Abstract
Purpose
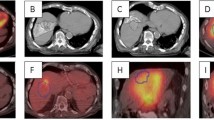
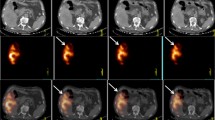
Radioembolization with 90Y microspheres is a locoregional radiation therapy for unresectable hepatic neoplasm. Non-target delivery of 90Y microspheres resulting in gastrointestinal (GI) symptoms is a recognized complication; there is minimal knowledge regarding the radiation effect to the gastric wall from left hepatic lobe 90Y treatments. Our aim was to study the incidence of GI complications when the target tissue (hepatic parenchyma ± tumor) is in close proximity to the gastric wall. We hypothesized that liver (tumor) to stomach proximity does not correlate with increased toxicity.
Methods
Between November 2011 and September 2013, we studied all patients who underwent left lobe radioembolization with 90Y glass microspheres. With Institutional Review Board (IRB) approval, we retrospectively reviewed MRI/CT images of these patients, identifying a subset of patients with the left hepatic lobe <1 cm from the gastric wall. Patients were seen in clinic 1 month posttreatment and subsequently at 3-month intervals. Short- and long-term gastric adverse events were tabulated.
Results
Ninety-seven patients successfully underwent left hepatic lobe 90Y microsphere radioembolization in which the average distance from the liver to the stomach wall was 1.0 ± 2.8 mm. The average dose for patients who received radioembolization to the left hepatic lobe was 109 ± 57 Gy. Fifty patients had tumor within 1 cm of the gastric wall. The average dose for patients who received radioembolization to the left hepatic lobe with tumor within 1 cm of the gastric wall was 121 ± 41 Gy. There were no reportable or recordable medical events. Of the patients, 34 % reported abdominal pain that was grade 1–2; 65 % of the patients reported no abdominal pain. None of the 97 patients developed a clinically evident GI ulcer.
Conclusion
Patients with left lobe tumors adjacent to or abutting the stomach do not exhibit acute or chronic radiation effects following radioembolization with glass microspheres.



Similar content being viewed by others
References
Dezarn WA, Cessna JT, DeWerd LA, Feng W, Gates VL, Halama J, et al. Recommendations of the American Association of Physicists in Medicine on dosimetry, imaging, and quality assurance procedures for 90Y microsphere brachytherapy in the treatment of hepatic malignancies. Med Phys 2011;38:4824–45.
TheraSphere® (US package insert). Rev. 11 ed. Ottawa, ON, Canada: Nordion Inc.; 2011.
Lewandowski RJ, Sato KT, Atassi B, Ryu RK, Nemcek Jr AA, Kulik L, et al. Radioembolization with 90Y microspheres: angiographic and technical considerations. Cardiovasc Intervent Radiol 2007;30:571–92. doi:10.1007/s00270-007-9064-z.
Salem R, Thurston KG. Radioembolization with 90yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 1: technical and methodologic considerations. J Vasc Interv Radiol 2006;17:1251–78. doi:10.1097/01.RVI.0000233785.75257.9A.
Garin E, Lenoir L, Rolland Y, Edeline J, Mesbah H, Laffont S, et al. Dosimetry based on 99mTc-macroaggregated albumin SPECT/CT accurately predicts tumor response and survival in hepatocellular carcinoma patients treated with 90Y-loaded glass microspheres: preliminary results. J Nucl Med 2012;53:255–63. doi:10.2967/jnumed.111.094235.
Bolch WE, Bouchet LG, Robertson JS, Wessels BW, Siegel JA, Howell RW, et al. MIRD pamphlet No. 17: the dosimetry of nonuniform activity distributions--radionuclide S values at the voxel level. Medical Internal Radiation Dose Committee. J Nucl Med 1999;40:11S–36S.
Naymagon S, Warner RR, Patel K, Harpaz N, Machac J, Weintraub JL, et al. Gastroduodenal ulceration associated with radioembolization for the treatment of hepatic tumors: an institutional experience and review of the literature. Dig Dis Sci 2010;55:2450–8. doi:10.1007/s10620-010-1156-y.
Song SY, Chung JW, Lim HG, Park JH. Nonhepatic arteries originating from the hepatic arteries: angiographic analysis in 250 patients. J Vasc Interv Radiol 2006;17:461–9. doi:10.1097/01.rvi.0000202718.16416.18.
Blanchard RJ, Morrow IM, Sutherland JB. Treatment of liver tumors with yttrium-90 microspheres alone. Can Assoc Radiol J 1989;40:206–10.
Carretero C, Munoz-Navas M, Betes M, Angos R, Subtil JC, Fernandez-Urien I, et al. Gastroduodenal injury after radioembolization of hepatic tumors. Am J Gastroenterol 2007;102:1216–20. doi:10.1111/j.1572-0241.2007.01172.x.
South CD, Meyer MM, Meis G, Kim EY, Thomas FB, Rikabi AA, et al. Yttrium-90 microsphere induced gastrointestinal tract ulceration. World J Surg Oncol 2008;6:93. doi:10.1186/1477-7819-6-93.
Murthy R, Brown DB, Salem R, Meranze SG, Coldwell DM, Krishnan S, et al. Gastrointestinal complications associated with hepatic arterial yttrium-90 microsphere therapy. J Vasc Interv Radiol 2007;18:553–61. doi:10.1016/j.jvir.2007.02.002. quiz 62.
Riaz A, Lewandowski RJ, Kulik LM, Mulcahy MF, Sato KT, Ryu RK, et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J Vasc Interv Radiol 2009;20:1121–30. doi:10.1016/j.jvir.2009.05.030. quiz 31.
Neff R, Abdel-Misih R, Khatri J, Dignazio M, Garcia M, Petrelli N, et al. The toxicity of liver directed yttrium-90 microspheres in primary and metastatic liver tumors. Cancer Invest 2008;26:173–7. doi:10.1080/07357900701512688.
Kennedy AS, Coldwell D, Nutting C, Murthy R, Wertman Jr DE, Loehr SP, et al. Resin 90Y-microsphere brachytherapy for unresectable colorectal liver metastases: modern USA experience. Int J Radiat Oncol Biol Phys 2006;65:412–25. doi:10.1016/j.ijrobp.2005.12.051.
Konda A, Savin MA, Cappell MS, Duffy MC. Radiation microsphere-induced GI ulcers after selective internal radiation therapy for hepatic tumors: an underrecognized clinical entity. Gastrointest Endosc 2009;70:561–7. doi:10.1016/j.gie.2009.03.015.
Szyszko T, Al-Nahhas A, Tait P, Rubello D, Canelo R, Habib N, et al. Management and prevention of adverse effects related to treatment of liver tumours with 90Y microspheres. Nucl Med Commun 2007;28:21–4. doi:10.1097/MNM.0b013e3280121a8f.
Mallach S, Ramp U, Erhardt A, Schmitt M, Häussinger D. An uncommon cause of gastro-duodenal ulceration. World J Gastroenterol 2008;14:2593–5.
Luo DL, Chan JK. Basophilic round bodies in gastric biopsies little known by pathologists: iatrogenic yttrium 90 microspheres deriving from selective internal radiation therapy. Int J Surg Pathol 2013;21:535–7. doi:10.1177/1066896913481057.
Chon YE, Seong J, Kim BK, Cha J, Kim SU, Park JY, et al. Gastroduodenal complications after concurrent chemoradiation therapy in patients with hepatocellular carcinoma: endoscopic findings and risk factors. Int J Radiat Oncol Biol Phys 2011;81:1343–51. doi:10.1016/j.ijrobp.2010.07.1986.
Yang MH, Lee JH, Choi MS, Park HS, Rhee PL, Kim JJ, et al. Gastrointestinal complications after radiation therapy in patients with hepatocellular carcinoma. Hepatogastroenterology 2005;52:1759–63.
Yoon H, Oh D, Park HC, Kang SW, Han Y, Lim DH, et al. Predictive factors for gastroduodenal toxicity based on endoscopy following radiotherapy in patients with hepatocellular carcinoma. Strahlenther Onkol 2013;189:541–6. doi:10.1007/s00066-013-0343-0.
Streitparth F, Pech M, Böhmig M, Ruehl R, Peters N, Wieners G, et al. In vivo assessment of the gastric mucosal tolerance dose after single fraction, small volume irradiation of liver malignancies by computed tomography-guided, high-dose-rate brachytherapy. Int J Radiat Oncol Biol Phys 2006;65:1479–86. doi:10.1016/j.ijrobp.2006.02.052.
Elschot M, Vermolen BJ, Lam MG, de Keizer B, van den Bosch MA, de Jong HW. Quantitative comparison of PET and bremsstrahlung SPECT for imaging the in vivo yttrium-90 microsphere distribution after liver radioembolization. PLoS One 2013;8:e55742. doi:10.1371/journal.pone.0055742.
Kao YH, Steinberg JD, Tay YS, Lim GK, Yan J, Townsend DW, et al. Post-radioembolization yttrium-90 PET/CT - part 2: dose–response and tumor predictive dosimetry for resin microspheres. EJNMMI Res 2013;3:57. doi:10.1186/2191-219X-3-57.
Compliance with ethical standards
ᅟ
Role of funding
There was no funding provided for this study.
Conflicts of interest
RJL and RS are advisors to BTG. The other authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gates, V.L., Hickey, R., Marshall, K. et al. Gastric injury from 90Y to left hepatic lobe tumors adjacent to the stomach: fact or fiction?. Eur J Nucl Med Mol Imaging 42, 2038–2044 (2015). https://doi.org/10.1007/s00259-015-3122-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-015-3122-6