Abstract
Global warming is leading to range shifts of marine species, threatening the structure and functioning of ecological communities and human populations that rely on them. The largest changes are seen in biogeographic transition zones, such as subtropical reef communities, where species range shifts are already causing substantial community reorganisation. This causes functional changes in communities over subtropical latitudes, though a baseline functional understanding remains elusive for many taxa. One key marine taxon are molluscs, which provide many ecosystem services, are important prey for fishes and are also fisheries targets themselves, but remain largely unstudied. Here, we examine the trait composition, functional diversity, and functional redundancy of mollusc assemblages along the tropical-to-temperate transition in Japan (25° to 35° Northern latitude). Specifically, we use a trait database of 88 mollusc species from 31 subtropical reefs along the Pacific coast of Japan to show that trait composition of mollusc assemblages changes continuously along the latitudinal gradient. We discover that functional diversity of mollusc assemblages decreases with increasing latitude, a pattern associated with declines in functional dispersion. Moreover, we find a clear distinction between tropical and subtropical mollusc assemblages, with substrate-attached, suspension feeding bivalves more abundant in the tropics and free-living gastropod grazers more prevalent at higher latitudes. Our trait-based evidence in this study shows a contraction and almost complete shift in the functioning of marine mollusc assemblages at biogeographic transition zones and our trait database facilitates further study. Our findings provide evidence of the changing taxonomic and functional composition of extant mollusc communities with latitude, pointing to potential pertinent changes and tropicalisation of these communities with rapid ocean warming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The diversity of species and their traits are directly related to ecosystem stability and ecological functioning (Hooper et al. 2005; Hughes et al. 2017). Despite the considerable ecosystem services provided by intact ecological functions, such as nutrient cycling and food provisions (Song et al. 2014), such functions are substantially threatened by anthropogenic pressures, including overexploitation and degradation. In particular, coral reef ecosystems are sentinel systems for global ecosystem collapse (Bland et al. 2017) and are already undergoing irreversible functional shifts (Hughes et al. 2017). Habitat degradation, overexploitation, and changes in climate regimes are causing widespread species and community reorganisations and declines in diversity (Wilson et al. 2010; Vergés et al. 2014). Localised extinctions of native species can lead to the loss of important ecosystem functions such as food provision (Micheli et al. 2014), and are projected for many coral reef ecosystems (Pratchett et al. 2018). By contrast, the alteration or expansion of species ranges into new areas, such as high-latitude reefs, can introduce new functions (e.g. provision of complex habitats by reef-building corals) and potentially disrupt ecosystem structure and functions (Vergés et al. 2014, 2019). Because of this, evaluating changes in ecosystem function with trait-based approaches is an important tool to help understand ecosystem stability (Worm et al. 2006). Trait-based approaches link directly to ecological functions (Mouillot et al. 2013, 2014), and changes in functioning over time can be better understood by considering current functional differences over spatial gradients such as latitude. It is plausible that different taxonomic groups provide different functions in a given ecosystem and that they will vary in their responses to environmental change. Currently, our understanding of functioning on reefs is limited to corals and reef fishes (e.g. Sommer et al. 2014; Mouillot et al. 2014). Considering the large diversity of organisms inhabiting reef assemblages, there is a need to assess the trait-based responses to environmental stressors for other key taxa (Przeslawski et al. 2008).
A comprehensive understanding of functional relationships in different systems and taxa is pivotal to predict and manage the fate of ecosystems. Functional ecology aims to understand the relationship between species diversity and ecological functioning (Micheli et al. 2014; Mouillot et al. 2014) by considering the suite of species traits present in different habitats. Species traits are typically defined as physical, behavioural, biochemical, and phenological characteristics that relate to species fitness (Laureto et al. 2015), and species with similar traits are expected to perform similar roles in ecosystems. Trait-based approaches can therefore help us understand how changes in species interactions and composition can lead to a loss of functioning (Petchey and Gaston 2002; Díaz et al. 2013; Madin et al. 2016). Theoretically, if all functionally significant traits are considered, and sufficient data are collected to accurately characterise them, the functions of an ecosystem can be estimated from the diversity of trait constituents (Violle et al. 2007).
The nature of trait composition and functional diversity can be estimated and explored using a range of summary metrics. Functional diversity (FD) is a measure of the diversity of traits in an ecosystem and has been used to characterise community functional response to disturbance (Mouillot et al. 2013; Rumm et al. 2018), and to evaluate fish community functional response to protected areas (Rincón-Díaz et al. 2018). Other indices are used to describe the distribution and range of traits in theoretical trait space, which itself can be visualised using ordination techniques (Cornwell et al. 2006; McWilliam et al. 2018). The three fundamental metrics used to characterise this space are functional richness, functional evenness, and functional dispersion (Mason et al. 2005; Laliberté and Legendre 2010). They attempt to capture the amount of trait space occupied by the species in a community, the distribution of traits within the trait space, and the variability and position of species trait grou**s in trait space, respectively. In practice, indices are selected in relation to specific research questions (Mouillot et al. 2013) and when combined have been used to identify functional measures of communities. For example, functional redundancy (Hubbell 2005) estimates the frequency of an expressed trait in an ecosystem in relation to the number of species in the habitat. A trait is considered redundant if it is expressed by many species in a community. Theoretically, a redundant function is more robust to species loss as the traits are provided by multiple species (Rosenfeld 2002).
On coral reefs, fish (Mouillot et al. 2014) and coral assemblages (McWilliam et al. 2018) typically show high functional redundancy, except for regions with low coral species richness. Significantly less work has been conducted to understand the relationships between species richness and functional redundancy on subtropical reefs. Species assemblages are generally thought to be more diverse in the tropics and less diverse in temperate regions (Mittelbach et al. 2007; Appeltans et al. 2012) and to vary along environmental gradients, such as a latitudinal gradient (Rivadeneira et al. 2002). However, it is not known if this is reflected in gradients of functional diversity and redundancy along biogeographic transition zones.
Subtropical reefs represent distinct ecosystems at the interface of temperate and tropical climates (Beger et al. 2014; Vergés et al. 2014, 2019). In these regions, highly diverse communities comprise tropical, temperate and endemic species at the limits of their geographic distribution and environmental tolerances (Vroom and Braun 2010). The influence of climate change on the community structure of these marginal ecosystems has already been recorded as regimes of environmental conditions change and disturbance becomes more pronounced (Suthers et al. 2011; Hobday and Lough 2011; Oliver et al. 2018; Kim et al. 2019). At the community level, this leads to migration, high species turnover, population fluctuations (Munday et al. 2008), and localised extinctions of marginal species in response to stochastic stress events (Beger et al. 2014). The poleward expansion of tropical species (Bennett et al. 2015) leads to their introduction to temperate ecosystems, with varied ecosystem level and socioeconomic implications (Madin et al. 2012). In fishes, large size and high mobility along with pelagic spawning indicate a greater potential for range expansion (Feary et al. 2014) whereas environmental tolerances are important in zooxanthellate corals (Mizerek et al. 2016). An example of range expansion comes from Tosa Bay, Japan (~ 33 degrees North), where overgrazing of temperate seaweeds by range-expanding tropical herbivorous fishes has resulted in a shift away from temperate kelp-dominated to coral-dominated systems (Vergés et al. 2014). Traits can also play an important role in determining which species move into higher latitude ecosystems. For example, on subtropical reefs in Australia, species traits such as size and morphology influence the establishment potential of zooxanthellate corals (Sommer et al. 2014; Mizerek et al. 2016) and of tropical fish vagrants on subtropical reefs (Feary et al. 2014). However, trait-mediated responses of other reef taxa are less well-known.
Trait and functional understanding of marine mollusc assemblages is limited globally, though there is evidence for contrasting trends in functional richness and functional evenness with increasing latitude in marine bivalves (Edie et al. 2018; Schumm et al. 2019). Molluscs play important roles in the marine environment such as food and habitat provision, predation, and grazing (Przeslawski et al. 2008). Within the phylum Mollusca, Bivalvia and Gastropoda show strong coupling with specific environmental conditions and habitat types (Zuschin et al. 2001; Schoepf et al. 2010; Fredston-Hermann et al. 2013). Species richness of some molluscs declines with increasing latitude (Roy et al. 2000), whilst other groups show no pattern (Miloslavich et al. 2013), and in some cases patterns differ between ocean basins (Valdovinos et al. 2003). Therefore, it is likely that mollusc assemblages will show a significant response to environmental changes on subtropical reefs. Moreover, their short life spans and limited mobility (Beauchard et al. 2017) make them ideal study species into the effects of environmental change. The lack of functional analyses of subtropical mollusc species currently hinders our understanding of the functional roles of molluscs in the context of current and near future range shifts. Addressing this knowledge gap, we quantify functional characteristics of mollusc species associated with subtropical coral communities along a tropical-to-temperate gradient in Japan. We consider a) whether trait composition and trait space change across the subtropical/tropical transition zone following environmental cues; b) how mollusc functional diversity metrics change along the latitudinal gradient; and c) to what degree mollusc functional redundancy is predicted by species richness changes. This approach provides insight to the functioning of mollusc communities within and across subtropical transition zones, contributing to our understanding of ecological function response to changes, and forms a basis for continued collation of trait information for marine molluscs.
Methods
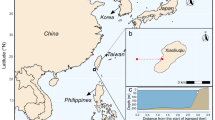
We surveyed a total of 31 sites along the Pacific coast of Japan in the summers of 2015 and 2016, ranging from ~ 24 to ~ 35 degrees Northern latitude (Fig. 1). The latitudinal extent ranges from tropical to temperate climates over ~ 10 degrees of latitude, a transition that is reflected in the ecological communities (Yamano et al. 2001; Beger et al. 2014). The tropical Ryukyu island chain in the south provides sequential habitats across the latitudinal gradient and is influenced by the northerly warm water Kuroshio Current. The current itself acts as a vector for larval dispersal and species introduction, facilitating high connectivity south to north (Yamano et al. 2011). The current splits at approximately 30 degrees latitude at Yakushima Island, and north from this point there is a distinct transition into subtropical ecosystems (Nakano 2004). A recent analysis has demonstrated a lack of relevant mollusc data in the region compared to more well-studied taxa such as hard corals and fishes (Reimer et al. 2019).
Environmental regimes in the study region along the west Pacific coast of Japan, showing a) a multidimensional scaling plot of environmental factors distinguishing five environmental climate regions (arrows indicate environmental variables that contribute to differences among sites), and b) the position of 31 study sites in the climate regions with typical coral reef or coral community habitat where molluscs were recorded
Mollusc species abundance data were collected from coral reefs and communities using visual sublittoral surveys along 25 × 1 m belt transects with between 3 and 5 replicates per site, building on standard methods used to quantify abundances for other reef taxa (Cinner et al. 2016). All analyses were conducted at the site-level with means calculated across transects. Site depths ranged between 6 and 10 metres, following the occurrence of coral communities. The focus of this survey was on macro mollusc species, which were identified to species level where possible following Okutani (2000). Only molluscs identified to species were included in the analyses to ensure that traits were identified and treated at the same taxonomic level. We calculated the mean richness and abundance of mollusc species at each site.
Environmental conditions at the study sites
The 31 study sites were grouped into clusters based on the similarity of their environmental parameters to provide a basis for functional comparison within and between the clusters. Environmental conditions chosen to group the sites related to coral survival due to the significant role corals play in sha** communities. The variables used were mean sea surface temperature (SST) (°C), diffuse light attenuation (m−1), pH, nitrate (mol m−3), phosphate (mol m−3), and calcite (mol m−3). These data were acquired from Bio-ORACLE at a spatial resolution of 5 arcminutes (Tyberghein et al. 2012; Assis et al. 2018). Sites were a greater distance apart than the resolution of the environmental data, allowing for a clear matching of environmental variables to sites. An Euclidean distance matrix was calculated for all sites based on standardised environmental variables. Environmental conditions at the sites were then plotted using non-metric multidimensional scaling (nMDS) to visualise the differences and similarities in environmental conditions among sites. Sites with similar environmental conditions were assigned to the same cluster—clusters were chosen based on visual interrogation of a dendrogram.
Species diversity
To improve our understanding of the system, and to aid our interpretation of the functional analyses, species richness was explored over latitude. Linear and polynomial regression analyses were used to investigate potential latitudinal trends in mollusc species richness. We also conducted a permutational analysis of variance (PERMANOVA) (number of permutations = 999, p-values adjusted to control false discovery rate (fdr)) using the ‘adonis’ function in the R package vegan version 2.5-6 (Oksanen et al. 2019), comparing species composition among ecoregions. This allowed statistical inference of turnover. Sakurajima was not included as an ecoregion, as it is a single site.
Species traits
We collated species traits data from online repositories, primary literature and books. In particular, ‘Marine mollusks in Japan’ (Okutani 2000) was referred to as it offered trait information specific to Japan. Traits from sources geographically close to Japan were included preferentially where possible to reduce the influence of plasticity of trait expression. This method was also used when species showed an affinity for multiple trait categories, for example, living on both sand and rock habitats. We chose seven traits to capture the ecological and functional relationships along the transition zone, such as traits linked to dispersal, trophic position, and habitat use. The number of traits included in the analyses were enough to minimise artificial functional redundancy that could arise from using too few traits, whilst also preventing excessive uniqueness of trait combinations caused by too many traits (Laureto et al. 2015). Trait classes represented biological, behavioural, and physiological features of species, and included two continuous and five categorical data formats:
-
Maximum depth (numerical): This trait reflects species affinity to deep or shallow areas. It correlates with many other environmental variables and can be viewed as a proxy of a species’ tolerance for temperature and physiological effects of pressure. Variation in depth ranges may also influence species responses to temperature changes. Even though we sampled all species at shallow reef sites of similar depths, we assume that species can survive up to the maximum depth recorded in the trait database.
-
Feeding (categorical): This trait captures a species’ main food source that determines its trophic level, discriminating predators, grazers, deposit feeders, and filter/suspension feeders. General feeding modes are used here to inclusively represent the diverse feeding of molluscs. The difference between feeding styles in temperate and tropical gastropod species has been shown to be distinct (Valentine et al. 2002), and the presence and absence of feeding modes can also reflect the availability of food resources in a habitat.
-
Habitat preference (categorical): This trait records the physical environment the species predominantly inhabits. It indicates the substrate type and associated environmental conditions such as energy levels and exposure that a species typically occupies. The trait levels are coral, mud, rock, and sand habitat preference.
-
Mobility (categorical): Mollusc species exhibit vastly different mobility—this is captured in a binary representation trait of mobile and sessile taxa. A species’ mobility conveys its role in an ecosystem for energy transmission (Hyndes et al. 2014), and can influence its response to disturbance (Tillin et al. 2006).
-
Position (categorical): This trait relates to how a species utilises a habitat and its physical positioning including burrowing, attached to substrate, and free-living species. This trait can indicate the likely role a species plays in a community and its sensitivity to certain stressors such as dredging and specific fishing practices (Buhl-Mortensen et al. 2015).
-
Size (numerical): Is a fundamental trait often used in trait studies (Paganelli et al. 2012; Ramírez-Ortiz et al. 2017) that has been shown to play a strong role in determining species’ responses to environmental parameters (Feary et al. 2014). Molluscs are diverse in their morphology, and to capture this in a single size metric, length was chosen from posterior to anterior where possible. A multidimensional metric could provide a better representation of size, though currently such data are not widely available.
-
Shape (categorical): This trait relates to many ecological factors, for example resistance to predation, ability to withstand wave energy, and position in a habitat. The shape trait takes the categories of conic, dorsoventrally compressed, laterally compressed, fusiform, ovate, turbinate, and vermiform. In this case, no distinction was made between hard shelled organisms and soft organisms (e.g. nudibranchs).
The traits chosen reflect not only ecological contexts, but also data availability. Trait-based analyses of other taxa usually include traits describing reproductive strategies (e.g. spawning type, parental care (Darling et al. 2012; Stuart-Smith et al. 2013; D’agata et al. 2014; Sommer et al. 2014; Mouillot et al. 2014), and behaviour [e.g. gregariousness (Stuart-Smith et al. 2013)]. The inclusion of these traits would be desirable, particularly those linked to dispersal capabilities (Blanco et al. 2019) that are particularly important due to the high connectivity provided by the Kuroshio Current (Yamano et al. 2011). However, we were unable to include such traits because they are not well known for the mollusc species sampled here.
Trait composition analysis
To examine trait compositional change over the subtropical transition zone, we calculated community weighted means (CWM) for each trait at each site using the R package FD version 1.0-12 (Laliberté et al. 2014). For continuous traits, community weighted means provides the average trait score for all species in a community weighted by their abundance; for categorical traits the metric provides abundance of each trait level in a community (Lavorel et al. 2008). Community weighted means of each site were calculated by combining the trait matrix and the site by species abundance matrix. A Bray–Curtis dissimilarity matrix was generated from the scaled community weighted means. An nMDS plot based on this distance matrix was then generated using the ‘metaMDS’ function in the R package vegan to allow for the identification of differences among sites based on trait composition. We also conducted a PERMANOVA (number of permutations = 999, p-values adjusted to control fdr) using the ‘adonis’ function to compare trait composition between ecoregions. This analysis did not include Sakurajima as an ecoregion.
The direction cosine and r2 values for each of the trait variables, environmental variables, and latitude were extracted from the outputs of the function ‘envfit’ in R package vegan and retained for nMDS axes 1 and 2. This method was used to explore which traits drive differences seen in the ordination and whether the differences could be explained by latitudinal effects. Due to the number of traits in the analysis, a permutation test was conducted on the r2 values to identify values that were unlikely to be observed by chance. This one-tailed test uses 999 random permutations to estimate the chance of observing the r2 value if there were no correlation between the trait and the ordination (Wang et al. 2012; Oksanen 2015).
Trait space
To explore the changes in trait diversity over latitude we used convex hulls (Cornwell et al. 2006; McWilliam et al. 2018). This approach identifies the spread of assemblages within trait space through ordination, with the most extreme values defining the vertices of the hull. A Gower dissimilarity matrix was generated from the trait data and used in a principal coordinate analysis (PCoA). To create the global convex hull, all species in the dataset were included in the analysis to represent the full trait space. Regional hulls were then generated by subsetting the species present at each ecoregion. Percentage occupancy of each regional hull within the global hull was calculated by dividing the area of each regional hull by the area of the global hull, using the packages FD, ade4 version 1.7-13 (Dray and Dufour 2007), and sp version 1.4-1 (Pebesma and Bivand 2005).
Functional indices
We calculated functional indices to quantify how mollusc community functioning changes across the latitudinal gradient. Following standard methodology, we generated functional diversity indices with the FD package based on two matrices—species abundance by site and trait by species. As trait data included both continuous and categorical traits, we chose Gower distance to create the underlying trait dissimilarity matrix. We calculated functional diversity RaoQ, functional richness FRic, and functional dispersion FDis as standard metrics to describe trait-based functional patterns (Laliberté and Legendre 2010). A Cailliez correction was applied to prevent negative eigenvalues from being generated from the non-Euclidean matrix. Rao’s Q is a functional diversity metric that considers both trait abundance and difference between species in a community (Botta-Dukát 2005). The two other metrics used—functional richness and dispersion—act to break down functional diversity into its component parts: functional richness considers the abundance of traits by measuring the trait space occupied by species in a community (Villéger et al. 2008), functional dispersion considers the difference between species in a community by estimating the distribution of species in trait space in terms of dispersion or clustering (Villéger et al. 2008). Diversity was not calculated for sites 14 (Yakushima, Yudomari) and 18 (Amami, Ankyaba) as each community had fewer than three functionally unique species.
We used regression analyses to explore how these three metrics change with latitude. A linear regression was first used to model the data, then a polynomial regression was used to test for the better fit of a curved line. Six models were created to explore the effect of latitude on functional diversity, functional richness, and functional dispersion. The model that best fit the data was decided by comparing the percentage of variability explained.
Functional redundancy
The degree of functional redundancy in the system was explored by regressing functional richness against species richness (Teichert et al. 2017). The level of functional redundancy can be estimated from the shape of the regression line. A linear association suggests that for each increase in species richness there is a subsequent increase in functional richness, signifying low functional redundancy as each new species adds new traits (Teichert et al. 2017). A saturation curve suggests that at some level of species richness, more species add redundant traits and therefore do not contribute to increased functional richness (Teichert et al. 2017).
Regression models were built to explore the relationship between species richness and functional richness. A linear and logarithmic model was generated, the model that best fit the data was chosen based on the amount of variability explained. The log regression was used to test for a saturation of functional richness with increasing species richness that is indicative of functional redundancy. To provide further support for the regression analysis, functional redundancy was calculated for all sites using R package SYNCSA version 1.3.3 (Debastiani and Pillar 2012). Functional redundancy is calculated as the difference between Rao’s Q and species diversity based on the functional dissimilarity (Ricotta et al. 2016). The change in functional redundancy with species richness was then investigated using a linear regression.
Results
This study recorded the abundance, distribution and trait information of 88 species of molluscs from 29 families over 10 degrees of latitude along the Pacific coast of Japan.
The environmental conditions at the study sites separated into five distinct clusters that were named to represent their environmental regime (Table 1, Fig. 1). Calcite was the only variable that did not correlate significantly with the ordination (Table 2). Sites at lower latitudes were characterised by higher SST and lower pH, nitrate, and phosphate (Table 1, Fig. 1). Sakurajima (Site 2) showed strong differences to all other sites, due to its high calcite and light attenuation, and was thus categorised into a separate cluster (cluster 5). Although this might have represented a data artefact arising from Sakurajima being situated in an enclosed bay that distorts environmental satellite data records, we believe that the island was accurately singled out as different, as it is exposed to high tidal and temperature fluctuations, and volcanism, leading to a highly distinct environment (Ono et al. 2007). The five clusters were then used to compare the functioning within and among regions that are environmentally similar.
Trait composition analysis
Trait composition of mollusc assemblages separated clearly along nMDS axis 1. Additionally, permutation tests of the r2 values of each trait showed traits and environmental variables that significantly correlated with the ordination (Fig. 2, Table 3). We found a continuous change of trait community composition associated with latitude, with the low latitude sites generally showing lower scores on the first axis than the higher latitude sites (Fig. 2). This was supported by a positive direction cosine and a significant r2 value for the latitude vector on axis 1 (Table 3). Indeed, axis 1 relates to changes in environmental conditions and community structure over latitude, as the clusters generally retained their latitudinal ranks. In particular, subtropical and tropical clusters show clear separation along axis 1, at approximately 30 degrees latitude. Axis 2 corresponds to variation within site clusters, particularly in cold tropical and subtropical clusters (Fig. 2) driven by differences in mollusc shape, feeding mode, and habitat preference (Fig. 2, Table 3). PERMANOVA indicated significant differences in trait composition between tropical and cold tropical vs subtropical and temperate ecoregions (Fig. 3).
Multivariate description of mollusc assemblages associated with coral communities at 31 sites based on their traits, with a) showing the position of sites in mollusc trait space, indicating a continuous change in trait composition with tropical and subtropical clusters showing differentiation from subtropical and temperate clusters, and b) showing factors that contribute most to differences among sites (blue arrows represent traits and the red arrow represents latitude)
Trait space of mollusc assemblages from coral communities along the Pacific coast of Japan. PCoA is used to generate hulls of mollusc species in trait space. The grey hull represents the full trait space occupied by species in the dataset, then subset into ecoregions: a) Tropical, b) Cold Tropical, c) Sakurajima, d) Subtropical, and e) Temperate. f) shows significance of functional change across ecoregions using Community Weigted Means, pairwise comparisons with permutation MANOVAs are shown amongst ecoregions, with Sakurajima removed
Trait space
The first two axes generated from the PCoA of communities explained 64.1% of the variation in the data (axis 1 = 39.6%, axis 2 = 24.5%). The tropical hull occupied 41% of the global hull area, the cold tropical hull occupied 89%, and the subtropical hull occupied 87.7%. The temperate hull occupied 7.3% and Sakurajima occupied 0.8% of the global trait space (Fig. 3).
Species diversity
Both linear and polynomial regression analyses indicated no trends in mollusc species richness over latitude (linear, F = 3.48, df = 1, 29, p = 0.072, r2 = 0.11; polynomial, F = 2.06, df = 2,28, p = 0.15, r2 = 0.13) (Fig. 4a). The PERMANOVA analysis showed significant differences in community composition between between tropical and cold tropical vs subtropical and temperate ecoregions (Supplementary material, Figure S1).
Changes of taxonomic and functional community indices of mollusc assemblages in coral communities along the Pacific coast of Japan with latitude, showing a) species richness, b) functional diversity (Rao’s Q), and c) functional dispersion, with increasing latitude. A polynomial regression is used to model functional diversity and dispersion
Functional diversity indices
Linear and polynomial models showed a significant negative relationship of functional diversity RaoQ with latitude, explaining ~ 34% and ~ 46% of variation in functional diversity, respectively (Table 4). We retained the polynomial model as a better representation of the data (Fig. 4b). Both the linear and polynomial models indicated no significant relationship of functional richness FRic with latitude (Table 4).
The linear model of functional dispersion FDis indicated a significant negative relationship with latitude, explaining ~ 32% of variation (Table 4). The polynomial model also showed a significant negative relationship of functional dispersion with latitude, explaining ~ 49% of the variation in the data, ~ 17% more than the linear model (Table 4). Therefore, the polynomial model was selected as a better fit (Fig. 4c).
Functional redundancy
The linear model showed a significant positive relationship between species richness on functional richness (r2 = 0.15, Table 5). The logarithmic model also showed a positive effect with an asymptote at higher species richness, explaining ~ 19% of the variability: ~ 4% more than the linear model (Table 5). Therefore, the logarithmic model was chosen as it explained more of the variability (Fig. 5). Functional redundancy showed no significant relationship with species richness (Table 5).
Discussion
Here, our results have shown that trait composition of coral community associated mollusc assemblages along the tropical-to-temperate transition in Japan varies continuously with latitude. The functional diversity of communities decreases with latitude, in concert with loss in functional dispersion, whereas gradients in functional richness are independent of latitude. Although we find some evidence for higher functional redundancy at higher species richness, the variability in the relationship indicates that species richness alone does not explain gradients in functional redundancy.
The changes in trait composition recorded in our study are generally continuous along latitudinal environmental gradients (Fig. 2a, Table 3) and there is a distinct separation of subtropical and tropical regions (Fig. 2a). Communities in these regions also occupy more of the trait space than those in tropical and temperate regions (Fig. 3). Further, communities in tropical and temperate regions have distinct trait spaces. Species traits collectively define functions (Violle et al. 2007; Laureto et al. 2015) and it is likely that the ecological functions provided by mollusc communities also change with latitude. Distribution patterns, feeding strategies and substrate relations of molluscs were found to be closely linked with habitat type in the Red Sea (Zuschin et al. 2001). In our study, mollusc assemblages at temperate and subtropical sites were characterised by species that are grazers, prefer rocky substrate, are free-living, mobile, and have a conical shape (Fig. 2b). At these higher latitude sites, the availability of hard substrate is more likely rock than coral (Nagai et al. 2011) and the prevalence of macroalgae (Sommer et al. unpublished) likely influences functional trait patterns in molluscs. Our finding that species are free-living and conic, shows that gastropod grazers are abundant at temperate and subtropical sites. Traits at tropical and cold tropical sites indicate a prevalence of species that prefer coral substrate, are suspension feeders, attached to substrate, and laterally compressed (Fig. 2b). Species included here are large in size, and include Pedum spondyloideum, Pinctada margaritifera, and Tridacna spp. This suggests tropical and cold tropical sites are dominated by bivalves. This distinction is reflected in trait space (Fig. 3) as species with higher values on axes 1 and 2 are bivalves. A conspicuous difference between tropical and temperate ecoregions in trait space is the prevalence of bivalve type species in the tropics, and comparably fewer of such species in temperate regions. This suggests that at tropical and cold tropical sites, large bivalves contribute to the utilisation of waste organic material and excess nutrients through suspension feeding (Neo et al. 2015), thus contributing to nutrient recycling on reefs. Large bivalves are also important ecosystem engineers by increasing habitat heterogeneity (Neo et al. 2015). At temperate and subtropical sites, gastropod herbivory is likely to affect algal community composition and competitive balances which could influence the establishment potential of corals (Hughes et al. 2007).
Multiple environmental variables change along marine latitudinal gradients. Of these, SST has been shown to strongly influence the palaeoecological marine mollusc assemblages in Japan (Ogasawara 1994), and extant assemblages over large spatial scales (Roy et al. 1998, 2000; Fernández et al. 2009). This effect was confirmed over our study region. However, the effect of many other environmental variables is also strong (Table 3), suggesting that mollusc assemblages are probably influenced by multiple processes. It is also possible that mollusc assemblages are not directly responding to environmental drivers such as SST, but instead responding to the effects of these variables on habitat-building taxa. For example, long- and short-term SST variability and light availability stongly influence distribution patterns in subtropical coral assemblages (Muir et al. 2015; Sommer et al. 2018) and scleractinian corals in turn influence ecosystem structure (Graham and Nash 2013). It is thus plausible that over relatively small scales, mollusc species that are strongly associated with certain coral types respond to changes in coral abundance. Therefore, variation in trait composition of mollusc assemblages is likely governed by a combination of biotic and abiotic drivers. To investigate this, the changes in trait composition of corals and other key taxa need to be considered over the same sites to explore the correlation between mollusc traits and other key taxa.
Of particular interest when considering trait composition is the difference between tropical and subtropical regions (Fig. 1), allowing us to understand how functioning of mollusc communities could alter with continued climate change. Our study shows a difference in trait composition between tropical and subtropical regions (Fig. 2). Though not directly quantified, the Kuroshio Current is likely to play a substantial role in the observed difference in trait composition between tropical and subtropical sites. The Kuroshio Current shows a strong influence on marine communities in the west Pacific through its effect on high-latitude abiotic conditions (Yatsu et al. 2013) and dispersal opportunities. The Kuroshio Current splits near the island of Yakushima at around 30 degrees latitude into northeasterly and northwesterly currents, causing significant gradients in associated environmental conditions (Chen et al. 2010). Reef accretion ceases at Yakushima (Veron and Minchin 1992; Nakano 2004). This region of oceanographic change coincides with strong gradients in mollusc trait composition observed here, as well as the distinct differences between tropical/cold tropical and subtropical/temperate regions (Fig. 2). Western boundary currents, such as the Kuroshio Current, are undergoing significant changes in intensity and temperature in response to climate change (Hobday and Lough 2011; Wu et al. 2012). It is likely that the associated oceanographic changes will alter ecosystem functioning along the tropical-to-temperate transition zone in Japan under climate change.
Our data show a trend of decreasing functional diversity of communities with increasing latitude (Fig. 4) over 10 degrees of latitude. Both functional diversity and functional dispersion show variability within clusters and also around the regression line. This variability suggests that factors other than latitudinal environmental gradients influence the functional diversity of mollusc assemblages. Valdovinos et al. (2003) show the importance of local scale processes in influencing mollusc diversity gradients. Similarly, we infer that small-scale factors, such as habitat features and associated small-scale variation in environmental conditions, probably also play a role in driving the observed patterns. For example, sites varied at small scales in their physical characteristics, with some showing a steeper slope profile, whilst others were more exposed. Local scale habitat characteristics therefore likely affect the amount of suitable habitat for mollusc species within an ecosystem, and associated patterns in trait composition. The consideration of small-scale habitat features in future work therefore has the potential to further improve our understanding of ecological dynamics of mollusc assemblages on these reefs.
Unlike previous studies on bivalves (Edie et al. 2018; Schumm et al. 2019), gradients in functional richness were not associated with latitude over the spatial scale of our study. Instead, it is the difference between species in communities that is associated with the changes in functional diversity, as functional dispersion declines at subtropical latitudes (Fig. 4c). It is expected that mollusc communities at subtropical sites will experience higher abiotic stress because communities are composed of tropical and temperature species at the edges of their latitudinal extents (Vroom and Braun 2010). Species similarity in areas of high abiotic stress can be indicative of environmental filtering—for example for high-latitude coral assemblages (Sommer et al. 2014; Sommer et al. 2017). Contrastingly, biotic interactions such as competition are thought to have the opposite effect, driving species differences (Cavender-Bares et al. 2004). Given the strong role of abiotic processes at transition zones for corals (Sommer et al. 2014, 2017; Mizerek et al., 2016) and the greater declines in functional dispersion of mollusc assemblages at subtropical sites in our study (Fig. 4b), it is plausible that environmental filtering also plays an important role in explaining the observed decrease in functional dispersion of mollusc assemblages at higher latitudes.
In light of the patterns in functional diversity, functional redundancy is important in buffering the functional stability of communities against species loss (Teichert et al. 2017). While our results show some evidence for functional redundancy at sites with higher species richness (Fig. 5), high variability suggests that our analyses do not capture the full picture of functional redundancy change. Evidence of functional redundancy is lacking for many subtropical marine species and to our knowledge has not been directly addressed in marine mollusc assemblages (Schumm et al. 2019). Fishes are best studied and despite reports of low functional redundancy for many systems (Micheli and Halpern 2005), there is evidence of functional redundancy in tropical assemblages (Mouillot et al. 2014). Our results are an important first step to identify potential redundancy in sites with higher species richness, but it is apparent that species richness alone does not explain gradients in functional redundancy.
Consideration of additional traits could further bolster our understanding of the functioning of mollusc assemblages, particularly larval development information (Fernández et al. 2009). The availability of mollusc traits was limiting, because information such as reproductive mode and larval duration were unavailable for many species. Moreover, size and shape vary greatly among mollusc species, such that for a tall but not long-shaped species, height may be a more appropriate measure of size than length. Where possible, length was used; however, in some cases, length measurements were not available. Traits were either categorical or continuous data formats, which in each case allows for one value or category to be recorded per species. Intra-species plasticity of traits is well documented in response to abiotic (Hollander et al. 2006) and biotic (Bourdeau 2009) factors, and probably also occurs for molluscs in Japan. For example, many of the species in the dataset exhibit an affinity for multiple substrate types and feeding modes. The methods used here do not allow for consideration of trait plasticity, however fuzzy coding of traits (Chevene et al. 1994) offers a promising alternative to simple categorisation of traits by accounting for variability of traits expressed by species (Fiorentino et al. 2017). We did not use fuzzy coding here as it requires accurately characterising the affinity of each species to every trait level used: such detail is not currently available. Functional diversity indices that incorporate fuzzy coded data are available (Villéger et al. 2017), and testing latitudinal functional change in mollusc communities that accounts for trait plasticity would provide increased scope for understanding and predicting community reassembly.
Understanding how ecosystems along the tropical-to-temperate transition vary along spatial and environmental gradients is a prerequisite to understanding how they function and how they might respond to climate change. This study considers the functioning of marine mollusc assemblages at biogeographic transition zones and highlights their changing roles along the latitudinal gradient. In particular, high abundance of substrate-attached, suspension feeding bivalves in the tropics points to their important role in nutrient cycling on coral reef ecosystems, while high prevalence of free-living, gastropod grazers at temperate and subtropical latitudes identifies molluscs as important herbivores on subtropical reefs. Declines in functional diversity with increasing latitude indicates a simplification of mollusc assemblages at higher latitudes. This study provides new insights for important taxa along biogeographic transition zones and establishes important baselines for future work. The strong biogeographic transition recorded in this study is linked to oceanographic features in the region, highlighting the potential for substantial shifts in trait composition and functioning of subtropical mollusc assemblages under climate change.
References
Appeltans W, Ahyong ST, Anderson G, Angel MV, Artois T, Bailly N, Bamber R, Barber A, Bartsch I, Berta A, Błażewicz-Paszkowycz M, Bock P, Boxshall G, Boyko CB, Brandão SN, Bray RA, Bruce NL, Cairns SD, Chan T-Y, Cheng L, Collins AG, Cribb T, Curini-Galletti M, Dahdouh-Guebas F, Davie PJF, Dawson MN, De Clerck O, Decock W, De Grave S, de Voogd NJ, Domning DP, Emig CC, Erséus C, Eschmeyer W, Fauchald K, Fautin DG, Feist SW, Fransen CHJM, Furuya H, Garcia-Alvarez O, Gerken S, Gibson D, Gittenberger A, Gofas S, Gómez-Daglio L, Gordon DP, Guiry MD, Hernandez F, Hoeksema BW, Hopcroft RR, Jaume D, Kirk P, Koedam N, Koenemann S, Kolb JB, Kristensen RM, Kroh A, Lambert G, Lazarus DB, Lemaitre R, Longshaw M, Lowry J, Macpherson E, Madin LP, Mah C, Mapstone G, McLaughlin PA, Mees J, Meland K, Messing CG, Mills CE, Molodtsova TN, Mooi R, Neuhaus B, Ng PKL, Nielsen C, Norenburg J, Opresko DM, Osawa M, Paulay G, Perrin W, Pilger JF, Poore GCB, Pugh P, Read GB, Reimer JD, Rius M, Rocha RM, Saiz-Salinas JI, Scarabino V, Schierwater B, Schmidt-Rhaesa A, Schnabel KE, Schotte M, Schuchert P, Schwabe E, Segers H, Self-Sullivan C, Shenkar N, Siegel V, Sterrer W, Stöhr S, Swalla B, Tasker ML, Thuesen EV, Timm T, Todaro MA, Turon X, Tyler S, Uetz P, van der Land J, Vanhoorne B, van Ofwegen LP, van Soest RWM, Vanaverbeke J, Walker-Smith G, Walter TC, Warren A, Williams GC, Wilson SP, Costello MJ (2012) The magnitude of global marine species diversity. Current Biology 22(23):2189–2202
Assis J, Tyberghein L, Bosch S, Verbruggen H, Serrão EA, De Clerck O (2018) Bio-ORACLE v.20: extending marine data layers for bioclimatic modelling. Global Ecology and Biogeography. 27(3):277–284
Beauchard O, Veríssimo H, Queirós AM, Herman PMJ (2017) The use of multiple biological traits in marine community ecology and its potential in ecological indicator development. Ecological Indicators. 76:81–96
Beger M, Sommer B, Harrison PL, Smith SDA, Pandolfi JM (2014) Conserving potential coral reef refuges at high latitudes. Diversity and Distributions. 20(3):245–257
Bennett S, Wernberg T, Harvey ES, Santana-Garcon J, Saunders BJ (2015) Tropical herbivores provide resilience to a climate-mediated phase shift on temperate reefs. Ecology Letters. 18(7):714–723
Blanco M, Ospina-Alvarez A, Navarrete S, Fernández M (2019) Influence of larval traits on dispersal and connectivity patterns of two exploited marine invertebrates in central Chile. Marine Ecology Progress Series. 612:1
Bland LM, Regan TJ, Dinh MN, Ferrari R, Keith DA, Lester R, Mouillot D, Murray NJ, Nguyen HA, Nicholson E (2017) Using multiple lines of evidence to assess the risk of ecosystem collapse. Proceedings of the Royal Society B: Biological Sciences. 284(1863):20170660
Botta-Dukát Z (2005) Rao’s quadratic entropy as a measure of functional diversity based on multiple traits. Journal of Vegetation Science. 16(5):533–540
Bourdeau PE (2009) Prioritized phenotypic responses to combined predators in a marine snail. Ecology. 90(6):1659–1669
Buhl-Mortensen L, Neat F, Koen-Alonso M, Hvingel C, Holte B (2015) Fishing impacts on benthic ecosystems: an introduction to the 2014 ICES symposium special issue. ICES Journal of Marine Science. 73(1):1–4
Cavender-Bares J, Ackerly DD, Baum DA, Bazzaz FA (2004) Phylogenetic overdispersion in Floridian oak communities. The American Naturalist. 163(6):823–843
Chen M-T, Chang Y-P, Yu P-S, Shiau L-J (2010) Marine records of East Asian monsoon variability over the past 5 Ma. Geological Society, London, Special Publications. 342(1):5–15
Chevene F, Doleadec S, Chessel D (1994) A fuzzy coding approach for the analysis of long-term ecological data. Freshwater Biology. 31(3):295–309
Cinner JE, Huchery C, MacNeil MA, Graham NAJ, McClanahan TR, Maina J, Maire E, Kittinger JN, Hicks CC, Mora C, Allison EH, D’Agata S, Hoey A, Feary DA, Crowder L, Williams ID, Kulbicki M, Vigliola L, Wantiez L, Edgar G, Stuart-Smith RD, Sandin SA, Green AL, Hardt MJ, Beger M, Friedlander A, Campbell SJ, Holmes KE, Wilson SK, Brokovich E, Brooks AJ, Cruz-Motta JJ, Booth DJ, Chabanet P, Gough C, Tupper M, Ferse SCA, Sumaila UR, Mouillot D (2016) Bright spots among the world’s coral reefs. Nature. 535(7612):416–419
Cornwell WK, Schwilk DW, Ackerly DD (2006) A trait-based test for habitat filtering: convex hull volume. Ecology. 87(6):1465–1471
D’agata S, Mouillot D, Kulbicki M, Andréfouët S, Bellwood DR, Cinner JE, Cowman PF, Kronen M, Pinca S, Vigliola L (2014) Human-mediated loss of phylogenetic and functional diversity in coral reef fishes. Current Biology. 24(5):555–560
Darling ES, Alvarez-Filip L, Oliver TA, McClanahan TR, Côté IM (2012) Evaluating life-history strategies of reef corals from species traits. Ecology Letters. 15(12):1378–1386
Debastiani VJ, Pillar VD (2012) SYNCSA—R tool for analysis of metacommunities based on functional traits and phylogeny of the community components: version 1.3.3. Bioinformatics. 28(15):2067–2068
Díaz S, Purvis A, Cornelissen JHC, Mace GM, Donoghue MJ, Ewers RM, Jordano P, Pearse WD (2013) Functional traits, the phylogeny of function, and ecosystem service vulnerability. Ecology and Evolution. 3(9):2958–2975
Dray S, Dufour A (2007) The ade4 package: implementing the duality diagram for ecologists. Version 1.7-13
Edie SM, Jablonski D, Valentine JW (2018) Contrasting responses of functional diversity to major losses in taxonomic diversity. Proceedings of the National Academy of Sciences. 115(4):732–737
Feary DA, Pratchett MS, Emslie M, Fowler AM, Figueira WF, Luiz OJ, Nakamura Y, Booth DJ (2014) Latitudinal shifts in coral reef fishes: why some species do and others do not shift. Fish and Fisheries. 15(4):593–615
Fernández M, Astorga A, Navarrete SA, Valdovinos C, Marquet PA (2009) Deconstructing latitudinal species richness patterns in the ocean: does larval development hold the clue? Ecology Letters. 12(7):601–611
Fiorentino D, Pesch R, Guenther C-P, Gutow L, Holstein J, Dannheim J, Ebbe B, Bildstein T, Schroeder W, Schuchardt B, Brey T, Wiltshire KH (2017) A ‘fuzzy clustering’ approach to conceptual confusion: how to classify natural ecological associations. Marine Ecology Progress Series. 584:17–30
Fredston-Hermann AL, O’Dea A, Rodriguez F, Thompson WG, Todd JA (2013) Marked ecological shifts in seagrass and reef molluscan communities since the mid-Holocene in the southwestern Caribbean. Bulletin of Marine Science. 89(4):983–1002
Graham NAJ, Nash KL (2013) The importance of structural complexity in coral reef ecosystems. Coral Reefs. 32(2):315–326
Hobday AJ, Lough JM (2011) Projected climate change in Australian marine and freshwater environments. Marine and Freshwater Research. 62(9):1000–1014
Hollander J, Collyer ML, Adams DC, Johannesson K (2006) Phenotypic plasticity in two marine snails: constraints superseding life history. Journal of Evolutionary Biology. 19(6):1861–1872
Hooper DU, Chapin FS III, Ewel JJ, Hector A, Inchausti P, Lavorel S, Lawton JH, Lodge DM, Loreau M, Naeem S, Schmid B, Setälä H, Symstad AJ, Vandermeer J, Wardle DA (2005) Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecological Monographs. 75(1):3–35
Hubbell SP (2005) Neutral theory in community ecology and the hypothesis of functional equivalence. Functional Ecology. 19(1):166–172
Hughes TP, Barnes ML, Bellwood DR, Cinner JE, Cumming GS, Jackson JBC, Kleypas J, van de Leemput IA, Lough JM, Morrison TH, Palumbi SR, van Nes EH, Scheffer M (2017) Coral reefs in the Anthropocene. Nature. 546:82
Hughes TP, Rodrigues MJ, Bellwood DR, Ceccarelli D, Hoegh-Guldberg O, McCook L, Moltschaniwskyj N, Pratchett MS, Steneck RS, Willis B (2007) Phase shifts, herbivory, and the resilience of coral reefs to climate change. Current Biology. 17(4):360–365
Hyndes GA, Nagelkerken I, McLeod RJ, Connolly RM, Lavery PS, Vanderklift MA (2014) Mechanisms and ecological role of carbon transfer within coastal seascapes. Biological Reviews. 89(1):232–254
Kim SW, Sampayo EM, Sommer B, Sims CA, del Gómez-Cabrera MC, Dalton SJ, Beger M, Malcolm HA, Ferrari R, Fraser N, Figueira WF, Smith SDA, Heron SF, Baird AH, Byrne M, Eakin CM, Edgar R, Hughes TP, Kyriacou N, Liu G, Matis PA, Skirving WJ, Pandolfi JM (2019) Refugia under threat: mass bleaching of coral assemblages in high-latitude eastern Australia. Global Change Biology. 25(11):3918–3931
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology. 91(1):299–305
Laliberté E, Legendre P, Shipley B (2014) FD: measuring functional diversity from multiple traits, and other tools for functional ecology. R package version 1.0-12
Laureto LMO, Cianciaruso MV, Samia DSM (2015) Functional diversity: an overview of its history and applicability. Natureza & Conservação. 13(2):112–116
Lavorel S, Grigulis K, McIntyre S, Williams NSG, Garden D, Dorrough J, Berman S, Quétier F, Thébault A, Bonis A (2008) Assessing functional diversity in the field – methodology matters! Functional Ecology. 22(1):134–147
Madin EMP, Ban NC, Doubleday ZA, Holmes TH, Pecl GT, Smith F (2012) Socio-economic and management implications of range-shifting species in marine systems. Global Environmental Change. 22(1):137–146
Madin JS, Hoogenboom MO, Connolly SR, Darling ES, Falster DS, Huang D, Keith SA, Mizerek T, Pandolfi JM, Putnam HM, Baird AH (2016) A trait-based approach to advance coral reef science. Trends in Ecology & Evolution. 31(6):419–428
Mason NWH, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos. 111(1):112–118
McWilliam M, Hoogenboom MO, Baird AH, Kuo C-Y, Madin JS, Hughes TP (2018) Biogeographical disparity in the functional diversity and redundancy of corals. Proceedings of the National Academy of Sciences. 115(12):3084–3089
Micheli F, Halpern BS (2005) Low functional redundancy in coastal marine assemblages. Ecology Letters. 8(4):391–400
Micheli F, Mumby PJ, Brumbaugh DR, Broad K, Dahlgren CP, Harborne AR, Holmes KE, Kappel CV, Litvin SY, Sanchirico JN (2014) High vulnerability of ecosystem function and services to diversity loss in Caribbean coral reefs. Biological Conservation. 171:186–194
Miloslavich P, Cruz-Motta JJ, Klein E, Iken K, Weinberger V, Konar B, Trott T, Pohle G, Bigatti G, Benedetti-Cecchi L, Shirayama Y, Mead A, Palomo G, Ortiz M, Gobin J, Sardi A, Díaz JM, Knowlton A, Wong M, Peralta AC (2013) Large-scale spatial distribution patterns of gastropod assemblages in rocky shores. PLoS One. 8(8):e71396–e71396
Mittelbach GG, Schemske DW, Cornell HV, Allen AP, Brown JM, Bush MB, Harrison SP, Hurlbert AH, Knowlton N, Lessios HA, McCain CM, McCune AR, McDade LA, McPeek MA, Near TJ, Price TD, Ricklefs RE, Roy K, Sax DF, Schluter D, Sobel JM, Turelli M (2007) Evolution and the latitudinal diversity gradient: speciation, extinction and biogeography. Ecology Letters. 10(4):315–331
Mizerek TL, Baird AH, Beaumont LJ, Madin J, Madin JS (2016) Environmental tolerance governs the presence of reef corals at latitudes beyond reef growth: coral traits in beyond reef assemblages. Global Ecology and Biogeography. 25(8):979–987
Mouillot D, Graham NAJ, Villéger S, Mason NWH, Bellwood DR (2013) A functional approach reveals community responses to disturbances. Trends in Ecology & Evolution. 28(3):167–177
Mouillot D, Villéger S, Parravicini V, Kulbicki M, Arias-González JE, Bender M, Chabanet P, Floeter SR, Friedlander A, Vigliola L, Bellwood DR (2014) Functional over-redundancy and high functional vulnerability in global fish faunas on tropical reefs. Proceedings of the National Academy of Sciences. 111(38):13757–13762
Muir PR, Wallace CC, Done T, Aguirre JD (2015) Limited scope for latitudinal extension of reef corals. Science. 348(6239):1135–1138
Munday PL, Jones GP, Pratchett MS, Williams AJ (2008) Climate change and the future for coral reef fishes. Fish and Fisheries. 9(3):261–285
Nagai S, Yoshida G, Tarutani K (2011) Change in species composition and distribution of algae in the coastal waters of western Japan. In: Global warming impacts-case studies on the economy, human health, and on urban and natural environments. IntechOpen
Nakano Y (2004) Coral Reefs of Japan: Distribution of Coral Reefs. In: Coral Reefs of Japan (Online). Ministry of the Environment, pp 1–13. https://www.env.go.jp/nature/biodic/coralreefs/reference/contents/0101.pdf. Accessed 30 Sept 2019
Neo ML, Eckman W, Vicentuan K, Teo SL-M, Todd PA (2015) The ecological significance of giant clams in coral reef ecosystems. Biological Conservation. 181:111–123
Ogasawara K (1994) Neogene paleogeography and marine climate of the Japanese Islands based on shallow-marine molluscs. Palaeogeography, Palaeoclimatology, Palaeoecology. 108(3):335–351
Oksanen J (2015) Vegan: an introduction to ordination. http://cran.r-project.org/web/packages/vegan/vignettes/introvegan.pdf. 8:19
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2019) vegan: Community Ecology Package. R package version 2.5-6
Okutani T (2000) Marine mollusks in Japan. Tokai University Press, Tokyo
Oliver ECJ, Donat MG, Burrows MT, Moore PJ, Smale DA, Alexander LV, Benthuysen JA, Feng M, Sen Gupta A, Hobday AJ, Holbrook NJ, Perkins-Kirkpatrick SE, Scannell HA, Straub SC, Wernberg T (2018) Longer and more frequent marine heatwaves over the past century. Nature Communications. 9(1):1324
Ono S, Reimer JD, Tsukahara J (2007) Thriving cnidarian community on the volcanic coastline of Sakurajima Taisho Lava Field, southern Japan. Coral Reefs. 26(1):95
Paganelli D, Marchini A, Occhipinti-Ambrogi A (2012) Functional structure of marine benthic assemblages using Biological Traits Analysis (BTA): a study along the Emilia-Romagna coastline (Italy, North-West Adriatic Sea). Estuarine, Coastal and Shelf Science. 96:245–256
Pebesma E, Bivand R (2005) Classes and methods for spatial data in R. Version 1.4-1. R News
Petchey OL, Gaston KJ (2002) Extinction and the loss of functional diversity. Proceedings of the Royal Society of London Series B: Biological Sciences. 269:1721–1727
Pratchett MS, Thompson CA, Hoey AS, Cowman PF, Wilson SK (2018) Effects of coral bleaching and coral loss on the structure and function of reef fish assemblages—coral bleaching: patterns, processes, causes and consequences In: van Oppen MJH, Lough JM (eds) Springer, Cham, pp 265–293. https://doi.org/10.1007/978-3-319-75393-5_11
Przeslawski R, Ahyong S, Byrne M, Wörheide G, Hutchings P (2008) Beyond corals and fish: the effects of climate change on noncoral benthic invertebrates of tropical reefs. Global Change Biology. 14(12):2773–2795
Ramírez-Ortiz G, Calderon-Aguilera LE, Reyes-Bonilla H, Ayala-Bocos A, Hernández L, Fernández Rivera-Melo F, López-Pérez A, Dominici-Arosamena A (2017) Functional diversity of fish and invertebrates in coral and rocky reefs of the Eastern Tropical Pacific. Marine Ecology. 38(4):e12447
Reimer JD, Biondi P, Lau YW, Masucci GD, Nguyen XH, Santos ME, Wee HB (2019) Marine biodiversity research in the Ryukyu Islands, Japan: current status and trends. PeerJ. 12(7):e6532
Ricotta C, de Bello F, Moretti M, Caccianiga M, Cerabolini BEL, Pavoine S (2016) Measuring the functional redundancy of biological communities: a quantitative guide. Methods in Ecology and Evolution. 7(11):1386–1395
Rincón-Díaz MP, Pittman SJ, Arismendi I, Heppell SS (2018) Functional diversity metrics detect spatio-temporal changes in the fish communities of a Caribbean marine protected area. Ecosphere. 9(10):e02433
Rivadeneira M, Fernández M, Navarrete S (2002) Latitudinal trends of species diversity in rocky intertidal herbivore assemblages: spatial scale and the relationship between local and regional species richness. Marine Ecology Progress Series. 245:23–131
Rosenfeld JS (2002) Functional redundancy in ecology and conservation. Oikos. 98(1):156–162
Roy K, Jablonski D, Valentine JW (2000) Dissecting latitudinal diversity gradients: functional groups and clades of marine bivalves. Proceedings of the Royal Society of London Series B: Biological Sciences. 267(1440):293–299
Roy K, Jablonski D, Valentine JW, Rosenberg G (1998) Marine latitudinal diversity gradients: tests of causal hypotheses. Proceedings of the National Academy of Sciences 95(7):3699–3702
Rumm A, Foeckler F, Dziock F, Ilg C, Scholz M, Harris RMB, Gerisch M (2018) Shifts in mollusc traits following floodplain reconnection: testing the response of functional diversity components. Freshwater Biology. 63(6):505–517
Schoepf V, Herler J, Zuschin M (2010) Microhabitat use and prey selection of the coral-feeding snail Drupella cornus in the northern Red Sea. Hydrobiologia. 641(1):45–57
Schumm M, Edie SM, Collins KS, Gómez-Bahamón V, Supriya K, White AE, Price TD, Jablonski D (2019) Common latitudinal gradients in functional richness and functional evenness across marine and terrestrial systems. Proceedings of the Royal Society B: Biological Sciences. 286(1908):20190745
Sommer B, Harrison PL, Beger M, Pandolfi JM (2014) Trait-mediated environmental filtering drives assembly at biogeographic transition zones. Ecology. 95(4):1000–1009
Sommer B, Beger M, Harrison PL, Babcock RC, Pandolfi JM (2018) Differential response to abiotic stress controls species distributions at biogeographic transition zones. Ecography 41:478–490
Sommer B, Sampayo EM, Beger M, Harrison PL, Babcock RC, Pandolfi JM (2017) Local and regional controls of phylogenetic structure at the high-latitude range limits of corals. Proceedings of the Royal Society B: Biological Sciences. 284(1861):20170915
Song Y, Wang P, Li G, Zhou D (2014) Relationships between functional diversity and ecosystem functioning: a review. Acta Ecologica Sinica. 34(2):85–91
Stuart-Smith RD, Bates AE, Lefcheck JS, Duffy JE, Baker SC, Thomson RJ, Stuart-Smith JF, Hill NA, Kininmonth SJ, Airoldi L, Becerro MA, Campbell SJ, Dawson TP, Navarrete SA, Soler GA, Strain EMA, Willis TJ, Edgar GJ (2013) Integrating abundance and functional traits reveals new global hotspots of fish diversity. Nature. 501:539
Suthers IM, Young JW, Baird ME, Roughan M, Everett JD, Brassington GB, Byrne M, Condie SA, Hartog JR, Hassler CS, Hobday AJ, Holbrook NJ, Malcolm HA, Oke PR, Thompson PA, Ridgway K (2011) The strengthening East Australian Current, its eddies and biological effects — an introduction and overview. Deep Sea Research Part II: Topical Studies in Oceanography. 58(5):538–546
Teichert N, Lepage M, Sagouis A, Borja A, Chust G, Ferreira MT, Pasquaud S, Schinegger R, Segurado P, Argillier C (2017) Functional redundancy and sensitivity of fish assemblages in European rivers, lakes and estuarine ecosystems. Scientific Reports. 7(1):1
Tillin HM, Hiddink JG, Jennings S, Kaiser MJ (2006) Chronic bottom trawling alters the functional composition of benthic invertebrate communities on a sea-basin scale. Marine Ecology Progress Series. 318:31–45
Tyberghein L, Verbruggen H, Pauly K, Troupin C, Mineur F, De Clerck O (2012) Bio-ORACLE: a global environmental dataset for marine species distribution modelling. Global Ecology and Biogeography. 21(2):272–281
Valdovinos C, Navarrete SA, Marquet PA (2003) Mollusk species diversity in the Southeastern Pacific: why are there more species towards the pole? Ecography. 26(2):139–144
Valentine J, Roy K, Jablonski D (2002) Carnivore/non carnivore ratios in northeastern Pacific marine gastropods. Marine Ecology Progress Series 228:153–163
Vergés A, McCosker E, Mayer-Pinto M, Coleman MA, Wernberg T, Ainsworth T, Steinberg PD (2019) Tropicalisation of temperate reefs: implications for ecosystem functions and management actions. Functional Ecology. 33(6):1000–1013
Vergés A, Steinberg PD, Hay ME, Poore AGB, Campbell AH, Ballesteros E, Heck KL Jr, Booth DJ, Coleman MA, Feary DA, Figueira W, Langlois T, Marzinelli EM, Mizerek T, Mumby PJ, Nakamura Y, Roughan M, van Sebille E, Gupta A Sen, Smale DA, Tomas F, Wernberg T, Wilson SK (2014) The tropicalization of temperate marine ecosystems: climate-mediated changes in herbivory and community phase shifts. Proceedings of the Royal Society B-Biological Sciences. 281(1789):20140846
Veron JEN, Minchin PR (1992) Correlations between sea surface temperature, circulation patterns and the distribution of hermatypic corals of Japan. Continental Shelf Research. 12(7):835–857
Villéger S, Mason NWH, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology. 89(8):2290–2301
Villéger S, Brosse S, Mouchet M, Mouillot D, Vanni MJ (2017) Functional ecology of fish: current approaches and future challenges. Aquat Sci 79(4):783–801
Violle C, Navas M-L, Vile D, Kazakou E, Fortunel C, Hummel I, Garnier E (2007) Let the concept of trait be functional! Oikos. 116(5):882–892
Vroom PS, Braun CL (2010) Benthic composition of a healthy subtropical reef: baseline species-level cover, with an emphasis on algae, in the Northwestern Hawaiian Islands. PLoS ONE. 5(3):e9733
Wang X, Eijkemans M, Wallinga J, Biesbroek G, Trzciński K, Sanders E, Bogaert D (2012) Multivariate approach for studying interactions between environmental variables and microbial communities. PLoS One. 7:e50267
Wilson SK, Fisher R, Pratchett MS, Graham NAJ, Dulvy NK, Turner RA, Cakacaka A, Polunin NVC (2010) Habitat degradation and fishing effects on the size structure of coral reef fish communities. Ecological Applications. 20(2):442–451
Worm B, Barbier EB, Beaumont N, Duffy JE, Folke C, Halpern BS, Jackson JBC, Lotze HK, Micheli F, Palumbi SR, Sala E, Selkoe KA, Stachowicz JJ, Watson R (2006) Impacts of biodiversity loss on ocean ecosystem services. Science. 314(5800):787–790
Wu L, Cai W, Zhang L, Nakamura H, Timmermann A, Joyce T, McPhaden MJ, Alexander M, Qiu B, Visbeck M, Chang P, Giese B (2012) Enhanced warming over the global subtropical western boundary currents. Nature Climate Change. 2:161
Yamano H, Hori K, Yamauchi M, Yamagawa O, Ohmura A (2001) Highest-latitude coral reef at Iki Island. Japan. Coral Reefs. 20(1):9–12
Yamano H, Sugihara K, Nomura K (2011) Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophysical Research Letters. 38(4):1
Yatsu A, Chiba S, Yamanaka Y, Ito S, Shimizu Y, Kaeriyama M, Watanabe Y (2013) Climate forcing and the Kuroshio/Oyashio ecosystem. ICES Journal of Marine Science. 70(5):922–933
Zuschin M, Hohenegger J, Steininger F (2001) Molluscan assemblages on coral reefs and associated hard substrata in the northern Red Sea. Coral Reefs. 20(2):107–116
Acknowledgements
This research was funded by grants from the Japanese Society for the Promotion of Science (JSPS) ‘Zuno-Junkan’ grant entitled “Studies on origin and maintenance of marine biodiversity and systematic conservation planning” to JDR, the Australian Research Council Centre of Excellence for Environmental Decisions (CE110001014) and an EU Marie Slodowska Curie Fellowship (TRIM-DLV-747102) to MB, and a University of Sydney Fellowship to BS. We specifically thank Dr. T Naruse, University of the Ryukyus for accommodation at Iriomote-jima Island, Okinawa, as well as Mr. S Dewa, Diving Service Umiannai in Kagoshima and Mr. K Nomura, Kushimoto Marine Park Center in Wakayama for arranging field surveys. We also thank Diving Service Toneriko at Iriomote-jima Island, Okinoshima Diving Service Marine Snow at Tateyama, Nakagi Marine Center at Shimoda, Yakushima Diving Service Mori-to-Umi at Yakushima Island, and Relax Diving Service and Aquadive Koholloa at Amami-Oshima Island for logistical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Topic Editor Alastair Harborne
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Floyd, M., Mizuyama, M., Obuchi, M. et al. Functional diversity of reef molluscs along a tropical-to-temperate gradient. Coral Reefs 39, 1361–1376 (2020). https://doi.org/10.1007/s00338-020-01970-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-020-01970-2