Abstract
Purpose
LY2334737 is an oral gemcitabine prodrug. This Phase I study assessed the safety and tolerability of LY2334737 in Japanese patients with solid tumors and evaluated pharmacokinetics (PK), pharmacodynamics, and antitumor activity.
Methods
Patients with advanced/metastatic solid tumors received escalating doses of LY2334737 once daily for 14 days, followed by a 7-day drug-free period. Cycles were repeated until discontinuation criteria were met.
Results
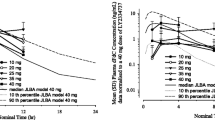
Of 13 patients treated, 3 received 20 mg/day, 6 received 30 mg/day, 4 received 40 mg/day. On the 40 mg dose, 3 patients experienced dose-limiting toxicities (DLTs): hepatic toxicities (e.g., Grade [G]3/4 transaminase and G1–3 bilirubin elevation) and G4 thrombocytopenia; all 3 showed features of disseminated intravascular coagulation. One additional DLT occurred on the 30 mg dose (G3 transaminase elevation). Exploratory pharmacogenetic analyses identified a genetic variation in the CES2 gene potentially associated with these DLTs. PK data showed no clear relationship between the AUC of gemcitabine and its incorporation into leukocyte DNA; 2 of the 3 DLT patients had high incorporation. Two patients (30 mg/day) achieved stable disease with progression-free survival lasting 135 and 155 days.
Conclusions
LY2334737 was tolerated by Japanese patients up to 30 mg/day. The toxicities observed at the 40 mg dose may require the development of alternative dosing schedules.


Similar content being viewed by others
References
Gemzar Summary of Product Characteristics, 31 August 2011. http://www.medicines.ie/medicine/8346/SPC/Gemzar/. Accessed 11 Feb 2013
Grunewald R, Kantarjian H, Keating MJ, Abbruzzese J, Tarassoff P, Plunkett W (1990) Pharmacologically directed design of the dose rate and schedule of 2′,2′-difluorodeoxycytidine (Gemcitabine) administration in leukemia. Cancer Res 50:6823–6826
Patel SR, Gandhi V, Jenkins J, Papadopolous N, Burgess MA, Plager C, Plunkett W, Benjamin RS (2001) Phase II clinical investigation of gemcitabine in advanced soft tissue sarcomas and window evaluation of dose rate on gemcitabine triphosphate accumulation. J Clin Oncol 19:3483–3489
Tempero M, Plunkett W, Ruiz Van Haperen V, Hainsworth J, Hochster H, Lenzi R, Abbruzzese J (2003) Randomized phase II comparison of dose-intense gemcitabine: thirty-minute infusion and fixed dose rate infusion in patients with pancreatic adenocarcinoma. J Clin Oncol 21:3402–3408
Gandhi V, Plunkett W, Du M, Ayres M, Estey EH (2002) Prolonged infusion of gemcitabine: clinical and pharmacodynamic studies during a phase I trial in relapsed acute myelogenous leukemia. J Clin Oncol 20:665–673
Abbruzzese JL, Grunewald R, Weeks EA, Gravel D, Adams T, Nowak B, Mineishi S, Tarassoff P, Satterlee W, Raber MN et al (1991) A phase I clinical, plasma, and cellular pharmacology study of gemcitabine. J Clin Oncol 9:491–498
Sakamoto H, Kitano M, Suetomi Y, Takeyama Y, Ohyanagi H, Nakai T, Yasuda C, Kudo M (2006) Comparison of standard-dose and low-dose gemcitabine regimens in pancreatic adenocarcinoma patients: a prospective randomized trial. J Gastroenterol 41:70–76
Takahashi Y, Mai M, Sawabu N, Nishioka K (2005) A pilot study of individualized maximum repeatable dose (iMRD), a new dose finding system, of weekly gemcitabine for patients with metastatic pancreas cancer. Pancreas 30:206–210
Pratt SE, Durland-Busbice S, Shepard RL, Donoho GP, Starling J, Wickremsinhe ER Mr, Perkins EJ, Dantzig AH (2013) Efficacy of low-dose oral metronomic dosing of the prodrug of gemcitabine, LY2334737, in human tumor xenografts. Mol Cancer Ther 12:481–490
Veltkamp SA, Jansen RS, Callies S, Pluim D, Visseren-Grul CM, Rosing H, Kloeker-Rhoades S, Andre VA, Beijnen JH, Slapak CA, Schellens JH (2008) Oral administration of gemcitabine in patients with refractory tumors: a clinical and pharmacologic study. Clin Cancer Res 14:3477–3486
Shipley LA, Brown TJ, Cornpropst JD, Hamilton M, Daniels WD, Culp HW (1992) Metabolism and disposition of gemcitabine, and oncolytic deoxycytidine analog, in mice, rats, and dogs. Drug Metab Dispos 20:849–855
Moysan E, Bastiat G, Benoit J-P (2013) Gemcitabine versus modified gemcitabine: a review of several promising chemical modifications. Mol Pharm 10:430–444
Bender DM, Bao J, Dantzig AH, Diseroad WD, Law KL, Magnus NA, Peterson JA et al (2009) Synthesis, crystallization, and biological evaluation of an orally active prodrug of gemcitabine. J Med Chem 52:6958–6961
Koolen SL, Witteveen PO, Jansen RS, Langenberg MH, Kronemeijer RH, Nol A, Garcia-Ribas I, Callies S, Benhadji KA, Slapak CA, Beijnen JH, Voest EE, Schellens JH (2011) Phase I study of oral gemcitabine prodrug (LY2334737) alone and in combination with erlotinib in patients with advanced solid tumors. Clin Cancer Res 17:6071–6082
Pratt SE, Durland-Busbice S, Shepard RL, Heinz-Taheny K, Iversen PW, Dantzig AH (2013) Human carboxylesterase 2 hydrolyzes the prodrug of gemcitabine (LY2334737) and confers prodrug sensitivity to cancer cells. Clin Cancer Res 19:1159–1168
Pratt SE, Durland-Busbice S, Shepard RL, Heinz-Taheny K, Iversen PW, Dantzig AH (2010) Identification of carboxylesterase 2 as a hydrolase that cleaves the prodrug of gemcitabine LY2334737. Abstr Clin Cancer Res 16(14 Suppl):B36
Xu G, Zhang W, Ma MK, McLeod HL (2002) Human Carboxylesterase 2 Is Commonly Expressed in Tumor Tissue and Is Correlated with Activation of Irinotecan. Clin Cancer Res 8:2605–2611
Le Tourneau C, Lee JJ, Siu LL (2009) Dose escalation methods in phase I cancer clinical trials. J Natl Cancer Inst 101:708–720
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst 92:205–216
Veltkamp SA, Hillebrand MJ, Rosing H, Jansen RS, Wickremsinhe ER, Perkins EJ, Schellens JH, Beijnen JH (2006) Quantitative analysis of gemcitabine triphosphate in human peripheral blood mononuclear cells using weak anion-exchange liquid chromatography coupled with tandem mass spectrometry. J Mass Spectrom 41:1633–1642
Wickremsinhe ER, Lutzke BS, Jones BR, Schultz GA, Freeman AB, Pratt SE, Bones AM, Ackermann BL (2010) Quantification of gemcitabine incorporation into human DNA by LC/MS/MS as a surrogate measure for target engagement. Anal Chem 82:6576–6583
Ministry of Health, Labour and Welfare of Japan (MHLW). Pharmaceuticals and medical devices safety information [MHLW Web site] [in Japanese]. http://www1.mhlw.go.Jp/houdou/1205/h05261_a_15.html#12. Accessed 10 June 2008
Ichikawa T, Sato H, Kaira K, Oh-I S, Kakizaki S, Sato K, Takagi H, Mori M (2008) Prolonged intrahepatic cholestasis after exposure to loxoprofen. Clin Ther 30:2402–2406
Veltkamp SA, Pluim D, van Eijndhoven MA, Bolijn MJ, Ong FH, Govindarajan R, Unadkat JD, Beijnen JH, Schellens JH (2008) New insights into the pharmacology and cytotoxicity of gemcitabine and 2′,2′-difluorodeoxyuridine. Mol Cancer Ther 7:2415–2425
Sugiyama E, Kaniwa N, Kim SR, Kikura-Hanajiri R, Hasegawa R, Maekawa K, Saito Y, Ozawa S, Sawada J, Kamatani N, Furuse J, Ishii H, Yoshida T, Ueno H, Okusaka T, Saijo N (2007) Pharmacokinetics of gemcitabine in Japanese cancer patients: The impact of a cytidine deaminase polymorphism. J Clin Oncol 25:32–42
Holzer TR, Heinz-Taheny K, Ballard DW, Powell EL, Dolled-Filhart MP, Christiansen J, Gustavson M, Biryukov J, Virayah J, Pratt SE, Shepard RL, Durland-Busbice SC, Dantzig AH (2010) Potential for patient tailoring for prodrug of gemcitabine (LY2334737) by assessment of carboxylesterase II expression in solid tumor biopsies. Abstr Clin Cancer Res 16(14 Suppl):B31
Faivre S, Olszanski A, Weigang-Koehler K, Riess H, Peng G, Callies S, Benhadji KA, Raymond E (2012) Phase I and pharmacokinetic (PK)/pharmacodynamic (PD) study of LY2334737, an oral gemcitabine prodrug, in patients (pts) with advanced solid tumors. Abstr J Clin Oncol 30(suppl):abstr 2554
Stuurman FE, Voest EE, Awada A, Witteveen PO, Bergeland T, Hals PA, Rasch W, Schellens JH, Hendlisz A (2013) Phase I study of oral CP-4126, a gemcitabine derivative, in patients with advanced solid tumors. Invest New Drugs (Epub ahead of print)
Acknowledgments
We would like to thank all patients for participating in the study. Karin Helsberg and Sarah Richardson, Trilogy Writing and Consulting, Frankfurt, Germany, provided medical writing support on behalf of Eli Lilly and Company. This work was supported by Eli Lilly and Company, Indianapolis, IN.
Conflict of interest
K.U., R.S., and T.M. are employees of Eli Lilly Japan, C.S. and K.A.B. are employees of Eli Lilly and Company. N.Y., H.N., Y.Y., K.U., and T.M. have no conflicts of interest to declare. T.T. has received honoraria from Eli Lilly Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamamoto, N., Nokihara, H., Yamada, Y. et al. Phase I study of oral gemcitabine prodrug (LY2334737) in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol 71, 1645–1655 (2013). https://doi.org/10.1007/s00280-013-2165-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-013-2165-2