Abstract
Purpose
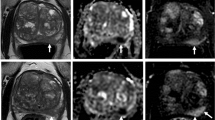
To investigate the performance of pre-surgery CT and multiparametric MRI (mpMRI) to identify lymph node (LN) metastases in the Michigan Urological Surgery Improvement Collaborative (MUSIC). Abdominopelvic CT and mpMRI are commonly used for intermediate- and high-risk prostate cancer (PCa) staging.
Methods
Retrospective analysis of the MUSIC registry identified patients undergoing robot-assisted radical prostatectomy (RP) between 3/2012 and 7/2018. Patients were classified according to pre-surgery imaging modality. Primary outcomes were operating characteristics of CT and mpMRI for detection of pathologic LN involvement (pN1).
Results
A total of 10,250 patients underwent RP and 3924 patients (38.3%) underwent CT and/or mpMRI prior to surgery. Suspicion for LN involvement was identified on 2.3% CT and 1.9% mpMRI. Overall, 391 patients were pN1(3.8%), including 0.1% low-, 2.1% intermediate-, and 10.9% high-risk PCa patients. Of 235 pN1 patients that underwent CT prior, far more had negative (91.1%) than positive (8.9%) findings, yielding sensitivity: 8.9%, specificity: 98.3%, negative predictive value (NPV): 92.1%, and positive predictive value (PPV): 32.3% for CT with regard to LN metastases. Similarly, more patients with pN1 disease had negative mpMRI (81.0%) then suspicious or indeterminate MRI (19.0%), yielding sensitivity: 19.0%, specificity: 97.3%, NPV: 95.9%, and PPV: 26.7%.
Conclusions
Abdominopelvic CT and mpMRI have clear limitations in identifying LN metastases. Additional clinicopathologic features should be considered when making management decisions, as 2.1% and 10.9% with intermediate-and high-risk cancer had metastatic LNs. The majority of pN1 patients had a negative CT or a negative/indeterminate mpMRI prior to RP. Pelvic LN dissection should be performed in RP patients with intermediate- or high-risk PCa, independent of preoperative imaging results.
Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2017) Cancer Statistics, 2017. CA Cancer J Clin 67(1):7–30
Talab SS, Preston MA, Elmi A, Tabatabaei S (2012) Prostate cancer imaging: what the urologist wants to know. Radiol Clin N Am 50(6):1015–1041
Crawford ED, Stone NN, Yu EY et al (2014) Challenges and recommendations for early identification of metastatic disease in prostate cancer. Urology 83(3):664–669
Pinto F, Totaro A, Palermo G et al (2012) Imaging in prostate cancer staging: present role and future perspectives. Urol Int 88(2):125–136
Hovels AM, Heesakkers RA, Adang EM et al (2008) The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol 63(4):387–395
Bayne CE, Williams SB, Cooperberg MR et al (2016) Treatment of the primary tumor in metastatic prostate cancer: current concepts and future perspectives. Eur Urol 69(5):775–787
Husband JE (2002) CT/MRI of nodal metastases in pelvic cancer. Cancer Imaging 2(2):123–129
Budiharto T, Joniau S, Lerut E et al (2011) Prospective evaluation of 11C-choline positron emission tomography/computed tomography and diffusion-weighted magnetic resonance imaging for the nodal staging of prostate cancer with a high risk of lymph node metastases. Eur Urol 60(1):125–130
Kiss B, Thoeny HC, Studer UE (2016) Current Status of Lymph Node Imaging in Bladder and Prostate Cancer. Urology 96:1–7
Calais J, Ceci F, Eiber M et al (2019) (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: a prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol 20:1286
Mansbridge M, Chung E, Rhee H (2019) The use of MRI and PET Imaging studies for prostate cancer management: brief update, clinical recommendations, and technological limitations. Med Sci (Basel) 7:8
Briganti A, Blute ML, Eastham JH et al (2009) Pelvic lymph node dissection in prostate cancer. Eur Urol 55(6):1251–1265
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69(1):16–40
Moschini M, Briganti A, Murphy CR et al (2016) Outcomes for patients with clinical lymphadenopathy treated with radical prostatectomy. Eur Urol 69(2):193–196
Cher ML, Dhir A, Auffenberg GB et al (2017) Appropriateness criteria for active surveillance of prostate cancer. J Urol 197(1):67–74
Risko R, Merdan S, Womble PR et al (2014) Clinical predictors and recommendations for staging computed tomography scan among men with prostate cancer. Urology 84(6):1329–1334
Lecouvet FE, Oprea-Lager DE, Liu Y et al (2018) Use of modern imaging methods to facilitate trials of metastasis-directed therapy for oligometastatic disease in prostate cancer: a consensus recommendation from the EORTC Imaging Group. Lancet Oncol 19(10):e534–e545
Cook GJR, Kulkarni M, Warbey VS (2019) PSMA PET/CT imaging for primary staging of intermediate and high-risk prostate cancer. BJU Int 124(3):357–358
Giganti F, Rosenkrantz AB, Villeirs G et al (2019) The evolution of MRI of the prostate: the past, the present, and the future. Am J Roentgenol. 2019:1–13
Kalapara AA, Nzenza T, Pan HY et al (2019) Detection and localisation of primary prostate cancer using (68) Ga-PSMA PET/CT compared with mpMRI and radical prostatectomy specimens. BJU Int. https://doi.org/10.1111/bju.14858. [Epub ahead of print]
Heidenreich A, Bastian PJ, Bellmunt J et al (2014) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65(1):124–137
Hurley P, Dhir A, Gao Y et al (2017) A statewide intervention improves appropriate imaging in localized prostate cancer. J Urol 197(5):1222–1228
Perez-Lopez R, Tunariu N, Padhani AR et al (2019) Imaging diagnosis and follow-up of advanced prostate cancer: clinical perspectives and state of the art. Radiology 292(2):273–286
Kim SJ, Lee SW (2019) Diagnostic accuracy of (F)18 flucholine PET/CT for preoperative lymph node staging in newly diagnosed prostate cancer patients; a systematic review and meta-analysis. Br J Radiol 92(1101):20190193
Maurer T, Gschwend JE, Rauscher I et al (2016) Diagnostic efficacy of (68)gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol 195(5):1436–1443
Koerber SA, Stach G, Kratochwil C et al (2019) Lymph node involvement in treatment-naive prostate cancer patients: correlation of PSMA-PET/CT imaging and Roach formula in 280 men in the Radiotherapeutic management. J Nucl Med 61:46
Afshar-Oromieh A, Vollnberg B, Alberts I et al (2019) Comparison of PSMA-ligand PET/CT and multiparametric MRI for the detection of recurrent prostate cancer in the pelvis. Eur J Nucl Med Mol Imaging 46(11):2289–2297
Meissner S, Janssen JC, Prasad V et al (2019) Accuracy of standard clinical 3T prostate MRI for pelvic lymph node staging: comparison to (68)Ga-PSMA PET-CT. Sci Rep 9(1):10727
Mottet N, van den Bergh RCN, Briers E et al (2018) EAU—ESTRO—ESUR—SIOG Guidelines on Prostate Cancer 2018. European Association of Urology Guidelines 2018 Edition. Arnhem, The Netherlands: European Association of Urology Guidelines Office
Roach M 3rd, Marquez C, Yuo HS et al (1994) Predicting the risk of lymph node involvement using the pre-treatment prostate specific antigen and Gleason score in men with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys 28(1):33–37
Acknowledgements
We would like to acknowledge the significant contribution of the clinical champions, urologists, and data abstractors in each participating practice. In addition, we would like to acknowledge the support provided by the Value Partnerships program at Blue Cross Blue Shield of Michigan. We would also like to acknowledge the support provided by the Betz Family Endowment for Cancer Research (RG0813-1036). Funding was provided in part by the Spectrum Health Foundation. We also acknowledge Crystal Farrell and Stephen Babitz for critical review of the manuscript and Sabrina Noyes for administrative support.
Author information
Authors and Affiliations
Consortia
Contributions
HP: data collection, manuscript writing. BR Lane: project development, data management, data analysis, manuscript writing/editing. JQ: data analysis. TK: data management, manuscript editing. JEM: project development, manuscript editing. AM: data management, manuscript editing. CMB: project development, data management, manuscript editing. JM: project development; manuscript editing. Michigan Urological Surgery Improvement Collaborative: project development, data collection and management.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest.
Research involving human participants
Each MUSIC practice obtained an exemption or approval for collaborative participation from a local Institutional Review Board.
Informed consent
Not applicable as this is a quality improvement study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Peabody, H., Lane, B.R., Qi, J. et al. Limitations of abdominopelvic CT and multiparametric MR imaging for detection of lymph node metastases prior to radical prostatectomy. World J Urol 39, 779–785 (2021). https://doi.org/10.1007/s00345-020-03227-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-020-03227-7