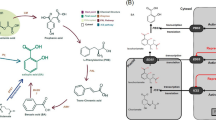
Abstract
Trichoderma viride has a wide range of applications in plant growth promotion, biological control, cellulase production, and biomass utilization. Salinity is a major limitation to Trichoderma strains in the natural environment and fermentation environment, and to improve the adaptability of Trichoderma to salt stress is of great significance to its applications in industry and agriculture. Histone acetylation plays important roles in the regulation of physiological and biochemical processes including various stress responses. GCN5 is the most representative histone acetylase, which plays vital roles in chromatin remodeling of promoters to facilitate the transcription activation. In this paper, we identified a GCN5-encoding gene TvGCN5 in T. viride Tv-1511, and characterized the function and regulating mechanism of TvGCN5-mediated acetylation of histone H3 in the salt adoption of Tv-1511, by constructions of the deletion mutants (Tv-1511-△GCN5) and overexpression mutants (Tv-1511-GCN5-OE) of TvGCN5. Results showed that compared with wild-type Tv-1511, the over-expression of TvGCN5 resulted in the longer mycelia diameter and more biomass under salt stress. Furthermore, Tv-1511-△GCN5 strains obtained the improved sodium (Na+) compartmentation and antioxidant capacity by upregulating the transcriptional levels of genes encoding PM H+-ATPase, vacuolar H+-ATPase, and antioxidant enzymes. Notably, the changes in the transcriptional expressions of these genes are tightly modulated by the TvGCN5-mediated acetylated level of histone H3 in their promoter regions. In all, these results reveal that TvGCN5 plays an important role in stress tolerance of T. viride Tv-1511, and provides potential insight to facilitate the application of epigenetic modulation in the expanding utilization of Trichoderma.
Key points
• Overexpresison of TvGCN5 improves the adoption of T. viride Tv-1511 to salt stress by increasing acetylation level of histone H3 on the promoter regions of sodium-transport and antioxidant-related genes, at H3K9ac, H3K14ac, H3K23ac, and H3K27ac.
• Overexprsison of TvGCN5 enhances the ion transport and compartmentation capacity by upregulating the expressions and activities of PM and vacuolar H+-ATPase to tolerate salt stress.
• Overexprsison of TvGCN5 promotes the antioxidant capacity by increasing the expressions and activities of antioxidant enzymes in response to salt stress.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Aghcheh RK, Kubicek CP (2015) Epigenetics as an emerging tool for improvement of fungal strains used in biotechnology. Appl Microbiol Biotechnol 99:6167–6181. https://doi.org/10.1007/s00253-015-6763-2
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341. https://doi.org/10.1093/jexbot/53.372.1331
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Apse MP, Blumwald E (2007) Na+ transport in plants. FEBS Lett 581:2247–2254. https://doi.org/10.1016/j.febslet.2007.04.014
Asai T, Morita S, Taniguchi T, Monde K, Oshima Y (2016) Epigenetic stimulation of polyketide production in Chaetomium cancroideum by an NAD+-dependent HDAC inhibitor. Org Biomol Chem 14:646–651. https://doi.org/10.1039/c5ob01595b
Benítez T, Rincón AM, Limón MC, Codón AC (2004) Biocontrol mechanisms of Trichoderma strains. Int Microbiol 7:249–260. https://doi.org/10.2436/im.v7i4.9480
Cánovas D, Marcos AT, Gacek A, Ramos MS, Strauss J (2014) The histone acetyltransferase GcnE (GCN5) plays a central role in the regulation of Aspergillus asexual development. Genetics 197:1175–1189. https://doi.org/10.1534/genetics.114.165688
Cohen R, Schocken J, Kaldis A, Vlachonasios KE, Hark AT, McCain ER (2009) The histone acetyltransferase GCN5 affects the inflorescence meristem and stamen development in Arabidopsis. Planta 230:1207–1221. https://doi.org/10.1007/s00425-009-1012-5
Danielle R (2021) Worth its salt: a histone acetyltransferase gene enhances salt tolerance in bread wheat. Plant Physiol 186:1752–1753. https://doi.org/10.1093/plphys/kiab248
Dixon D, Fordham-Skelton T (1998) Plant-microbe interactions. Curr Opin Plant Biol 1:283–284. https://doi.org/10.1016/S1369-5266(98)80029-0
Druzhinina IS, Seidl-Seiboth V, Herrera-Estrella A, Horwitz BA, Kenerley CM, Monte E (2011) Trichoderma: the genomics of opportunistic success. Nat Rev Microbiol 9:749–759. https://doi.org/10.1038/nrmicro2637
Fan A, Mi W, Liu Z, Zeng G, Zhang P, Hu Y (2017) Deletion of a histone acetyltransferase leads to the pleiotropic activation of natural products in Metarhizium robertsii. Org Lett 19:1686–1689. https://doi.org/10.1021/acs.orglett.7b00476
Ferreiro JA, Powell NG, Karabetsou N, Mellor J, Waters R (2006) Roles for Gcn5p and Ada2p in transcription and nucleotide excision repair at the Saccharomyces cerevisiae MET16 gene. Nucleic Acids Res 34:976–985. https://doi.org/10.1093/nar/gkj501
Flowers TJ, Yeo AR (1995) Breeding for salinity resistance in crop plants—where next? Australian J Plant Physiol 22:875–884. https://doi.org/10.1071/PP9950875
Gao XH, Ren ZH, Zhao YX, Zhang H (2003) Overexpression of SOD2 increases salt tolerance of Arabidopsis. Plant Physiol 133:1873–1881. https://doi.org/10.1104/pp.103.026062
Gévaudant F, Duby G, von Stedingk E, Zhao R, Morsomme P, Boutry M (2007) Expression of a constitutively activated plasma membrane H+-ATPase alters plant development and increases salt tolerance. Plant Physiol 144:1763–1776. https://doi.org/10.1104/pp.107.103762
Gusakov AV (2011) Alternatives to Trichoderma reesei in biofuel production. Trends Biotechnol 29:419–425. https://doi.org/10.1016/j.tibtech.2011.04.004
Harman GE, Howell CR, Viterbo A, Chet I, Lorito M (2004) Trichoderma species: opportunistic, avirulent plant symbionts. Nat Rev Microbiol 2:43–56. https://doi.org/10.1038/nrmicro797
Haruta M, Sussma MR (2012) The effect of a genetically reduced plasma membrane proton motive force on vegetative growth of Arabidopsis. Plant Physiol 158:1158–1171. https://doi.org/10.1104/pp.111.189167
Hasegawa PM (2013) Sodium (Na+) homeostasis and salt tolerance of plants. Environ Exp Bot 92:19–31. https://doi.org/10.1016/j.envexpbot.2013.03.001
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499. https://doi.org/10.1146/annurev.arplant.51.1.463
Hermosa R, Viterbo A, Chet L, Monte E (2012) Plant-beneficial effects of Trichoderma and of its genes. Microbiology 158:17–25. https://doi.org/10.1099/mic.0.052274-0
Huh GH, Damsz B, Matsumoto TK, Reddy MP, Rus AM, Ibeas JI, Narasimhan ML, Bressan RA, Hasegawa PM (2002) Salt causes ion disequilibrium-induced programmed cell death in yeast and plants. Plant J 29:649–659. https://doi.org/10.1046/j.0960-7412.2001.01247.x
Jenuwein T, Allis CD (2001) Translating the histone code. Science 293:1074–1080. https://doi.org/10.1126/science.1063127
Kim JY, Yang W, Forner J, Lohmann JU, Noh B, Noh YS (2018) Epigenetic reprogramming by histone acetyltransferase HAG1/AtGCN5 is required for pluripotency acquisition in Arabidopsis. EMBO J 37:e98726. https://doi.org/10.15252/embj.201798726
Kishkevich A, Cooke SL, Harris MRA, Bruin RD (2019) Gcn5 and Rpd3 have a limited role in the regulation of cell cycle transcripts during the G1 and S phases in Saccharomyces cerevisiae. Sci Rep 9:10686. https://doi.org/10.1038/s41598-019-47170-z
Kronzucker HJ, Britto DT (2010) Sodium transport in plants: a critical review. New Phytol 189:54–81. https://doi.org/10.1111/j.1469-8137.2010.03540.x
Lee KK, Workman JL (2007) Histone acetyltransferase complexes: one size doesn’t fit all. Nat Rev Mol Cell Biol 8:284–295. https://doi.org/10.1038/nrm2145
Marcel N, Boysen JM, Thomas K, Knig CC, Falk H, Thomas M, Brakhage AA (2021) Yeast two-hybrid screening reveals a dual function for the histone acetyltransferase GcnE by controlling glutamine synthesis and development in Aspergillus fumigatus. Curr Genet 65:523–538. https://doi.org/10.1007/s00294-018-0891-z
Marmorstein R, Zhou MM (2014) Writers and readers of histone acetylation: structure, mechanism, and inhibition. CSH Persp Biol 6:a018762. https://doi.org/10.1101/cshperspect.a018762
Mello-de-Sousa TM, Rassinger A, Pucher ME (2015) The impact of chromatin remodelling on cellulase expression in Trichoderma reesei. BMC Genomics 16:588. https://doi.org/10.1186/s12864-015-1807-7
Mittler R, Vanderauwera S, Suzuki N, Miller G, Tognetti VB, Vandepoele K (2011) ROS signaling: the new wave? Trends Plant Sci. https://doi.org/10.1016/j.tplants.2011.03.007.0-309
Mohamed ALA, Haggag WM (2006) Biocontrol potential of salinity tolerant mutants of Trichoderma harzianum against Fusarium oxysporum Potencial de biocontrole de mutantes sal-tolerantes de Trichoderma harzianum contra Fusarium oxysporum. Braz J Microbiol. https://doi.org/10.1590/S1517-83822006000200016
Mukherjee PK, Horwitz BA, Herrera-Estrella A, Schmoll M, Kenerley CM (2013) Trichoderma research in the genome era. Annu Rev Phytopathol 51:105–129. https://doi.org/10.1146/annurev-phyto-082712-102353
Mutlu B, Puigserver P (2020) Gcn5 acetyltransferase in cellular energetic and metabolic processes. BBA Gene Regul Mech 1864:194626. https://doi.org/10.1016/j.bbagrm.2020.194626
Nakari-Setälä T, Penttilä M (1995) Production of Trichoderma reesei cellulases on glucose-containing media. Appl Environ Microbio l 61:3650–3655. https://doi.org/10.1007/BF00871823
Okumura M, Inoue S, Kuwata K, Kinoshita T (2016) Photosynthesis activates plasma membrane H+-ATPase via sugar accumulation. Plant Physiol 171:580–589. https://doi.org/10.1104/pp.16.00355
Omoto E, Nagao H, Taniguchi M, Miyake H (2013) Localization of reactive oxygen species and change of antioxidant capacities in mesophyll and bundle sheath chloroplasts of maize under salinity. Physiol Plantarum 149:1–12. https://doi.org/10.1111/ppl.12017
Palmgren MG (2001) Plant plasma membrane H+-ATPases: powerhouses for nutrient uptake. Annu Rev Plant Physiol 52:817–845. https://doi.org/10.1146/annurev.arplant.52.1.817
Pii Y, Mimmo T, Tomasi N, Terzano R, Cesco S, Crecchio C (2015) Microbial interactions in the rhizosphere: beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process: a review. Biol Fert Soil 51:403–415. https://doi.org/10.1007/s00374-015-0996-1
Poças-Fonseca MJ, Cabral CG, Manfrão-Netto JHC (2020) Epigenetic manipulation of filamentous fungi for biotechnological applications: a systematic review. Biotechnol Lett 42:885–904. https://doi.org/10.1007/s10529-020-02871-8
Qiu N, Chen M, Guo J, Bao H, Ma X (2007) Coordinate up-regulation of V-H+-ATPase and vacuolar Na+/H+ antiporter as a response to NaCl treatment in a C3 halophyte Sueda salsa. Plant Sci 172:1218–1225. https://doi.org/10.1007/s11104-018-3679-5
Rosier A, Flávio HV, Bais HP (2018) Defining plant growth promoting rhizobacteria molecular and biochemical networks in beneficial plant-microbe interactions. Plant Soil 428:35–55. https://doi.org/10.1007/s11104-018-3679-5
Schuster A, Schmoll M (2010) Biology and biotechnology of Trichoderma. Appl Microbiol Biot 87:787–799. https://doi.org/10.1007/s00253-010-2632-1
Seiboth B, Karimi RA, Phatale PA, Linke R (2012) The putative protein methyltransferase LAE1 controls cellulase gene expression in Trichoderma reesei. Mol Microbiol 84:1150–1164. https://doi.org/10.1111/j.1365-2958.2012.08083.x
Servet C, Silva NCE, Zhou DX (2010) Histone acetyltransferase AtGCN5/HAG1 is a versatile regulator of developmental and inducible gene expression in Arabidopsis. Mol Plant 3:670–677. https://doi.org/10.1093/mp/ssq018
Shabala S, Cuin TA (2008) Potassium transport and plant salt tolerance. Physiol Plantarum 133:651–669. https://doi.org/10.1111/j.1399-3054.2007.01008.x
Shabala S (2009) Salinity and programmed cell death: unraveling mechanisms for ion specific signaling. J Exp Bot 60:709–712. https://doi.org/10.1093/jxb/erp013
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 135:1–9. https://doi.org/10.1155/2012/217037
Silva P, Geros H (2009) Regulation by salt of vacuolar H+-ATPase and H+-pyrophosphatase activities and Na+/H+ exchange. Plant Signal Behav 4:718–726. https://doi.org/10.4161/psb.4.8.9236
Studt L, Schmidt FJ, Jahn L, Sieber CMK, Connolly LR, Niehaus EM, Freitag M, Humpf HU, Tudzynski B (2013) Two histone deacetylases, FfHda1 and FfHda2, are important for Fusarium fujikuroi secondary metabolism and virulence. Appl Environ Microbiol 79:7719–7734. https://doi.org/10.1128/AEM.01557-13
Talbot NJ (2007) Fungal genomics goes industrial. Nat Biotechnol 25:542–543. https://doi.org/10.1038/nbt0507-542
Teng Y, Yu Y, Waters R (2002) The Saccharomyces cerevisiae histone acetyltransferase GCN5 has a role in the photoreactivation and nucleotide excision repair of UV-induced cyclobutane pyrimidine dimers in the MFA2 gene. J Mol Biol 316:489–499. https://doi.org/10.1006/jmbi.2001.5383
Vinale F, Sivasithamparam K, Ghisalberti EL, Marra R, Barbetti MJ, Li H, Woo SL, Lorito M (2008) A novel role for Trichoderma secondary metabolites in the interactions with plants. Physiol Mol Plant P 72:80–86. https://doi.org/10.1016/j.pmpp.2008.05.005
Wang J, Bao JT, Li XR, Liu YB (2010) Salt stress induced apoptotic-like changes in Thellungiella halophila. Sci Cold Arid Reg 2: 31–42. CNKI:SUN:HAQK.0.2010–01–006
Wang L, Mizzen C, Ying C, Candau R, Barlev N, Brownell J, Allis CD, Berger SL (1997) Histone acetyltransferase activity is conserved between yeast and human GCN5 and is required for complementation of growth and transcriptional activation. Mol Cell Biol 17:519–527. https://doi.org/10.1128/MCB.17.1.519
Wang T, **ng J, Liu Z, Zheng M, Yao Y, Hu ZR, Peng HR, **n MM, Zhou DX, Ni ZF (2019) Histone acetyltransferase gcn5-mediated regulation of long non-coding RNA At4 contributes to phosphate starvation response in Arabidopsis. J Exp Bot 70:6337–6348. https://doi.org/10.1093/jxb/erz359
**n Q, Gong Y, Lv X, Chen G, Liu W (2013) Trichoderma reesei histone acetyltransferase GCN5 regulates fungal growth, conidiation, and cellulase gene expression. Curr Microbiol 67:580–589. https://doi.org/10.1007/s00284-013-0396-4
Xu W, Shi W, Jia L, Liang J, Zhang J (2012) Tft6, and Tft7, two different members of tomato 14–3-3 gene family, play distinct roles in plant adaption to low phosphorus stress. Plant Cell Environ 7:1047–1048. https://doi.org/10.1111/j.1365-3040.2012.02497.x
Yamaguchi T, Blumwald E (2005) Develo** salt-tolerant crop plants: challenges and opportunities. Trends Plant Sci 10:615–620. https://doi.org/10.1016/j.tplants.2005.10.002
Yamauchi S, Shimazaki K (2017) Determination of H+-ATPase activity in Arabidopsis guard cell protoplasts through H+-pum** measurement and H+-ATPase quantification. Bio-protocol 7:e2653. https://doi.org/10.21769/BioProtoc.2653
Yamauchi S, Takemiya A, Sakamoto T, Kurata T, Tsutsumi T, Kinoshita T, Shimazaki K (2016) The plasma membrane H+-ATPase AHA1 plays a major role in stomatal opening in response to blue light. Plant Physiol 171:2731–2743. https://doi.org/10.1104/pp.16.01581
Zeilinger S, Omann M (2007) Trichoderma biocontrol: signal transduction pathways involved in host sensing and mycoparasitism. Gene Regul Syst Biol 1:227–234. https://doi.org/10.1007/s002880050558
Zhang JL, Tang L, Huang R, Li Z, Wei L, Jiang L, Liu C, Yu X, Zhu HW, Chen GZ, Zhang XX (2021) Trichoderma: A treasure house of structurally diverse secondary metabolites with medicinal importance. Front Microbiol 12:723828. https://doi.org/10.3389/fmicb.2021.723828
Zhang X, Zhou S, Fu Y, Su Z, Wang X, Sun C (2006) Identification of a drought tolerant introgression line derived from Dongxiang common wild rice (O. rufipogon Griff.). Plant Mol Biol 62:247–259. https://doi.org/10.1007/s11103-006-9018-x
Zhao S, Zhang X, Li HT (2018) Beyond histone acetylation-writing and erasing histone acylations. Curr Opin Struc Biol 53:169–177. https://doi.org/10.1016/j.sbi.2018.10.001
Zheng M, Liu X, Lin JC, Liu XY, Wang ZY, **n MM, Yao YY, Peng HR, Zhou DX, Ni ZF, Sun QX, Hu ZR (2018) Histone acetyltransferase GCN5 contributes to cell wall integrity and salt stress tolerance by altering expression of cellulose synthesis genes. Plant J 97:587–602. https://doi.org/10.1111/tpj.14144
Funding
This research is supported by the National Natural Science Foundation of China (21607169; 41977125; 41907033; 21254356), the National Natural Science Foundation of Shandong Province (ZR202102270296), the Natural Science Foundation of Guangdong Province (2019A1515011948), the Youth Science Funds of Shandong Academy of Sciences (2020QN0019), the Program for Innovation Team in the University of **an (2019GXRC033), the Key R&D Program of Yantai (2019ZDCX016), and the Special Science and Technology Project of Rizhao (2019CXZX2205).
Author information
Authors and Affiliations
Contributions
ZL conceived the project; ZL and ZL designed the experiments; ZL, HZ, CJC, ZZ, JC, and KG carried out the experiments; ZL and HZ analyzed the data; ZL, HZ, and ZL wrote the article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Competing interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Z., Zhang, H., Cai, C. et al. Histone acetyltransferase GCN5-mediated lysine acetylation modulates salt stress aadaption of Trichoderma. Appl Microbiol Biotechnol 106, 3033–3049 (2022). https://doi.org/10.1007/s00253-022-11897-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-11897-z