Abstract
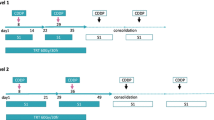
Aims: The aims of this study were to assess the efficacy and toxicity of concurrent chemoradiotherapy with divided schedule of cisplatin and vinorelbine in patients with locally advanced non-small-cell lung cancer (NSCLC). Methods: Patients with previously untreated, unresectable, and stage IIIA or IIIB NSCLC were eligible if they had a performance status of 0 or 1, were 75 years or younger, and had adequate organ function. Twenty-six patients (24 men and 2 women; median age, 66 years; age range, 42–75 years) were enrolled. Both cisplatin (40 mg/m2) and vinorelbine (20 mg/m2) were given on days 1 and 8 every 3 weeks. Beginning on day 2 of chemotherapy, thoracic radiotherapy was given for approximately 6 weeks (2 Gy per fraction; total dose, 60 Gy). Results: Five of the 26 patients achieved a complete response, and 16 achieved a partial response for an overall response rate of 80.8% (95% confidence interval, 60.6–93.4%). The median survival time was 23 months (range, 4–43 months). Overall survival rates at 1 and 2 years were 80 and 56%, respectively. Hematologic toxicities included grade 3–4 neutropenia in 84.6% of patients, grade 3–4 thrombocytopenia in 3.8%, and grade 3–4 anemia in 61.5%. Two patients (7.7%) had grade 3 radiation esophagitis that resolved completely without dilation. Grade 3–4 radiation pneumonitis occurred in two patients (7.7%) and was treated with corticosteroids. Both patients had a good partial resolution of symptoms and radiographic abnormalities. There were no treatment-related deaths. The actual delivered dose intensities for both cisplatin and vinorelbine were 79.5%. Radiotherapy was completed in 96% of patients. Conclusion: Concurrent chemoradiotherapy with cisplatin and vinorelbine administered on a divided schedule is effective and well tolerated in patients with locally advanced NSCLC.

Similar content being viewed by others
References
Choy H, Akerley W, Safran H, Graziano S, Chung C, Williams T, Cole B, Kennedy T (1998) Multiinstitutional phase II trial of paclitaxel, carboplatin, and concurrent radiation therapy for locally advanced non-small-cell lung cancer. J Clin Oncol 16:3316–3322
Cros S, Wright M, Morimoto M, Lataste H, Couzinier JP, Krikorian A (1989) Experimental antitumor activity of navelbine. Semin Oncol 16(suppl 4):15–20
Curren WJ, Scott CB, Langer CL, Komaki R, Lee JS, Hauser S, Movsas B, Wasserman T, Sause W, Cox JD (2003) Long-term benefit is observed in a phase III comparison of sequential vs concurrent chemo-radiation for patients with unresected stage III NSCLC: RTOG 9410. Proc Am Soc Clin Oncol 22:621
Depierre A, Lemarie E, Dabouis G, Garnier G, Jacoulet P, Dalphin JC (1991) A phase II study of navelbine (vinorelbine) in the treatment of non-small cell lung cancer. Am J Clin Oncol 14:115–119
Dewit L (1987) Combined treatment of radiation and cis-diammine-dichloroplatinum II: A review of experimental and clinical data. Int J Radiat Oncol Bio Phys 13:403–426
Dillman RO, Herndon J, Seagren SL, Eaton WL Jr, Green MR (1996) Improved survival in stage III non-small-cell lung cancer: seven-year follow-up of cancer and leukemia group B (CALGB) trial. J Natl Cancer Inst 88:1210–1215
Edelman MJ, Gandara DR (1996) Promising new agents in the treatment of non-small cell lung cancer. Cancer Chemother Pharmacol 37:385–393
Edelstein MP, Wolfe LA III, Duch DS (1996) Potentiation of radiation therapy by vinorelbine (Navelbine) in non-small cell lung cancer. Semin Oncol 23:41–47
Furuse K, Fukuoka M, Kawahara M, Nishiwaki H, Takada Y, Kudoh S, Katagami N, Ariyoshi Y (1999) Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 17:2692–2699
Goa KL, Faulds D (1994) Vinorelbine-a review of its pharmacological properties and clinical use in cancer chemotherapy. Drugs Aging Adis Int 5:200–234
Kelly K, Crowley J, Burn PA Jr, Presant CA, Grevstad PK, Moinpour CM, Ramsey SD, Wozniak AJ, Weiss GR, Moore DF, Israel VK, Livingston RB, Gandara D (2001) Randomized phase III trial of paclitaxel plus carboplatin versus vinorelbine plus cisplatin in the treatment of patients with advanced non-small-cell-lung-cancer: a Southwest Oncology Group trial. J Clin Oncol 19:3210–3218
Kiura K, Ueoka H, Segawa Y, Tabata M, Kamei H, Takigawa N, Hiraki S, Watanabe Y, Bessho A, Eguchi K, Okimoto N, Harita S, Takemoto M, Hiraki Y, Harada M, Tanimoto M (2003) Phase I/II study of docetaxel and cisplatin with concurrent thoracic radiation therapy for locally advanced non-small-cell lung cancer. Br J Cancer 89:795–802
Lau D, Leigh B, Gandara D, Edelman M, Morgan R, Israel V, Lara P, Wilder R, Ryu J, Doroshow J (2001) Twice-weekly paclitaxel and weekly carboplatin with concurrent thoracic radiation followed by carboplatin/paclitaxel consolidation for stage III non-small-cell lung cancer: a California cancer consortium phase II trial. J Clin Oncol 19:442–447
Le Chevalier T, Brisgand D, Douilland JY, Pujol JL, Alberola V, Monnier A, Riviere A, Lianes P, Chomy P, Cigolari S, Gottfried M, Ruffie P, Panizo A, Gaspard MH, Ravaioli A, Besenval M, Besson F, Martinez A, Berthaud P, Tursz T (1994) Randomized study of vinorelbine and cisplatin versus vindesine and cisplatin versus vinorelbin alone in advanced non-small cell lung cancer: results of a European multicentre trial including 612 patients. J Clin Oncol 12:360–367
Masters GA, Haraf DJ, Hoffman PC, Drinkard LC, Krauss SA, Ferguson MK, Oiak J, Samuels BL, Golomb HM, Vokes EE (1998) Phase I study of vinorelbine, cisplatin, and concomitant thoracic radiation in the treatment of advanced chest malignancies. J Clin Oncol 16:2157–2163
Non-small Cell Lung Cancer Collaborative Group (1995) Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomized clinical trails. BMJ 311:899–909
Perez CA, Stanley K, Grundy G, Hanson W, Rubin P, Kramer S, Brady LW, Marks JE, Perez-Tamayo R, Brown GS, Concannon JP, Rotman M (1982) Impact of irradiation technique and tumor extent in tumor control and survival of patients with unresectable non-oat cell carcinoma of the lung. Cancer 50:1091–1099
Pritchard RS, Anthony SP (1996) Chemotherapy plus radiotherapy compared with radiotherapy alone in the treatment of locally advanced, unresectable, non-small-cell lung cancer. Ann Intern Med 125:723–729
Robert F, Spencer SA, Childs HA III, Zhang R, Meredith RF, Wheeler RH, Hawkins MH, Carey D (2002) Concurrent chemoradiation therapy with cisplatin and paclitaxel for locally advanced non-small cell lung cancer: long-term follow-up of a phase I trial. Lung Cancer 37:189–199
Sause WT, Scott C, Taylor S, Johnson D, Livingston R, Komaki R, Emami B, Curran WJ, Byhardt RW, Turrisi AT, Dar AR, Cox JD (1995) Radiation therapy oncology group (RTOG) 88–08 and Eastern cooperative oncology group (ECOG) 4588: preliminary results of a phase III trial in regionally advanced unresectable non-small-cell lung cancer. J Natl Cancer Inst 87:198–205
Scagliotti GV, Marinis FD, Rinaldi M, Crino L, Gridelli C, Ricci S, Matano E, Boni C, Marangolo M, Failla G, Altavilla G, Adamo V, Ceribelli A, Clerici M, Costanzo FD, Frontini L, Tonato M (2002) Phase III randomized trial comparing three platinum-based doublets in advanced non-small-cell lung cancer. J Clin Oncol 20:4285–4291
Segawa Y, Ueoka H, Kiura K Kamei H, Tabata M, Sakae K, Hiraki Y, Kawahara S, Eguchi K, Hiraki S, Harada M (2000) A phase II study of cisplatin and 5-fluorouracil with concurrent hyperfactionated thoracic radiation for locally advanced non-small-cell lung cancer: a preliminary report from the Okayama lung cancer study group. Br J Cancer 82:104–111
Sekine I, Noda L, Oshita F, Yamada K, Tanaka M, Yamashita K, Nokihara H, Yamamoto N, Kunitoh H, Ohe Y, Tamura T, Kodama T, Sumi M, Saijo N (2004) Phase I study of cisplatin, vinorelbine, and concurrent thoracic radiotherapy for unresectable stage III non-small cell lung cancer. Cancer Sci 95:691–695
Solomon B, Ball DL, Richardson G, Smith JG, Millward M, Macmanus M, Michael M, Wirth A, Okane C, Muceniekas L, Ryan G, Rischin D (2003) Phase I/II study of concurrent twice-weekly paclitaxel and weekly cisplatin with radiation therapy for stage III non-small cell lung cancer. Lung Cancer 41: 353–361
Vokes EE, Herndon JE II, Crawford J, Leopold KA, Perry MC, Miller AA, Green MR (2002) Randomized phase II study of cisplatin with gemcitabine or paclitaxel or vinorelbine as induction chemotherapy followed by concomitant chemoradiotherapy for stage IIIB non-small-cell lung cancer: cancer and leukemia group B study 9431. J Clin Oncol 20:4191–4198
Zatloukal PV, Petruzelka L, Zemanova M, Havel L, Janku F, Judas L, Kubik A, Krepela E, Fiala P, Pecen L (2004) Concurrent versus sequential chemoradiotherapy with cisplatin and vinorelbine in locally advanced non-small cell lung cancer: a randomized study. Lung Cancer 46:87–98
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirose, T., Mizutani, Y., Ohmori, T. et al. The combination of cisplatin and vinorelbine with concurrent thoracic radiation therapy for locally advanced stage IIIA or IIIB non-small-cell lung cancer. Cancer Chemother Pharmacol 58, 361–367 (2006). https://doi.org/10.1007/s00280-005-0164-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-005-0164-7