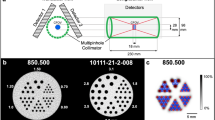
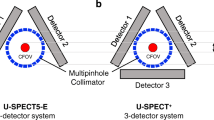
Abstract
Purpose
We compared the performance of three commercial small-animal μSPECT scanners equipped with multipinhole general purpose (GP) and multipinhole high-resolution (HR) collimators designed for imaging mice.
Methods
Spatial resolution, image uniformity, point source sensitivity and contrast recovery were determined for the U-SPECT-II (MILabs), the NanoSPECT-NSO (BioScan) and the X-SPECT (GE) scanners. The pinhole diameters of the HR collimator were 0.35 mm, 0.6 mm and 0.5 mm for these three systems respectively. A pinhole diameter of 1 mm was used for the GP collimator. To cover a broad field of imaging applications three isotopes were used with various photon energies: 99mTc (140 keV), 111In (171 and 245 keV) and 125I (27 keV). Spatial resolution and reconstructed image uniformity were evaluated in both HR and a GP mode with hot rod phantoms, line sources and a uniform phantom. Point source sensitivity and contrast recovery measures were additionally obtained in the GP mode with a novel contrast recovery phantom developed in-house containing hot and cold submillimetre capillaries on a warm background.
Results
In hot rod phantom images, capillaries as small as 0.4 mm with the U-SPECT-II, 0.75 mm with the X-SPECT and 0.6 mm with the NanoSPECT-NSO could be resolved with the HR collimators for 99mTc. The NanoSPECT-NSO achieved this resolution in a smaller field-of-view (FOV) and line source measurements showed that this device had a lower axial than transaxial resolution. For all systems, the degradation in image resolution was only minor when acquiring the more challenging isotopes 111In and 125I. The point source sensitivity with 99mTc and GP collimators was 3,984 cps/MBq for the U-SPECT-II, 620 cps/MBq for the X-SPECT and 751 cps/MBq for the NanoSPECT-NSO. The effects of volume sensitivity over a larger object were evaluated by measuring the contrast recovery phantom in a realistic FOV and acquisition time. For 1.5-mm rods at a noise level of 8 %, the contrast recovery coefficient (CRC) was 42 %, 37 % and 34 % for the U-SPECT-II, X-SPECT and NanoSPECT-NSO, respectively. At maximal noise levels of 10 %, a CRCcold of 70 %, 52 % and 42 % were obtained for the U-SPECT-II, X-SPECT and NanoSPECT-NSO, respectively. When acquiring 99mTc with the GP collimators, the integral/differential uniformity values were 30 %/14 % for the U-SPECT-II, 50 %/30 % for the X-SPECT and 38 %/25 % for the NanoSPECT-NSO. When using the HR collimators, these uniformity values remained similar for U-SPECT-II and X-SPECT, but not for the NanoSPECT-NSO for which the uniformity deteriorated with larger volumes.
Conclusion
We compared three μSPECT systems by acquiring and analysing mouse-sized phantoms including a contrast recovery phantom built in-house offering the ability to measure the hot contrast on a warm background in the submillimetre resolution range. We believe our evaluation addressed the differences in imaging potential for each system to realistically image tracer distributions in mouse-sized objects.








Similar content being viewed by others
References
Mankoff DA. A definition of molecular imaging. J Nucl Med. 2007;48:18N, 21N.
Cherry SR. In vivo molecular and genomic imaging: new challenges for imaging physics. Phys Med Biol. 2004;49:R13–48.
Bailey DL, Karp JS, Surti S. Physics and instrumentation in PET, in positron emission tomography: basic science and clinical practice. Philadelphia: Springer; 2003. p. 41–67.
Ishizu K, Mukai T, Yonekura Y, Pagani M, Fujita T, Magata Y, et al. Ultra-high resolution SPECT system using four pinhole collimators for small animal studies. J Nucl Med. 1995;36:2282–7.
Jaszczak RJ, Li J, Wang H, Zalutsky MR, Coleman RE. Pinhole collimation for ultra-high-resolution, small-field-of-view SPECT. Phys Med Biol. 1994;39:425–37.
McElroy DP, MacDonald LR, Beekman FJ, Yuchuan Wang, Patt BE, Iwanczyk JS, et al. Evaluation of A-SPECT: a desktop pinhole SPECT system for small animal imaging. Nuclear Science Symposium Conference Record, 2001 IEEE, vol. 3, pp. 1835–1839, 2001. doi:10.1109/NSSMIC.2001.1008699.
Weber DA, Ivanovic M, Franceschi D, Strand SE, Erlandsson K, Franceschi M, et al. Pinhole SPECT: an approach to in vivo high resolution SPECT imaging in small laboratory animals. J Nucl Med. 1994;35:342–8.
Beekman FJ, Vastenhouw B. Design and simulation of a high-resolution stationary SPECT system for small animals. Phys Med Biol. 2004;49:4579–92.
Beekman FJ, van der Have F, Vastenhouw B, van der Linden AJA, van Rijk PP, Burbach JPH, et al. U-SPECT-I: a novel system for submillimeter-resolution tomography with radiolabeled molecules in mice. J Nucl Med. 2005;46:1194–200.
Wilson DW, Barrett HH, Furenlid LR. A new design for a SPECT small-animal imager. Nuclear Science Symposium Conference Record, 2001 IEEE, vol. 3, pp. 1826–1829, 2001.doi:10.1109/NSSMIC.2001.1008697.
Meikle SR, Kench P, Weisenberger AG, Wojcik R, Smith MF, Majewski S, et al. A prototype coded aperture detector for small animal SPECT. Nuclear Science Symposium Conference Record, 2001 IEEE, vol. 3, pp. 1580–1584, 2001.doi:10.1109/NSSMIC.2001.1008641.
Liu Z, Kastis GA, Stevenson GD, Barrett HH, Furenlid LR, Kupinski MA, et al. Quantitative analysis of acute myocardial infarct in rat hearts with ischemia-reperfusion using a high-resolution stationary SPECT system. J Nucl Med. 2002;43:933–9.
Rowe RK, Aarsvold JN, Barrett HH, Chen JC, Klein WP, Moore BA, et al. A stationary hemispherical SPECT imager for 3-dimensional brain imaging. J Nucl Med. 1993;34:474–80.
Schramm NU, Ebel G, Engeland U, Schurrat T, Behe M, Behr TM. High-resolution SPECT using multipinhole collimation. IEEE Trans Nucl Sci. 2003;50:315–20.
Lackas C, Schramm NU, Hoppin JW, Engeland U, Wirrwar A, Halling H. T-SPECT: a novel imaging technique for small animal research. IEEE Trans Nucl Sci. 2005;52:181–7.
Kim H, Furenlid LR, Crawford MJ, Wilson DW, Barber HB, Peterson TE, et al. SemiSPECT: a small-animal single-photon emission computed tomography (SPECT) imager based on eight cadmium zinc telluride (CZT) detector arrays. Med Phys. 2006;33:465–74.
Miller BW, Furenlid LR, Moore SK, Barber HB, Nagarkar VV, Barrett HH. System Integration of FastSPECT III, a Dedicated SPECT Rodent-Brain Imager Based on BazookaSPECT Detector Technology. IEEE Nucl Sci Symp Conf Rec (1997) 2009, Oct. 24 2009-Nov. 1 2009;4004–4008.
Beekman F, van der Have F. The pinhole: gateway to ultra-high-resolution three-dimensional radionuclide imaging. Eur J Nucl Med Mol Imaging. 2007;34:151–61.
Wu C, de Jong JR, van Andel HA G, van der Have F, Vastenhouw B, Laverman P, et al. Quantitative multi-pinhole small-animal SPECT: uniform versus non-uniform Chang attenuation correction. Phys Med Biol. 2011;56:N183–93.
Constantinesco A, Choquet P, Monassier L, Israel-Jost V, Mertz L. Assessment of left ventricular perfusion, volumes, and motion in mice using pinhole gated SPECT. J Nucl Med. 2005;46:1005–11.
Golestani R, Wu C, Tio RA, Zeebregts CJ, Petrov AD, Beekman FJ, et al. Small-animal SPECT and SPECT/CT: application in cardiovascular research. Eur J Nucl Med Mol Imaging. 2010;37:1766–77.
Auricchio A, Acton PD, Hildinger M, Louboutin JP, Plossl K, O’Connor E, et al. In vivo quantitative noninvasive imaging of gene transfer by single-photon emission computerized tomography. Hum Gene Ther. 2003;14:255–61.
Kumar SR, Deutscher SL, Figueroa SD. Tumor targeting and SPECT imaging properties of an (111)In-labeled galectin-3 binding peptide in prostate carcinoma. Nucl Med Biol. 2009;36:137–46.
Gambini JP, Cabral P, Alonso O, Savio E, Figueroa SD, Zhang X, et al. Evaluation of 99mTc-glucarate as a breast cancer imaging agent in a xenograft animal model. Nucl Med Biol. 2011;38:255–60.
Luo SN, Wang Y, Lin JG, Qiu L, Cheng W, Zhai HZ, et al. Animal studies of (99m)Tc-i-PIDP: a new bone imaging agent. Appl Radiat Isotopes. 2011;69:1169–75.
Booij J, de Bruin K, de Win MML, Lavini C, den Heeten GJ, Habraken JBA. Imaging of striatal dopamine transporters in rat brain with single pinhole SPECT and co-aligned MRI is highly reproducible. Nucl Med Biol. 2003;30:643–9.
Bennink RJ, Hamann J, de Bruin K, ten Kate FJ, van Deventer SJ, te Velde AA. Dedicated pinhole SPECT of intestinal neutrophil recruitment in a mouse model of dextran sulfate sodium-induced colitis. J Nucl Med. 2005;46:526–31.
Goorden MC, Beekman FJ. High-resolution tomography of positron emitters with clustered pinhole SPECT. Phys Med Biol. 2010;55:1265–77.
van der Have F, Vastenhouw B, Rentmeester M, Beekman FJ. System calibration and statistical image reconstruction for ultra-high resolution stationary pinhole SPECT. IEEE Trans Med Imaging. 2008;27:960–71.
Miller BW, Van Holen R, Barrett H, Furenlid L. A system calibration and fast iterative reconstruction method for next-generation SPECT imagers. IEEE Trans Nucl Sci. 2012;59:1990–6.
Branderhorst W, Vastenhouw B, van der Have F, Blezer ELA, Bleeker WK, Beekman FJ. Targeted multi-pinhole SPECT. Eur J Nucl Med Mol Imaging. 2011;38:552–61.
Mok GS, Tsui BM, Beekman FJ. The effects of object activity distribution on multiplexing multi-pinhole SPECT. Phys Med Biol. 2011;56:2635–50.
Vunckx K, Suetens P, Nuyts J. Effect of overlap** projections on reconstruction image quality in multipinhole SPECT. IEEE Trans Med Imaging. 2008;27:972–83.
Cherry SR, Sorensen J, Phelps ME. Physics in nuclear medicine. Philadelphia: Saunders; 2012.
Vastenhouw B, Beekman F. Submillimeter total-body murine imaging with U-SPECT-I. J Nucl Med. 2007;48:487–93.
Vaissier PE, Goorden MC, Vastenhouw B, van der Have F, Ramakers RM, Beekman FJ. Fast spiral SPECT with stationary gamma-cameras and focusing pinholes. J Nucl Med. 2012;53:1292–9.
van der Have F, Vastenhouw B, Ramakers RM, Branderhorst W, Krah JO, Ji C, et al. U-SPECT-II: an ultra-high-resolution device for molecular small-animal imaging. J Nucl Med. 2009;50:599–605.
Wyckhuys T, Staelens S, Van Nieuwenhuyse B, Deleye S, Hallez H, Vonck K, et al. Hippocampal deep brain stimulation induces decreased rCBF in the hippocampal formation of the rat. Neuroimage. 2010;52:55–61.
Van Steenkiste C, Staelens S, Deleye S, De Vos F, Vandenberghe S, Geerts A, et al. Measurement of porto-systemic shunting in mice by novel three-dimensional micro-single photon emission computed tomography imaging enabling longitudinal follow-up. Liver Int. 2010;30:1211–20.
Wu C, van der Have F, Vastenhouw B, Dierckx RAJO, Paans AMJ, Beekman FJ. Absolute quantitative total-body small-animal SPECT with focusing pinholes. Eur J Nucl Med Mol Imaging. 2010;37:2127–35.
Hoeben BAW, Molkenboer-Kuenen JDM, Oyen WJG, Peeters WJM, Kaanders JHAM, Bussink J, et al. Radiolabeled cetuximab: dose optimization for epidermal growth factor receptor imaging in a head-and-neck squamous cell carcinoma model. Int J Cancer. 2011;129:870–8.
De Vos F, De Bruyne S, Wyffels L, Boos TL, Staelens S, Deleye S, et al. In vivo evaluation of [(123)I]-4-(2-(bis(4-fluorophenyl)methoxy)ethyl)-1-(4-iodobenzyl)piperidine, an iodinated SPECT tracer for imaging the P-gp transporter. Nucl Med Biol. 2010;37:469–77.
Vangestel C, Van de Wiele C, Mees G, Mertens K, Staelens S, Reutelingsperger C, et al. Single-photon emission computed tomographic imaging of the early time course of therapy-induced cell death using technetium 99m tricarbonyl His-annexin A5 in a colorectal cancer xenograft model. Mol Imaging. 2012;11:135–47.
Vangestel C, Van de Wiele C, Van Damme N, Staelens S, Pauwels P, Reutelingsperger CP, et al. (99)mTc-(CO)(3) His-annexin A5 micro-SPECT demonstrates increased cell death by irinotecan during the vascular normalization window caused by bevacizumab. J Nucl Med. 2011;52:1786–94.
Vervoort L, Burvenich I, Staelens S, Dumolyn C, Waegemans E, Van Steenkiste M, et al. Preclinical evaluation of monoclonal antibody 14C5 for targeting pancreatic cancer. Cancer Biother Radiopharm. 2010;25:193–205.
Blanckaert P, Burvenich I, Staelens S, De Bruyne S, Moerman L, Wyffels L, et al. Effect of cyclosporin A administration on the biodistribution and multipinhole muSPECT imaging of [123I]R91150 in rodent brain. Eur J Nucl Med Mol Imaging. 2009;36:446–53.
Mok GSP, Yu JH, Du Y, Wang YC, Tsui BMW. Evaluation of a multi-pinhole collimator for imaging small animals with different sizes. Mol Imaging Biol. 2012;14:60–9.
Goins BA, Soundararajan A, Bao A, Phillips WT, Perez R. [(186)Re]Liposomal doxorubicin (Doxil): in vitro stability, pharmacokinetics, imaging and biodistribution in a head and neck squamous cell carcinoma xenograft model. Nucl Med Biol. 2009;36:515–24.
Phillips WT, Head HW, Dodd GD, Bao A, Soundararajan A, Garcia-Rojas X, et al. Combination radiofrequency ablation and intravenous radiolabeled liposomal doxorubicin: imaging and quantification of increased drug delivery to tumors. Radiology. 2010;255:405–14.
Lin K, Hsiao I-T, Wietholt C, Chung Y, Chen C, Yen T. Performance evaluation of an animal SPECT using modified NEMA standards. J Nucl Med. 2008;49 Suppl 1:402P.
Schramm NU, Lackas C, Hoppin JW, Forrer F, de Jong M. The NanoSPECT/CT: a high-sensitivity small-animal SPECT/CT with submillimeter spatial resolution. Eur J Nucl Med Mol Imaging. 2006;47: Supl 1:233P.
Schramm NU, Lackas C, Gershman B, Norenberg JP, de Jong M. Improving resolution, sensitivity and applications for the NanoSPECT/CT: a high-performance SPECT/CT imager for small-animal research. Eur J Nucl Med Mol Imaging. 2007;34:S226–7.
Gershman B, Hoppin J, Schramm N, Lackas C, Norenberg J. Evaluation of the quantification capabilities of a NanoSPECT/CT as a function of angular sampling, counting statistics, reconstruction parameters and the dynamic range of measured activity. J Nucl Med. 2007;48:433P.
Pesnel S, Guminski Y, Pillon A, Lerondel S, Imbert T, Guilbaud N, et al. (99m)Tc-HYNIC-spermine for imaging polyamine transport system-positive tumours: preclinical evaluation. Eur J Nucl Med Mol Imaging. 2011;38:1832–41.
McLarty K, Cornelissen B, Cai Z, Scollard DA, Costantini DL, Done SJ, et al. Micro-SPECT/CT with 111In-DTPA-pertuzumab sensitively detects trastuzumab-mediated HER2 downregulation and tumor response in athymic mice bearing MDA-MB-361 human breast cancer xenografts. J Nucl Med. 2009;50:1340–8.
National Electrical Manufacturers Association. Standards Publication NU 1–2007. Rosslyn: National Electrical Manufacturers Association; 2007.
National Electrical Manufacturers Association. Performance measurements of positron emission tomographs. Rosslyn: National Electrical Manufacturers Association; 2001.
Branderhorst W, Vastenhouw B, Beekman FJ. Pixel-based subsets for rapid multi-pinhole SPECT reconstruction. Phys Med Biol. 2010;55:2023–34.
Van Holen R, Staelens S, Vandenberghe S. Tomographic image quality of rotating slat versus parallel hole-collimated SPECT. Phys Med Biol. 2011;56:7205–22.
Harteveld AA, Meeuwis AP, Disselhorst JA, Slump CH, Oyen WJ, Boerman OC, et al. Using the NEMA NU 4 PET image quality phantom in multipinhole small-animal SPECT. J Nucl Med. 2011;52:1646–53.
Visser EP, Harteveld AA, Meeuwis AP, Disselhorst JA, Beekman FJ, Oyen WJ, et al. Image quality phantom and parameters for high spatial resolution small-animal SPECT. J Nucl Med. 2011;52:1646–53.
DiFilippo FP, Gallo SL, Klatte RS, Patel S. A fillable micro-hollow sphere lesion detection phantom using superposition. Phys Med Biol. 2010;55:5363–81.
Liow JS, Strother SC. The convergence of object dependent resolution in maximum-likelihood based tomographic image-reconstruction. Phys Med Biol. 1993;38:55–70.
Wilson DW, Tsui BM, Barrett HH. Noise properties of the EM algorithm: II. Monte-Carlo simulations. Phys Med Biol. 1994;39:847–71.
Barrett HH, Wilson DW, Tsui BM. Noise properties of the EM algorithm: I. Theory. Phys Med Biol. 1994;39:833–46.
Mok GS, Wang Y, Tsui BM. Quantification of the multiplexing effects in multi-pinhole small animal SPECT: a simulation study. IEEE Trans Nucl Sci. 2009;56:2636–43.
Magota K, Kubo N, Kuge Y, Nishijima KI, Zhao S, Tamaki N. Performance characterization of the Inveon preclinical small-animal PET/SPECT/CT system for multimodality imaging. Eur J Nucl Med Mol Imaging. 2011;38:742–52.
Boisson F, Zahra D, Parmar A, Meikle S, Rellbac A. Mouse imaging capabilities of the Inveon SPECT system using single and multi-pinhole collimators dedicated to mouse studies. World Molecular Imaging Congress. 2012. Abstract P139.
Meikle SR, Kench P, Kassiou M, Banati RB. Small animal SPECT and its place in the matrix of molecular imaging technologies. Phys Med Biol. 2005;50:R45–61.
Acknowledgments
This work was supported by the University of Antwerp, the Research Foundation – Flanders (FWO), the Belgian Science Policy Office (BELSPO), iMinds and Ghent University. The authors would like to thank Peter Laverman of Radboud University Nijmegen, Ruud Ramakers of the University Medical Center Utrecht, Ciara Finucane and Jerome Burnet of Queen Mary, University of London, and Alessandro Passeri of the University of Florence for their cooperation and technical assistance with, respectively, the U-SPECT-II, the NanoSPECT-NSO and the X-SPECT measurements.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributions
S.D. performed and analysed the experiments, took part in the phantom design and construction, contributed to the writing and made all the figures. R.V.H. performed the X-SPECT experiments, contributed to the phantom design, the experimental setup and proofreading. J.V. took part in the experimental setup, image analysis and proofreading. S.V.D.B. contributed to the initial experimental setup and took part in the proofreading. Si.St. provided the motivation for the introduction of μSPECT for the investigation of antibodies and peptides in preference to μPET and contributed to the proofreading, St.St. set up the framework, originated and contributed to the phantom design, designed the experimental setups, and took part in the writing, discussion and proofreading.
Rights and permissions
About this article
Cite this article
Deleye, S., Van Holen, R., Verhaeghe, J. et al. Performance evaluation of small-animal multipinhole μSPECT scanners for mouse imaging. Eur J Nucl Med Mol Imaging 40, 744–758 (2013). https://doi.org/10.1007/s00259-012-2326-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-012-2326-2