Abstract
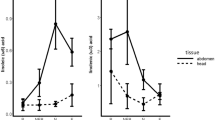
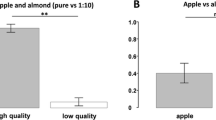
Honey bee colonies require adequate pollen for maintenance and growth. Pollens vary in nutritional value, and a balanced diet is achieved by mixing pollens with complementary essential nutrients. We tested subjective evaluation of pollens by foragers in colonies deprived of one of two essential fatty acids (eFAs), alpha-linolenic acid (omega-3) or linoleic acid (omega-6). We used four pollens, two rich in omega-3 and two rich in omega-6. A colony in an observation hive was allowed to forage for 2–5 days on a single pollen source. The following day, we repeatedly presented one of three pollens: the same pollen that the bees had been collecting the previous days, a novel pollen that was similarly deficient in omega-3 or omega-6, and a novel pollen that complemented their eFA deficiency. We measured the rate of waggle dances, which reflects on the strength of recruitment effort, of foragers returning to the observation hive from each of the pollens. Dance rates did not differ between the four pollens, but they were the highest to the “complementary” pollen and the lowest to the “same” pollen. Furthermore, this effect was greater for pollen combinations with greater eFA disparity between the same and the complementary pollens. Our findings support the ability of bees to balance colony eFA intake. Conditioning of the proboscis extension response (PER) tests showed that pollen foragers discriminated well between the four pollen odors, but the mechanisms by which bees assess pollen eFA composition remain to be elucidated. Differential dancing would recruit foragers to pollens that balance colony nutritional needs.
Significance statement
Honey bee foragers use dance communication to recruit nestmates to floral sources, with higher dance rates representing greater subjective evaluation of the floral source by the dancer. We fed colonies for a few days with a single pollen source, which was poor in one eFA (omega-3 or omega-6) and rich in the other. We then recorded the dances of foragers that collected each of the three pollens: the same pollen that the bees had been collecting the previous days, a novel pollen that was similarly deficient in omega-3 or omega-6, and a novel pollen that complemented their eFA deficiency. Dance rates were the highest to the “complementary” pollen and the lowest to the “same” pollen, showing that a colony could differentially recruit foragers to pollens that balance its nutritional needs.





Similar content being viewed by others
References
Afik O, Dag A, Kerem Z, Shafir S (2006a) Analyses of avocado (Persea americana) nectar properties and their perception by honey bees (Apis mellifera). J Chem Ecol 32:1949–1963
Afik O, Dag A, Shafir S (2006b) The effect of avocado (Persea americana) nectar composition on its attractiveness to honey bees (Apis mellifera). Apidologie 37(3):317–325
Afik O, Dag A, Shafir S (2008) Honeybee, Apis mellifera, round dance is influenced by trace components of floral nectar. Anim Behav 75:371–377
Alaux C, Ducloz F, Crauser D, Le Conte Y (2010) Diet effects on honeybee immunocompetence. Biol Lett 6(4):562–565
Archer CR, Kohler A, Pirk CWW, Oosthuizen V, Apostolides Z, Nicolson SW (2014) Antioxidant supplementation can reduce the survival costs of excess amino acid intake in honeybees. J Insect Physiol 71:78–86
Arien Y, Dag A, Zarchin S, Masci T, Shafir S (2015) Omega-3 deficiency impairs honey bee learning. Proc Natl Acad Sci U S A 112(51):15761–15766
Avni D, Dag A, Shafir S (2009) Pollen sources for honey bees in Israel: source, periods of shortage and influence on population growth. Isr J Plant Sci 57(3):263–275
Avni D, Hendriksma HP, Dag A, Uni Z, Shafir S (2014) Nutritional aspects of honey bee-collected pollen and constraints on colony development in the eastern Mediterranean. J Insect Physiol 69:65–73
Beekman M, Preece K, Schaerf TM (2016) Dancing for their supper: do honeybees adjust their recruitment dance in response to the protein content of pollen? Insect Soc 63(1):117–126
Bertazzini M, Medrzycki P, Bortolotti L, Maistrello L, Forlani G (2010) Amino acid content and nectar choice by forager honeybees (Apis mellifera L.) Amino Acids 39(1):315–318
Brenna JT (2011) Animal studies of the functional consequences of suboptimal polyunsaturated fatty acid status during pregnancy, lactation and early post-natal life. Matern Child Nutr 7(Suppl 2):59–79
Brodschneider R, Crailsheim K (2010) Nutrition and health in honey bees. Apidologie 41(3):278–294
Camazine S (1993) The regulation of pollen foraging by honey bees—how foragers assess the colony need for pollen. Behav Ecol Sociobiol 32(4):265–272
Camazine S, Crailsheim K, Hrassnigg N, Robinson GE, Leonhard B, Kropiunigg H (1998) Protein trophallaxis and the regulation of pollen foraging by honey bees (Apis mellifera L.) Apidologie 29(1–2):113–126
Carter C, Shafir S, Yehonatan L, Palmer RG, Thornburg R (2006) A novel role for proline in plant floral nectars. Naturwissenschaften 93(2):72–79
Chittka L, Thomson JD (eds) (2001) Cognitive ecology of pollination: animal behavior and floral evolution. Cambridge University Press, Cambridge
Cohen AC (2004) Insect diets. CRC Press, Boca Raton
Cook SM, Awmack CS, Murray DA, Williams IH (2003) Are honey bees’ foraging preferences affected by pollen amino acid composition? Ecol Entomol 28(5):622–627
Cook SM, Sandoz JC, Martin AP, Murray DA, Poppy GM, Williams IH (2005) Could learning of pollen odours by honey bees (Apis mellifera) play a role in their foraging behaviour? Physiol Entomol 30(2):164–174
de Brito Sanchez MG (2011) Taste perception in honey bees. Chem Senses 36(8):675–692
de Groot AP (1953) Protein and amino acid requirements of the honeybee (Apis mellifica L.) Phys Comp Oec 3:195–285
Di Pasquale G, Salignon M, Le Conte Y, Belzunces LP, Decourtye A, Kretzschmar A, Suchail S, Brunet JL, Alaux C (2013) Influence of pollen nutrition on honey bee health: do pollen quality and diversity matter? PLoS One 8(8):e72016
Dobson HEM (1988) Survey of pollen and pollenkitt lipids—chemical cues to flower visitors. Am J Bot 75(2):170–182
Dobson HEM, Bergstrom G (2000) The ecology and evolution of pollen odors. Plant Syst Evol 222(1–4):63–87
Donaldson-Matasci M, Dornhaus A (2014) Dance communication affects consistency, but not breadth, of resource use in pollen-foraging honey bees. PLoS One 9(10):e107527
Dreller C, Tarpy DR (2000) Perception of the pollen need by foragers in a honeybee colony. Anim Behav 59:91–96
Dussutour A, Simpson SJ (2009) Communal nutrition in ants. Curr Biol 19(9):740–744
Feldlaufer MF, Knox DA, Lusby WR, Shimanuki H (1993) Antimicrobial activity of fatty acids against Bacillus larvae, the causative agent of American foulbrood disease. Apidologie 24(2):95–99
Frisch K (1967) The dance language and orientation of bees. Harvard University Press, Cambridge
Gardner KE, Seeley TD, Calderone NW (2008) Do honeybees have two discrete dances to advertise food sources? Anim Behav 75:1291–1300
Griffin SR, Smith ML, Seeley TD (2012) Do honeybees use the directional information in round dances to find nearby food sources? Anim Behav 83(6):1319–1324
Gruter C, Ratnieks FLW (2011) Flower constancy in insect pollinators—adaptive foraging behavior or cognitive limitation? Commun Integr Biol 4(6):633–636
Hanley ME, Franco M, Pichon S, Darvill B, Goulson D (2008) Breeding system, pollinator choice and variation in pollen quality in British herbaceous plants. Funct Ecol 22(4):592–598
Hendriksma HP, Shafir S (2016) Honey bee foragers balance colony nutritional deficiencies. Behav Ecol Sociobiol 70:509–517
Hendriksma HP, Oxman KL, Shafir S (2014) Amino acid and carbohydrate tradeoffs by honey bee nectar foragers and their implications for plant-pollinator interactions. J Insect Physiol 69:56–64
Hilgert-Moreira SB, Nascher CA, Callegari-Jacques SM, Blochtein B (2014) Pollen resources and trophic niche breadth of Apis mellifera and Melipona obscurior (Hymenoptera, Apidae) in a subtropical climate in the Atlantic rain forest of southern Brazil. Apidologie 45(1):129–141
Kessler SC, Tiedeken EJ, Simcock KL, Derveau S, Mitchell J, Softley S, Stout JC, Wright GA (2015) Bees prefer foods containing neonicotinoid pesticides. Nature 521(7550):74–76
Kitaoka TK, Nieh JC (2009) Bumble bee pollen foraging regulation: role of pollen quality, storage levels, and odor. Behav Ecol Sociobiol 63(4):501–510
Kohler A, Pirk CW, Nicolson SW (2012) Honeybees and nectar nicotine: deterrence and reduced survival versus potential health benefits. J Insect Physiol 58(2):286–292
Konzmann S, Lunau K (2014) Divergent rules for pollen and nectar foraging bumblebees—a laboratory study with artificial flowers offering diluted nectar substitute and pollen surrogate. PLoS One 9(3):e91900
Leger DW, Didrichsons IA (1994) An assessment of data pooling and some alternatives. Anim Behav 48(4):823–832
Leonhardt SD, Bluthgen N (2012) The same, but different: pollen foraging in honeybee and bumblebee colonies. Apidologie 43(4):449–464
Lihoreau M, Charleston MA, Senior AM, Clissold FJ, Raubenheimer D, Simpson SJ, Buhl J (2017) Collective foraging in spatially complex nutritional environments. Philos Trans R Soc Lond Ser B Biol Sci 372(1727):20160238-1
London-Shafir I, Shafir S, Eisikowitch D (2003) Amygdalin in almond nectar and pollen—facts and possible roles. Plant Syst Evol 238(1–4):87–95
Manning R (2001) Fatty acids in pollen: a review of their importance for honey bees. Bee World 82(2):60–75
Manning R, Rutkay A, Eaton L, Dell B (2007) Lipid-enhanced pollen and lipid-reduced flour diets and their effect on the longevity of honey bees (Apis mellifera L.) Aust J Entomol 46:251–257
Mapalad KS, Leu D, Nieh JC (2008) Bumble bees heat up for high quality pollen. J Exp Biol 211(Pt 14):2239–2242
Nicholls E, de Ibarra NH (2014) Bees associate colour cues with differences in pollen rewards. J Exp Biol 217(Pt 15):2783–2788
Pankiw T, Page RE, Fondrk K (1998) Brood pheromone stimulates pollen foraging in honey bees (Apis mellifera). Behav Ecol Sociobiol 44:193–198
Paoli PP, Donley D, Stabler D, Saseendranath A, Nicolson SW, Simpson SJ, Wright GA (2014) Nutritional balance of essential amino acids and carbohydrates of the adult worker honeybee depends on age. Amino Acids 46(6):1449–1458
Pernal SF, Currie RW (2000) Pollen quality of fresh and 1-year-old single pollen diets for worker honey bees (Apis mellifera L.) Apidologie 31(3):387–409
Pernal SF, Currie RW (2001) The influence of pollen quality on foraging behavior in honeybees (Apis mellifera L.) Behav Ecol Sociobiol 51(1):53–68
Pernal SF, Currie RW (2002) Discrimination and preferences for pollen-based cues by foraging honeybees, Apis mellifera L. Anim Behav 63:369–390
Piskorski R, Kroder S, Dorn S (2011) Can pollen headspace volatiles and pollenkitt lipids serve as reliable chemical cues for bee pollinators? Chem Biodivers 8(4):577–586
Rasheed SA, Harder LD (1997) Economic motivation for plant species preferences of pollen-collecting bumble bees. Ecol Entomol 22(2):209–219
Raveret-Richter M, Waddington KD (1993) Past foraging experience influences honey bee dance behaviour. Anim Behav 46:123–128
Robertson AW, Mountjoy C, Faulkner BE, Roberts MV, Macnair MR (1999) Bumble bee selection of Mimulus guttatus flowers: the effects of pollen quality and reward depletion. Ecology 80(8):2594–2606
Roulston TH, Cane JH, Buchmann SL (2000) What governs protein content of pollen: pollinator preferences, pollen-pistil interactions, or phylogeny? Ecol Monogr 70(4):617–643
Ruedenauer FA, Spaethe J, Leonhardt SD (2015) How to know which food is good for you: bumblebees use taste to discriminate between different concentrations of food differing in nutrient content. J Exp Biol 218(Pt 14):2233–2240
Schmickl T, Crailsheim K (2004) Inner nest homeostasis in a changing environment with special emphasis on honey bee brood nursing and pollen supply. Apidologie 35(3):249–263
Schmidt JO (1982) Pollen foraging preferences of honey bees. Southwest Entomol 7(4):255–259
Schmidt JO, Thoenes SC, Levin MD (1987) Survival of honey bees, Apis mellifera (Hymenoptera:Apidae), fed various pollen sources. Ann Entomol Soc Am 80:176–183
Seeley TD (1995) The wisdom of the hive: the social physiology of honey bee colonies. Harvard University Press, Cambridge, MA
Seeley TD, Towne WF (1992) Tactics of dance choice in honey bees: do foragers compare dances? Behav Ecol Sociobiol 30:59–69
Seeley TD, Mikheyev AS, Pagano GJ (2000) Dancing bees tune both duration and rate of waggle-run production in relation to nectar-source profitability. J Comp Physiol A-Sens Neural Behav Physiol 186(9):813–819
Shafir S (1994) Intransitivity of preferences in honey bees: support for ‘comparative’ evaluation of foraging options. Anim Behav 48:55–67
Shafir S, Yehonatan L (2014) Comparative evaluations of reward dimensions in honey bees: evidence from two-alternative forced choice proboscis-extension conditioning. Anim Cogn 17(3):633–644
Shafir S, Waite TA, Smith BH (2002) Context-dependent violations of rational choice in honeybees (Apis mellifera) and gray jays (Perisoreus canadensis). Behav Ecol Sociobiol 51(2):180–187
Shafir S, Bechar A, Weber EU (2003) Cognition-mediated coevolution—context-dependent evaluations and sensitivity of pollinators to variability in nectar rewards. Plant Syst Evol 238(1–4):195–209
Shafir S, Menda G, Smith BH (2005) Caste-specific differences in risk sensitivity in honeybees, Apis mellifera. Anim Behav 69:859–868
Simopoulos AP (2002) The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 56(8):365–379
Simpson SJ, Raubenheimer D (2012) The nature of nutrition: a unifying framework from animal adaptation to human obesity. Princeton University Press, Princeton
Simpson SJ, Clissold FJ, Lihoreau M, Ponton F, Wilder SM, Raubenheimer D (2014) Recent advances in the integrative nutrition of arthropods. Annu Rev Entomol 60:293–311
Sklan D, Budowski P (1979) Cholesterol metabolism in the liver and intestine of the chick: effect of dietary cholesterol, taurocholic acid and cholestyramine. Lipids 14:386–390
Stabler D, Paoli PP, Nicolson SW, Wright GA (2015) Nutrient balancing of the adult worker bumblebee (Bombus terrestris) depends on the dietary source of essential amino acids. J Exp Biol 218(5):793–802
Stephens DW, Krebs JR (1986) Foraging theory. Princeton University Press, Princeton
Stephens DW, Brown JS, Ydenberg RC (eds) (2007) Foraging: behavior and ecology. The University of Chicago Press, Chicago
Vaudo AD, Patch HM, Mortensen DA, Tooker JF, Grozinger CM (2016a) Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc Natl Acad Sci U S A 113(28):E4035–E4042
Vaudo AD, Stabler D, Patch HM, Tooker JF, Grozinger CM, Wright GA (2016b) Bumble bees regulate their intake of essential protein and lipid pollen macronutrients. J Exp Biol 219(Pt 24):3962–3970
Waddington KD (1982) Honey bee foraging profitability and round dance correlates. J Comp Physiol A 148:297–301
Waddington KD (2001) Subjective evaluation and choice behavior by nectar- and pollen-collecting bees. In: Chittka L, Thompson JD (eds) Cognitive ecology of pollination: animal behavior and floral evolution. Cambridge University Press, Cambridge, pp 41–60
Waddington KD, Kirchner WH (1992) Acoustical and behavioral correlates of profitability of food sources in honey bee round dances. Ethology 92(1):1–6
Waddington KD, Nelson CM, Page RE (1998) Effects of pollen quality and genotype on the dance of foraging honey bees. Anim Behav 56:35–39
Winston M (1987) The biology of the honey bee. Harvard University Press, Cambridge
Wright GA, Thomson MG, Smith BH (2005) Odour concentration affects odour identity in honeybees. Proc Biol Sci 272(1579):2417–2422
Ydenberg RC (1998) Behavioral decisions about foraging and predator avoidance. In: Dukas R (ed) Cognitive ecology: the evolutionary ecology of information processing and decision making. University of Chicago Press, Chicago, pp 343–378
Acknowledgements
We thank Haim Kalev for beekee** assistance, Shahar Pincovici for assistance with bee dance recordings, and Zehava Uni for fatty acid analyses.
Funding
This research was supported by the Israel Science Foundation (grant no. 1456/10).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human rights
For this type of study, formal consent is not required.
Statement on the welfare of animals
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Informed consent was obtained from all individual participants included in the study. The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Additional information
Communicated by C. Grozinger
Rights and permissions
About this article
Cite this article
Zarchin, S., Dag, A., Salomon, M. et al. Honey bees dance faster for pollen that complements colony essential fatty acid deficiency. Behav Ecol Sociobiol 71, 172 (2017). https://doi.org/10.1007/s00265-017-2394-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-017-2394-1