Abstract
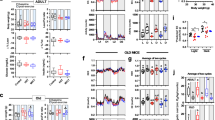
The main goal of the study was to explore the genetic architecture underlying muscle weight in old mice. Weight of soleus, tibialis anterior (TA), extensor digitorum longus (EDL), and gastrocnemius muscles was measured in the C57BL/6J (B6) and DBA/2J (D2) strains and derivative generations: a panel of the BXD recombinant inbred (RI) strains and a B6D2 F2 intercross at the age of 800 days. The between-strain difference in muscle weight (B6 > D2) ranged between 16% and 38%. Linkage analysis identified suggestive quantitative trait loci (QTL) on Chromosomes (Chr) 2, 6, 7, 8, 19, and X that influenced muscle weight in the 800-day-old group. Comparison of weights at 200, 500, and 800 days revealed a variable effect of age among the four muscles. Linkage analysis in the B6D2 F2 population combined across the three different age groups identified muscle-, sex-, and age-specific QTL on Chr 1, 2, 3, 5, 6, 8, 9, 11, 13, 17, X, and Y. Genetic factors that influence the rate of weight change (within-strain weight difference at two ages) over the lifespan of BXD RIs were mapped to the markers D2Mit369 and D3Mit130 at the genome-wide p < 0.05 for TA muscle in males (between 200 and 800 days) and females (between 500 and 800 days), respectively. Analysis of all age groups supported previous findings that the genetic effects may be muscle-, age-, and sex-specific.





Similar content being viewed by others
References
Arden NK, Spector TD (1997) Genetic influences on muscle strength, lean body mass, and bone mineral density: a twin study. J Bone Miner Res 12: 2076–2081
Brockmann GA, Kratzsch J, Haley CS, Renne U, Schwerin M, et al. (2000) Single QTL effects, epistasis, and pleiotropy account for two-thirds of the phenotypic F(2) variance of growth and obesity in DU6i x DBA/2 mice. Genome Research 10: 1941–1957
Broman KW (2001) Review of statistical methods for QTL map** in experimental crosses. Lab Anim (NY) 30: 44–52
Broman KW, Wu H, Sen S, Churchill GA (2003) R/qtl: QTL map** in experimental crosses. Bioinformatics 19: 889–890
Brooks SV, Faulkner JA (1988) Contractile properties of skeletal muscles from young, adult and aged mice. J Physiol 404: 71–82
Brooks SV, Faulkner JA (1991) Forces and powers of slow and fast skeletal muscles in mice during repeated contractions. J Physiol 436: 701–710
Carmelli D, Reed T (2000) Stability and change in genetic and environmental influences on hand-grip strength in older male twins. J Appl Physiol 89: 1879–1883
Cheverud JM, Routman EJ, Duarte FA, van Swinderen B, Cothran K, et al. (1996) Quantitative trait loci for murine growth. Genetics 142: 1305–1319
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait map**. Genetics 138: 963–871
Eddinger TJ, Moss RL, Cassens RG (1985) Fiber number and type composition in extensor digitorum longus, soleus, and diaphragm muscles with aging in Fisher 344 rats. J Histochem Cytochem 33: 1033–1041
Edstrom L, Larsson L (1987) Effects of age on contractile and enzyme-histochemical properties of fast- and slow-twitch single motor units in the rat. J Physiol 392: 129–145
Engel AG, Yamamoto M, Fischbeck KH. (1994) Dystrophinopathies. In Myology, Vol. 2, Engel AG, Franzini-Armstrong C (eds.) (New York: McGraw– Hill), pp 1133–1187
Florini JR, Ewton DZ (1989) Skeletal muscle fiber types and myosin ATPase activity do not change with age or growth hormone administration. J Gerontol 44: B110–B117
Frederiksen H, Gaist D, Petersen HC, Hjelmborg J, McGue M, et al. (2002) Hand grip strength: a phenotype suitable for identifying genetic variants affecting mid- and late-life physical functioning. Genet Epidemiol 23: 110–122
Georges M, Charlier C, Cockett N (2003) The callipyge locus: evidence for the trans interaction of reciprocally imprinted genes. Trends Genet 19: 248–252
Griggs RC, Markesbery WR (1994) Distal Myopathies. In Myology, Vol. 2, Engel AG, Franzini-Armstrong C (eds.) (New York: McGraw–Hill), pp 1246–1257
Grobet L (1997) A deletion in the bovine myostatin gene causes the double-muscled phenotype in cattle. [see comments.]. Nat Genet 17: 71–74
Hadchouel J, Carvajal JJ, Daubas P, Bajard L, Chang T, et al. (2003) Analysis of a key regulatory region upstream of the Myf5 gene reveals multiple phases of myogenesis, orchestrated at each site by a combination of elements dispersed throughout the locus. Development 130: 3415–3426
Hartl DL, Jones EW (2002) Human karyotypes and chromosome behaviour. In Essential genetics: a genomics perspective (Sudbury, MA: Jones and Bartlett Publishers), pp 167–207
Hennig R, Lomo T (1985) Firing patterns of motor units in normal rats. Nature 314: 164–166
Hepple RT, Ross KD, Rempfer AB (2004) Fiber atrophy and hypertrophy in skeletal muscles of late middle-aged Fischer 344 x Brown Norway F1-hybrid rats. J Gerontol A Biol Sci Med Sci 59: 108–117
Janssen I, Heymsfield SB, Wang ZM, Ross R (2000) Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J Appl Physiol 89: 81–88
Kambadur R, Sharma M, Smith TP, Bass JJ (1997) Mutations in myostatin (GDF8) in double-muscled Belgian Blue and Piedmontese cattle. Genome Res 7: 910–916
Lander E, Kruglyak L (1995) Genetic dissection of complex traits: guidelines for interpreting and reporting linkage results. Nat Genet 11: 241–247
Larsson L, Ansved T (1988) Effects of age on the motor unit. A study on single motor units in the rat. Ann NY Acad Sci 515: 303–313
Larsson L, Ansved T (1995) Effects of ageing on the motor unit. Prog Neurobiol 45: 397–458
Larsson L, Salviati G (1989) Effects of age on calcium transport activity of sarcoplasmic reticulum in fast- and slow-twitch rat muscle fibres. J Physiol 419: 253–264
Larsson L, Biral D, Campione M, Schiaffino S (1993) An age-related type IIB to IIX myosin heavy chain switching in rat skeletal muscle. Acta Physiol Scand 147: 227–234
Lionikas A, Blizard DA, Vandenbergh DJ, Glover MG, Stout JT, et al. (2003) Genetic architecture of fast- and slow-twitch skeletal muscle weight in 200-day-old mice of the C57BL/6J and DBA/2J lineage. Physiol Genomics 16: 141–152
Lionikas A, Blizard DA, Gerhard GS, Vandenbergh DJ, Stout JT, et al. (2005) Genetic determinants of weight of fast- and slow-twitch skeletal muscle in 500-day old mice of the C57BL/6J and DBA/2J lineage. Physiol Genomics 21: 184–192
Lynch NA, Metter EJ, Lindle RS, Fozard JL, Tobin JD, et al. (1999) Muscle quality. I. Age-associated differences between arm and leg muscle groups. J Appl Physiol 86: 188–194
Mahoney J, Sager M, Dunham NC, Johnson J (1994) Risk of falls after hospital discharge. J Am Geriatr Soc 42: 269–274
Malek M, Dekkers JC, Lee HK, Baas TJ, Prusa K, et al. (2001) A molecular genome scan analysis to identify chromosomal regions influencing economic traits in the pig. II. Meat and muscle composition. Mamm Genome 12: 637–645
Masinde GL, Li X, Gu W, Hamilton–Ulland M, Mohan S, et al. (2002) Quantitative trait loci that harbor genes regulating muscle size in (MRL/MPJ × SJL/J) F(2) mice. Funct Integr Genomics 2: 120–125
McCarter R, McGee J (1987) Influence of nutrition and aging on the composition and function of rat skeletal muscle. J Gerontol 42: 432–441
McPherron AC, Lee SJ (1997) Double muscling in cattle due to mutations in the myostatin gene. Proc Natl Acad Sci USA 94: 12457–12461
Milan D, Bidanel JP, Iannuccelli N, Riquet J, Amigues Y, et al. (2002) Detection of quantitative trait loci for carcass composition traits in pigs. Genet Sel Evol 34: 705–728
Reed RL, Pearlmutter L, Yochum K, Meredith KE, Mooradian AD (1991a) The relationship between muscle mass and muscle strength in the elderly. J Am Geriatr Soc 39: 555–561
Reed T, Fabsitz RR, Selby JV, Carmelli D (1991b) Genetic influences and grip strength norms in the NHLBI twin study males aged 59–69. Ann Hum Biol 18: 425–432
Rocha JL, Eisen EJ, Van Vleck LD, Pomp D (2004) A large-sample QTL study in mice: II. Body composition. Mamm Genome 15: 100–113
Schuelke M, Wagner KR, Stolz LE, Hubner C, Riebel T, et al. (2004) Myostatin mutation associated with gross muscle hypertrophy in a child. N Engl J Med 350: 2682–2688
Summerbell D, Ashby PR, Coutelle O, Cox D, Yee S, et al. (2000) The expression of Myf5 in the develo** mouse embryo is controlled by discrete and dispersed enhancers specific for particular populations of skeletal muscle precursors. Development 127: 3745–3757
Szabo G, Dallmann G, Muller G, Patthy L, Soller M, et al. (1998) A deletion in the myostatin gene causes the compact (Cmpt) hypermuscular mutation in mice. Mamm Genome 9: 671–672
Van Laere AS, Nguyen M, Braunschweig M, Nezer C, Collette C, et al. (2003) A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 425: 832–836
Vandenbergh DJ, Heron K, Peterson R, Shpargel KB, Woodroffe A, et al. (2003) Simple tests to detect errors in high-throughput genotype data in the molecular laboratory. J Biomol Tech 14: 9–16
Varga L, Szabo G, Darvasi A, Muller G, Sass M, et al. (1997) Inheritance and map** of Compact (Cmpt), a new mutation causing hypermuscularity in mice. Genetics 147: 755–764
Varga L, Muller G, Szabo G, Pinke O, Korom E, et al. (2003) Map** modifiers affecting muscularity of the myostatin mutant (Mstn(Cmpt-dl1Abc)) compact mouse. Genetics 165: 257–267
Varona L, Ovilo C, Clop A, Noguera JL, Perez-Enciso M, et al. (2002) QTL map** for growth and carcass traits in an Iberian by Landrace pig intercross: additive, dominant and epistatic effects. Genet Res 80: 145–154
Weindruch R, Kayo T, Lee CK, Prolla TA (2001) Microarray profiling of gene expression in aging and its alteration by caloric restriction in mice. J Nutr 131: 918S–923S
Weindruch R, Kayo T, Lee CK, Prolla TA (2002) Gene expression profiling of aging using DNA microarrays. Mech Ageing Dev 123: 177–193
Willems ME, Stauber WT (2000) Changes in force by repeated stretches of skeletal muscle in young and old female Sprague Dawley rats. Aging (Milano) 12: 478–481
Acknowledgments
The authors are grateful to Dr. F. Yu and Dr. N. Sharkey, Ms. C. Vandenberg, A. Lionikiene, and Mr. M. G. Glover for excellent help with the tissue harvesting, and Ms. S. Lingenfelter and K. Seymour for excellent technical assistance. The study was supported by grants from the National Institutes of Health (AG14731, AG00276, AR45627, AR47318), the European Commission (QLK6-CT-2000-0530), and the Swedish Medical Research Council (8651).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lionikas, A., Blizard, D.A., Vandenbergh, D.J. et al. Genetic determinants of weight of fast- and slow-twitch skeletal muscles in old mice. Mamm Genome 17, 615–628 (2006). https://doi.org/10.1007/s00335-005-0177-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-005-0177-x