Abstract
Objectives
To analyze the value of amide proton transfer-weighted imaging (APTWI) and diffusion kurtosis imaging (DKI) in differentiating cervical cancer (CC) pathological type, grade, and stage.
Methods
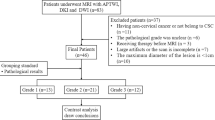
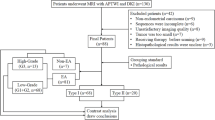
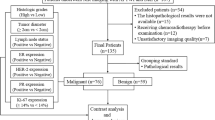
One hundred and twelve women underwent pelvic APTWI and DKI. The magnetization transfer ratio asymmetry (MTRasym, 3.5 ppm), apparent kurtosis coefficient (Kapp), and non-Gaussian diffusion coefficient (Dapp) were calculated by histological subtype, grade, and stage. The differences, efficacy, and correlation between parameters were determined.
Results
The MTRasym(3.5 ppm) and Dapp values of the adenocarcinoma (CA) group were higher than those of the cervical squamous carcinoma (CSC) group, while the Kapp values were lower than those of the CSC group. The MTRasym(3.5 ppm) and Kapp values of the high-grade group were higher than those of the low-grade group, while the Dapp values were lower than those of the low-grade group. The Dapp values of the advanced-stage group were lower than those of the early-stage group, while the Kapp values were greater than those of the early-stage group. The Kapp showed the highest efficacy in differentiating CSC and CA, high- and low-grade CC, and advanced- and early-stage CC. In the CSC and CA groups, both the Kapp and Dapp were highly correlated with pathological grade, and the MTRasym(3.5 ppm) was weakly correlated with pathological grade. The Kapp, Dapp, and MTRasym(3.5 ppm) were all weakly correlated with pathological stage.
Conclusion
Both DKI and APTWI can be used in preliminary evaluations of CC, but DKI has advantages in the identification of pathological type, grade, and stage.
Key Points
• PTWI and DKI provide new information regarding cervical cancer.
• MTRasym(3.5 ppm), D app , and K app are valid parameters to characterize tissue microstructure.
• DKI is superior to APTWI in the study of cervical cancer.





Similar content being viewed by others
Abbreviations
- APTWI:
-
Amide proton transfer-weighted imaging
- AUC:
-
Area under the curve
- CA:
-
Cervical adenocarcinoma
- CC:
-
Cervical cancer
- CSC:
-
Cervical squamous carcinoma
- D app :
-
Mean diffusivity
- DKI:
-
Diffusion kurtosis imaging
- FIGO:
-
International Federation of Gynecology and Obstetrics
- ICC:
-
Intraclass correlation coefficient
- K app :
-
Mean kurtosis
- MRI:
-
Magnetic resonance imaging
- MTRasym(3.5 ppm):
-
The asymmetric magnetization transfer ratio at 3.5 ppm
- RF:
-
Radio frequency
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
References
Small W, Bacon MA, Bajaj A et al (2017) Cervical cancer: a global health crisis. Cancer 123:2404–2412
Berek JS, Matsuo K, Grubbs BH et al (2019) Multidisciplinary perspectives on newly revised 2018 FIGO staging of cancer of the cervix uteri. J Gynecol Oncol 30:e40
Jonska-Gmyrek J, Gmyrek L, Zolciak-Siwinska A, Kowalska M, Kotowicz B (2019) Adenocarcinoma histology is a poor prognostic factor in locally advanced cervical cancer. Curr Med Res Opin 35:595–601
Jung EJ, Byun JM, Kim YN et al (2017) Cervical adenocarcinoma has a poorer prognosis and a higher propensity for distant recurrence than squamous cell carcinoma. Int J Gynecol Cancer 27:1228–1236
Fujiwara H, Yokota H, Monk B et al (2014) Gynecologic Cancer InterGroup (GCIG) consensus review for cervical adenocarcinoma. Int J Gynecol Cancer 24:S96–S101
Pol FJ, Zusterzeel PL, van Ham MA, Kuijpers DA, Bulten J, Massuger LF (2015) Satellite lymphovascular space invasion: an independent risk factor in early stage cervical cancer. Gynecol Oncol 138:579–584
Shoji T, Takatori E, Saito T et al (2013) Neoadjuvant chemotherapy using platinum- and taxane-based regimens for bulky stage Ib2 to IIb non-squamous cell carcinoma of the uterine cervix. Cancer Chemother Pharmacol 71:657–662
Kalliala I, Anttila A, Nieminen P, Halttunen M, Dyba T (2014) Pregnancy incidence and outcome before and after cervical intraepithelial neoplasia: a retrospective cohort study. Cancer Med 3:1512–1516
Moukarzel LA, Angarita AM, VandenBussche C et al (2017) Preinvasive and invasive cervical adenocarcinoma: preceding low-risk or negative pap result increases time to diagnosis. J Low Genit Tract Dis 21:91–96
Jensen JH, Helpern JA, Ramani A, Lu H, Kaczynski K (2005) Diffusional kurtosis imaging: the quantification of non-Gaussian water diffusion by means of magnetic resonance imaging. Magn Reson Med 53:1432–1440
Jensen JH, Helpern JA (2010) MRI quantification of non-Gaussian water diffusion by kurtosis analysis. NMR Biomed 23:698–710
Yin J, Sun H, Wang Z, Ni H, Shen W, Sun PZ (2018) Diffusion kurtosis imaging of acute infarction: comparison with routine diffusion and follow-up MR imaging. Radiology 287:651–657
Hempel JM, Schittenhelm J, Bisdas S et al (2018) In vivo assessment of tumor heterogeneity in WHO 2016 glioma grades using diffusion kurtosis imaging: diagnostic performance and improvement of feasibility in routine clinical practice. J Neuroradiol 45:32–40
Sun K, Chen X, Chai W et al (2015) Breast cancer: diffusion kurtosis MR imaging-diagnostic accuracy and correlation with clinical-pathologic factors. Radiology 277:46–55
Suo S, Chen X, Wu L et al (2014) Non-Gaussian water diffusion kurtosis imaging of prostate cancer. Magn Reson Imaging 32:421–427
Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090
Zhou J, Lal B, Wilson DA, Laterra J, van Zijl PC (2003) Amide proton transfer (APT) contrast for imaging of brain tumors. Magn Reson Med 50:1120–1126
Jeong HK, Han K, Zhou J et al (2017) Characterizing amide proton transfer imaging in haemorrhage brain lesions using 3T MRI. Eur Radiol 27:1577–1584
Joo B, Han K, Choi YS et al (2018) Amide proton transfer imaging for differentiation of benign and atypical meningiomas. Eur Radiol 28:331–339
Jiang S, Eberhart CG, Lim M et al (2019) Identifying recurrent malignant glioma after treatment using amide proton transfer-weighted MR imaging: a validation study with image-guided stereotactic biopsy. Clin Cancer Res 25:552–561
Ohno Y, Yui M, Koyama H et al (2016) Chemical exchange saturation transfer MR imaging: preliminary results for differentiation of malignant and benign thoracic lesions. Radiology 279:578–589
Dula AN, Dewey BE, Arlinghaus LR et al (2015) Optimization of 7-T chemical exchange saturation transfer parameters for validation of glycosaminoglycan and amide proton transfer of fibroglandular breast tissue. Radiology 275:255–261
Takayama Y, Nishie A, Sugimoto M et al (2016) Amide proton transfer (APT) magnetic resonance imaging of prostate cancer: comparison with Gleason scores. MAGMA 29:671–679
Wang P, Thapa D, Wu G, Sun Q, Cai H, Tuo F (2018) A study on diffusion and kurtosis features of cervical cancer based on non-Gaussian diffusion weighted model. Magn Reson Imaging 47:60–66
Li B, Sun H, Zhang S, Wang X, Guo Q (2019) The utility of APT and IVIM in the diagnosis and differentiation of squamous cell carcinoma of the cervix: a pilot study. Magn Reson Imaging 63:105–113
Dappa E, Elger T, Hasenburg A, Düber C, Battista MJ, Hötker AM (2017) The value of advanced MRI techniques in the assessment of cervical cancer: a review. Insights Imaging 8:471–481
Rosenkrantz AB, Padhani AR, Chenevert TL et al (2015) Body diffusion kurtosis imaging: basic principles, applications, and considerations for clinical practice. J Magn Reson Imaging 42:1190–1202
Tee YK, Donahue MJ, Harston GW, Payne SJ, Chappell MA (2014) Quantification of amide proton transfer effect pre- and post-gadolinium contrast agent administration. J Magn Reson Imaging 40:832–838
Zheng S, van der Bom IM, Zu Z, Lin G, Zhao Y, Gounis MJ (2014) Chemical exchange saturation transfer effect in blood. Magn Reson Med 71:1082–1092
Argüello-Ramírez J, Pérez-Cárdenas E, Delgado-Chávez R, Solorza-Luna G, Villa-Treviño S, Arenas-Huertero F (2004) Matrix metalloproteinases-2, -3, and -9 secreted by explants of benign and malignant lesions of the uterine cervix. Int J Gynecol Cancer 14:333–340
Zou T, Yu H, Jiang C et al (2018) Differentiating the histologic grades of gliomas preoperatively using amide proton transfer-weighted (APTW) and intravoxel incoherent motion MRI. NMR Biomed 31:e3850
Takayama Y, Nishie A, Togao O et al (2018) Amide proton transfer MR imaging of endometrioid endometrial adenocarcinoma: association with histologic grade. Radiology 286:909–917
Tang Y, Dundamadappa SK, Thangasamy S et al (2014) Correlation of apparent diffusion coefficient with Ki-67 proliferation index in grading meningioma. AJR Am J Roentgenol 202:1303–1308
Whittaker CS, Coady A, Culver L, Rustin G, Padwick M, Padhani AR (2009) Diffusion-weighted MR imaging of female pelvic tumors: a pictorial review. Radiographics 29:759–774 discussion 774-778
Togao O, Yoshiura T, Keupp J et al (2014) Amide proton transfer imaging of adult diffuse gliomas: correlation with histopathological grades. Neuro Oncol 16:441–448
Togao O, Hiwatashi A, Yamashita K et al (2017) Grading diffuse gliomas without intense contrast enhancement by amide proton transfer MR imaging: comparisons with diffusion- and perfusion-weighted imaging. Eur Radiol 27:578–588
Yue W, Meng N, Wang J et al (2019) Comparative analysis of the value of diffusion kurtosis imaging and diffusion-weighted imaging in evaluating the histological features of endometrial cancer. Cancer Imaging 19:9
Acknowledgments
We acknowledge the support received from the National Natural Science Foundation of China and Henan Medical Science and Technology Research Program. In addition, Nan Meng wants to thank, in particular, the patience, care, and support from **g Sun over the past years. I love you forever.
Funding
This study has received funding by the National Natural Science Foundation of China (grants 81271565 and 31470047), and the National Clinical Key Specialty of China and the Henan Medical Science and Technology Research Program (grants 2018020357 and 2018020367).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Meiyun Wang.
Conflict of interest
One of the authors of this manuscript (Kaiyu Wang) is an employee of GE Healthcare. The remaining authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Some study subjects or cohorts have been previously reported in Magnetic Resonance Imaging.
Methodology
• Prospective
• Diagnostic or prognostic study
• Performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meng, N., Wang, X., Sun, J. et al. Application of the amide proton transfer-weighted imaging and diffusion kurtosis imaging in the study of cervical cancer. Eur Radiol 30, 5758–5767 (2020). https://doi.org/10.1007/s00330-020-06884-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-06884-9