from a european community-based aging cohort while cortical aging-related tau astrogliopathy (ARTAG) is highly prevalent. J Neuropathol Exp Neurol 78:398–405. https://doi.org/10.1093/jnen/nlz017
Haneuse S, Schildcrout J, Crane P, Sonnen J, Breitner J, Larson E (2009) Adjustment for selection bias in observational studies with application to the analysis of autopsy data. Neuroepidemiology 32:229–239. https://doi.org/10.1159/000197389
Iverson GL, Luoto TM, Karhunen PJ, Castellani RJ (2019) Mild chronic traumatic encephalopathy neuropathology in people with no known participation in contact sports or history of repetitive neurotrauma. J Neuropathol Exp Neurol 78:615–625. https://doi.org/10.1093/jnen/nlz045
James BD, Wilson RS, Boyle PA, Trojanowski JQ, Bennett DA, Schneider JA (2016) TDP-43 stage, mixed pathologies, and clinical Alzheimer's-type dementia. Brain 139:2983–2993. https://doi.org/10.1093/brain/aww224
Jellinger KA, Korczyn AD (2018) Are dementia with Lewy bodies and Parkinson's disease dementia the same disease? BMC Med 16:34. https://doi.org/10.1186/s12916-018-1016-8
Kress BT, Iliff JJ, **a M, Wang M, Wei HS, Zeppenfeld D et al (2014) Impairment of paravascular clearance pathways in the aging brain. Ann Neurol 76:845–861. https://doi.org/10.1002/ana.24271
Lee EB, Kinch K, Johnson VE, Trojanowski JQ, Smith DH, Stewart W (2019) Chronic traumatic encephalopathy is a common co-morbidity, but less frequent primary dementia in former soccer and rugby players. Acta Neuropathol 138:389–399. https://doi.org/10.1007/s00401-019-02030-y
Ling H, Holton JL, Shaw K, Davey K, Lashley T, Revesz T (2015) Histological evidence of chronic traumatic encephalopathy in a large series of neurodegenerative diseases. Acta Neuropathol 130:891–893. https://doi.org/10.1007/s00401-015-1496-y
Ling H, Morris HR, Neal JW, Lees AJ, Hardy J, Holton JL et al (2017) Mixed pathologies including chronic traumatic encephalopathy account for dementia in retired association football (soccer) players. Acta Neuropathol 133:337–352. https://doi.org/10.1007/s00401-017-1680-3
Litvan I, Hauw JJ, Bartko JJ, Lantos PL, Daniel SE, Horoupian DS et al (1996) Validity and reliability of the preliminary NINDS neuropathologic criteria for progressive supranuclear palsy and related disorders. J Neuropathol Exp Neurol 55:97–105. https://doi.org/10.1097/00005072-199601000-00010
Mackenzie IR, Neumann M, Bigio EH, Cairns NJ, Alafuzoff I, Kril J et al (2010) Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathol 119:1–4. https://doi.org/10.1007/s00401-009-0612-2
McKee AC, Cairns NJ, Dickson DW, Folkerth RD, Keene CD, Litvan I et al (2016) The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol 131:75–86. https://doi.org/10.1007/s00401-015-1515-z
McKee AC, Cantu RC, Nowinski CJ, Hedley-Whyte ET, Gavett BE, Budson AE et al (2009) Chronic traumatic encephalopathy in athletes: progressive tauopathy after repetitive head injury. J Neuropathol Exp Neurol 68:709–735. https://doi.org/10.1097/NEN.0b013e3181a9d503
McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, Daneshvar DH et al (2013) The spectrum of disease in chronic traumatic encephalopathy. Brain 136:43–64. https://doi.org/10.1093/brain/aws307
McKeith IG (2006) Consensus guidelines for the clinical and pathologic diagnosis of dementia with Lewy bodies (DLB): report of the Consortium on DLB International Workshop. J Alzheimers Dis 9:417–423. https://doi.org/10.3233/jad-2006-9s347
Mez J, Daneshvar DH, Abdolmohammadi B, Chua AS, Alosco ML, Kiernan PT et al (2020) Duration of American football play and chronic traumatic encephalopathy. Ann Neurol 87:116–131. https://doi.org/10.1002/ana.25611
Mez J, Daneshvar DH, Kiernan PT, Abdolmohammadi B, Alvarez VE, Huber BR et al (2017) Clinicopathological evaluation of chronic traumatic encephalopathy in players of American football. JAMA 318:360–370. https://doi.org/10.1001/jama.2017.8334
Mez J, Solomon TM, Daneshvar DH, Murphy L, Kiernan PT, Montenigro PH et al (2015) Assessing clinicopathological correlation in chronic traumatic encephalopathy: rationale and methods for the UNITE study. Alzheimers Res Ther 7:62. https://doi.org/10.1186/s13195-015-0148-8
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW et al (2012) National Institute on Aging-Alzheimer's Association guidelines for the neuropathologic assessment of Alzheimer's disease: a practical approach. Acta Neuropathol 123:1–11. https://doi.org/10.1007/s00401-011-0910-3
Mooradian AD (1988) Effect of aging on the blood-brain barrier. Neurobiol Aging 9:31–39. https://doi.org/10.1016/s0197-4580(88)80013-7
Newell KL, Hyman BT, Growdon JH, Hedley-Whyte ET (1999) Application of the National Institute on Aging (NIA)-Reagan Institute criteria for the neuropathological diagnosis of Alzheimer disease. J Neuropathol Exp Neurol 58:1147–1155. https://doi.org/10.1097/00005072-199911000-00004
Serrano-Pozo A, Qian J, Muzikansky A, Monsell SE, Montine TJ, Frosch MP et al (2016) Thal amyloid stages do not significantly impact the correlation between neuropathological change and cognition in the Alzheimer disease continuum. J Neuropathol Exp Neurol 75:516–526. https://doi.org/10.1093/jnen/nlw026
Stein TD, Montenigro PH, Alvarez VE, **a W, Crary JF, Tripodis Y et al (2015) Beta-amyloid deposition in chronic traumatic encephalopathy. Acta Neuropathol 130:21–34. https://doi.org/10.1007/s00401-015-1435-y
Stern RA, Daneshvar DH, Baugh CM, Seichepine DR, Montenigro PH, Riley DO et al (2013) Clinical presentation of chronic traumatic encephalopathy. Neurology 81:1122–1129. https://doi.org/10.1212/WNL.0b013e3182a55f7f
Tibshirani R, Walther G, Hastie T (2001) Estimating the number of clusters in a data set via the gap statistic. J R Stat Soc Ser B (Stat Methodol) 63:411–423
Udeochu JC, Shea JM, Villeda SA (2016) Microglia communication: Parallels between aging and Alzheimer's disease. Clin Exp Neuroimmunol 7:114–125. https://doi.org/10.1111/cen3.12307
Vonsattel JP, Aizawa H, Ge P, DiFiglia M, McKee AC, MacDonald M et al (1995) An improved approach to prepare human brains for research. J Neuropathol Exp Neurol 54:42–56. https://doi.org/10.1097/00005072-199501000-00006
Vonsattel JP, Del Amaya MP, Keller CE (2008) Twenty-first century brain banking. Processing brains for research: the Columbia University methods. Acta Neuropathol 115:509–532. https://doi.org/10.1007/s00401-007-0311-9
Wilson RS, Nag S, Boyle PA, Hizel LP, Yu L, Buchman AS et al (2013) Brainstem aminergic nuclei and late-life depressive symptoms. JAMA Psychiatry 70:1320–1328. https://doi.org/10.1001/jamapsychiatry.2013.2224
Woerman AL, Aoyagi A, Patel S, Kazmi SA, Lobach I, Grinberg LT et al (2016) Tau prions from Alzheimer's disease and chronic traumatic encephalopathy patients propagate in cultured cells. Proc Natl Acad Sci USA 113:E8187–E8196. https://doi.org/10.1073/pnas.1616344113
**ng CY, Tarumi T, Liu J, Zhang Y, Turner M, Riley J et al (2017) Distribution of cardiac output to the brain across the adult lifespan. J Cereb Blood Flow Metab 37:2848–2856. https://doi.org/10.1177/0271678X16676826
Acknowledgements
We would like to acknowledge the many families who contributed to this work, Christopher Nowinski, Ph.D. and Lisa McHale from the Concussion Legacy Foundation, and the clinical and neuropathology research staff of the BU CTE Center, VA Boston Healthcare System and Edith Nourse Rogers VA Medical Center This work was supported by grant funding from: NIA (AG057902, AG06234, RF1AG054156, K23AG046377), NINDS (U54NS115266, U01NS086659, and K23NS102399), National Institute of Aging Boston University AD Center (P30AG13846; supplement 0572063345-5); Department of Veterans Affairs Merit Award (I01-CX001038), the Nick and Lynn Buoniconti Foundation, and BU-CTSI Grant Number 1UL1TR001430. JC is funded by the Alzheimer’s Association (AARF-17-529888). The views, opinions, and/or findings contained in this article are those of the authors and should not be construed as an official Veterans Affairs or Department of Defense position, policy, or decision, unless so designated by other official documentation. Funders did not have a role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alosco, M.L., Cherry, J.D., Huber, B.R. et al. Characterizing tau deposition in chronic traumatic encephalopathy (CTE): utility of the McKee CTE staging scheme. Acta Neuropathol 140, 495–512 (2020). https://doi.org/10.1007/s00401-020-02197-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-020-02197-9