Abstract
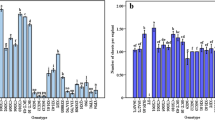
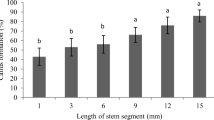
Factors influencing reliable shoot regeneration from leaf explants of rapeseed (Brassica napus L.) were examined. Addition of AgNO3 to callus induction medium was significantly effective for shoot regeneration in all three genotypes initially tested. When 48 genotypes subsequently were surveyed, a large variation of shoot regenerability was observed, ranging from 100 to 0% in frequency of bud formation and from 7.5 to 0 in the number of buds per explant. A significant correlation (r=0.84) was observed between the frequency of bud formation and the number of buds per explant. The shoot regenerability from leaf explants was not related to that from cotyledonary explants (r=0.28). Histological observations showed that an organized structure developed from calluses produced at vascular bundle tissues after 7 days of culture on callus induction medium, and they developed shoot apical meristems one week after transfer onto shoot induction medium. Regenerated plantlets were obtained 2 months after the initiation of culture and they normally flowered and set seeds. No alterations of morphology or DNA contents were observed in regenerated plants and their S1 progenies.





Similar content being viewed by others
Abbreviations
- BAs :
-
6-benzylaminopurine
- NAAs :
-
1-naphthaleneacetic acid.
- PGRs :
-
plant growth regulators.
- FCMs :
-
flow cytometry
References
Akasaka Y, Daimon H, Mii M (2000) Improved plant regeneration from cultured leaf segments in peanut (Arachis hypogaea L.) by limited exposure to thidiazuron. Plant Sci 156:169–175
Boulter ME, Croy E, Simpson P, Shields R, Croy PRD, Shirsat AH (1990) Transformation of Brassica napus L. (oilseed rape) using Agrobacterium tumefaciense and Agrobacterium rhizogenes a comparison. Plant Sci 70:91–99
Burnett L, Arnoldo M, Yarrow S, Huang B (1994) Enhancement of shoot regeneration from cotyledon explants of Brassica rapa ssp. oleifera through pre-treatment with auxin and cytokinin and use of ethylene inhibitors. Plant Cell Tissue Organ Culture 37:253–256
Chi GL, Pua EC, Goh CJ (1991) Role of ethylene on de novo shoot regeneration from cotyledonary explants of Brassica campestris ssp. Pekinensis (Lour) Olesson in vitro. Plant Physiol 196:178–183
De Block M, De Brouwer D, Tenning P (1989) Transformation of Brassica napus and Brassica oleracea using Agrobacterium tumefaciens and the expression of the bar and neo genes in the transgenic plants. Plant Physiol 91:694–701
Dunwell JM (1981) In vitro regeneration from excised leaf discs of three Brassica species. J Exp Bot 32:789–799
Ezura H, Yuhashi K, Yasuta T, Minamisawa K (2000) Effect of ethylene on Agrobacterium tumefaciens-mediated gene transfer to melon. Plant Breed 119:75–79
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures if soybean root cells. Exp Cell Res 50:151–158
Hachey JE, Sharma KK, Moloney MM (1991) Efficient shoot regeneration of Brassica campestris using cotyledon explants cultured in vitro. Plant Cell Rep 9:549–554
Herve P, Jauneau A, Paques M, Marien J-N, Boudet AM, Teulieres C (2001) A procedure for shoot organogenesis in vitro from leaves and nodes of an elite Eucalyptus gunnii clone: comparative histology. Plant Sci 161:645–653
Khan MR, Rashid H, Ansar M, Chaudry Z (2003) High frequency shoot regeneration and Agrobacterium-mediated DNA transfer in Canola (Brassica napus). Plant Cell Tissue Organ Culture 75:223–231
Moleney MM, Walker JM, Sharma KK (1989) High-efficiency transformation of Brassica napus using Agrobacterium vectors. Plant Cell Rep 8:238–242
Murakami T, Ono Y, Takahata Y (1995) Phytohormonal and genotypic factors affecting shoot regeneration from cotyledonary explant of radish (Raphanus sativus L.). Plant Tissue Culture Lett 12:321–323
Ono Y, Takahata Y, Kaizuma N (1994) Effect of genotype on shoot regeneration from cotyledonary explants of rapeseed (Brassica napus L.). Plant Cell Rep 14:13–17
Ono Y, Takahata Y (2000) Genetic analysis of shoot regeneration from cotyledonary explants in Brassica napus. Theor Appl Genet 100:895–898
Pechan PM, Keller WA (1998) Identification of potentially embryogenic microspores of Brassica napus L. Physiol Plant 14:377–384
Pieron S, Boxus P, Dekegek D (1998) Histological study of nodule morphogenesis from Cichorium intybus L. leaves cultivated in vitro. In Vitro Cell Biol-Plant 34:87–93
Pua EC, Chi GL (1993) De novo shoot morphogenesis and plant growth of mustard (Brassica juncea) in vitro relation to ethylene. Physiol Plant 88:467–474
Pua EC, Sim GE, Chi GL, Kong LF (1996) Synergistic effect of ethylene inhibitors and putrescine on shoot regeneration from hypocotyls explants of Chinese radish (Raphanus sativus L. var longipinnatus Bailey) in vitro. Plant Cell Rep 15:685–690
Pua EC, Lim TS (2004) Transgenic oilseed Brassicas. In: Pua EC, Douglad CJ (eds) Biotechnology in agriculture and forestry, vol 54. Springer-Verlag, Berlin Heidelberg, pp 195–224
Shu W, Loh CS (1991) Secondary embryogenesis from thin cell layers of Brassica napus ssp. oleifera. New Phytol 119:427–432
Stringam GR (1979) Regeneration in leaf-callus cultures of haploid rapeseed (Brassica napus L.) Z. Pflanzenphysiology 92:459–462
Takasaki T, Hatakeyama K, Ojima K, Watanabe M, Toriyama K, Hinata K (1996) Effects of various factors (hormone combinations, genotypes and antibiotics) on shoot regeneration from cotyledon explants in Brassica rapa L. Plant Tissue Culture Lett 13:177–180
Zhang FL, Takahata Y, Xu JB (1998) Medium and genotype factors influencing shoot regeneration from cotyledonary explants of Chinese cabbage (Brassica campestris L. ssp. pekinensis). Plant Cell Rep 17:780–786
Acknowledgements
We thank Dr Y. Ono (Fukushima Fruit Trees Experiment Station) for providing data and Dr Y. Sasaki (Genesis Research Institute, Inc.) and Dr M. Watanabe (Iwate University) for their critical comments on this experiment. We thank Dr K. Mishiba (Iwate Biotechnology Research Center) for his assistance in FCM analysis
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Ozias-Akins
Rights and permissions
About this article
Cite this article
Akasaka-Kennedy, Y., Yoshida, H. & Takahata, Y. Efficient plant regeneration from leaves of rapeseed (Brassica napus L.): the influence of AgNO3 and genotype. Plant Cell Rep 24, 649–654 (2005). https://doi.org/10.1007/s00299-005-0010-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-005-0010-8