Abstract
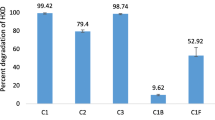
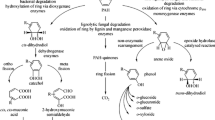
Polycyclic aromatic hydrocarbons (PAHs) and volatile organic compounds (VOCs) are important indoor contaminants. Their hydrophobic nature hinders the possibility of biological abatement using biofiltration. Our aim was to establish whether the use of a consortium of Fusarium solani and Rhodococcus erythropolis shows an improved performance (in terms of mineralization rate and extent) towards the degradation of formaldehyde, as a slightly polar VOC; toluene, as hydrophobic VOC; and benzo[α]pyrene (BaP) as PAH at low concentrations compared to a single-species biofilm in serum bottles with vermiculite as solid support to mimic a biofilter and to relate the possible improvements with the surface hydrophobicity and partition coefficient of the biomass at three different temperatures. Results showed that the hydrophobicity of the surface of the biofilms was affected by the hydrophobicity of the carbon source in F. solani but it did not change in R. erythropolis. Similarly, the partition coefficients of toluene and BaP in F. solani biomass (both as pure culture and consortium) show a reduction of up to 38 times compared to its value in water, whereas this reduction was only 1.5 times in presence of R. erythropolis. Despite that increments in the accumulated CO2 and its production rate were found when F. solani or the consortium was used, the mineralization extent of toluene was below 25%. Regarding BaP degradation, the higher CO2 production rates and percent yields were obtained when a consortium of F. solani and R. erythropolis was used, despite a pure culture of R. erythropolis exhibits poor mineralization of BaP.



Similar content being viewed by others
References
Aguiar L, Mendes A, Pereira C, Neves P, Mendes D, Teixeira JP (2014) Biological air contamination in elderly care centers: Geria project. J Toxicol Environ Heal Part A 77:944–958. doi:10.1080/15287394.2014.911135
Alinejad A, Zamir SM, Shojaosadati SA (2017) Different strategies for transient-state operation of a biotrickling filter treating toluene vapor. Appl Microbiol Biotechnol. doi:10.1007/s00253-016-8075-6
Arriaga S, Revah S (2005a) Removal of n-hexane by Fusarium solani with a gas-phase biofilter. J Ind Microbiol Biotechnol 32:548–553. doi:10.1007/s10295-005-0247-9
Arriaga S, Revah S (2005b) Improving hexane removal by enhancing fungal development in a microbial consortium biofilter. Biotechnol Bioeng 90:107–115. doi:10.1002/bit.20424
Barro R, Regueiro J, Llompart M, Garcia-Jares C (2009) Analysis of industrial contaminants in indoor air: part 1. Volatile organic compounds, carbonyl compounds, polycyclic aromatic hydrocarbons and polychlorinated biphenyls. J Chromatogr A 1216:540–566. doi:10.1016/j.chroma.2008.10.117
Boonchan S, Britz ML, Stanley GA (2000) Degradation and mineralization of high molecular weight polycyclic aromatic hydrocarbons by defined fungal bacterial cocultures. Appl Environ Microbiol 66:1007–1019. doi:10.1128/AEM.66.3.1007-1019.2000
Brown SK, Sim MR, Abramson MJ, Gray CN (1994) Concentrations of volatile organic compounds in indoor air—a review. Indoor Air 4:123–134. doi:10.1111/j.1600-0668.1994.t01-2-00007.x
Burge PS (2004) Sick building syndrome. Occup Environ Med 61:185–190. doi:10.1136/oem.2003.008813
Cavalieri EL, Rogan EG (1995) Central role of radical cations in metabolic activation of polycyclic aromatic hydrocarbons. Xenobiotica 25:677–688. doi:10.3109/00498259509061885
Cheng Z, Lu L, Kennes C, Yu J, Chen J (2016) Treatment of gaseous toluene in three biofilters inoculated with fungi/bacteria: microbial analysis, performance and starvation response. J Hazard Mater 303:83–93. doi:10.1016/j.jhazmat.2015.10.017
Colome S, McCunney RJ, Samet JM, Swankin D (1996) Indoor air pollution: an introduction for health professionals. American Lung Association, the American Medical Association, the U.S. Consumer Product Safety Commission, and the U.S. Environmental Protection Agency. Washington (DC), pp 1–22
Davison BH, Barton JW, Klasson KT, Francisco AB (2000) Influence of high biomass concentrations on alkane solubilities. Biotechnol Bioeng 68:279–284. doi:10.1002/(SICI)1097-0290(20000505)68:3<279::AID-BIT6>3.0.CO;2-P
De Carvalho CCCR, Fatal V, Alves SS, Da Fonseca MMR (2007) Adaptation of Rhodococcus erythropolis cells to high concentrations of toluene. Appl Microbiol Biotechnol 76:1423–1430. doi:10.1007/s00253-007-1103-9
Deeb RA, Alvarez-Cohen L (1999) Temperature effects and substrate interactions during the aerobic biotransformation of BTEX mixtures by toluene-enriched consortia and Rhodococcus rhodochrous. Biotechnol Bioeng 62:526–536. doi:10.1002/(SICI)1097-0290(19990305)62:5<526::AID-BIT4>3.0.CO;2-8
Deshusses MA, Hamer G, Dunn IJ (1995) Behavior of biofilters for waste air biotreatment. 1. Dynamic model development. Environ Sci Technol 29:1048–1058. doi:10.1021/es00004a027
Díaz-Robles LA, Fu JS, Vergara-Fernández A, Etcharren P, Schiappacasse LN, Reed GD, Silva MP (2014) Health risks caused by short term exposure to ultrafine particles generated by residential wood combustion: a case study of Temuco, Chile. Environ Int 66:174–181. doi:10.1016/j.envint.2014.01.017
Estévez E, Veiga MC, Kennes C (2005) Biodegradation of toluene by the new fungal isolates Paecilomyces variotii and Exophiala oligosperma. J Ind Microbiol Biotechnol 32:33–37. doi:10.1007/s10295-004-0203-0
Estrada JM, Hernández S, Muñoz R, Revah S (2013) A comparative study of fungal and bacterial biofiltration treating a VOC mixture. J Hazard Mater 250–251:190–197. doi:10.1016/j.jhazmat.2013.01.064
Fayeulle A, Veignie E, Slomianny C, Dewailly E, Munch JC, Rafin C (2014) Energy-dependent uptake of benzo[a]pyrene and its cytoskeleton-dependent intracellular transport by the telluric fungus Fusarium solani. Environ Sci Pollut Res 21:3515–3523. doi:10.1007/s11356-013-2324-3
Goldman R, Enewold L, Pellizzari E, Beach JB, Bowman ED, Krishnan SS, Shields PG (2001) Smoking increases carcinogenic polycyclic aromatic hydrocarbons in human lung tissue. Cancer Res 61:6367–6371
Heitkamp MA, Franklin W, Cerniglia CE (1988) Microbial metabolism of polycyclic aromatic hydrocarbons: isolation and characterization of a pyrene-degrading bacterium. Appl Environ Microbiol 54:2549–2555
Hidalgo A, Lopategi M, Prieto JA (2002) Formaldehyde removal in synthetic and industrial wastewater by Rhodococcus erythropolis UPV-1. Appl Microbiol Biotechnol 58:260–264. doi:10.1007/s00253-001-0876-5
** Y, Veiga M, Kennes C (2006) Performance optimization of the fungal biodegradation of α-pinene in gas-phase biofilter. Process Biochem 41:1722–1728. doi:10.1016/j.procbio.2006.03.020
Juteau P, Larocque R, Rho D, LeDuy A (1999) Analysis of the relative abundance of different types of bacteria capable of toluene degradation in a compost biofilter. Appl Microbiol Biotechnol 52:863–868
Kamisaka Y, Noda N (2001) Intracellular transport of phosphatidic acid and phosphatidylcholine into lipid bodies in an oleaginous fungus, Mortierella ramanniana var. angulispora. J Biochem 129:19–26
Liu K, Han W, Pan W-P, Riley JT (2001) Polycyclic aromatic hydrocarbon (PAH) emissions from a coal-fired pilot FBC system. J Hazard Mater 84:175–188. doi:10.1016/S0304-3894(01)00196-0
Luengas A, Barona A, Hort C, Gallastegui G, Platel V, Elias A (2015) A review of indoor air treatment technologies. Rev Environ Sci Bio/Technology 14:499–522. doi:10.1007/s11157-015-9363-9
Malhautier L, Quijano G, Avezac M, Rocher J, Fanlo JL (2014) Kinetic characterization of toluene biodegradation by Rhodococcus erythropolis: towards a rationale for microflora enhancement in bioreactors devoted to air treatment. Chem Eng J 247:199–204. doi:10.1016/j.cej.2014.02.099
Mendes A, Aelenei D, Papoila AL, Carreiro-Martins P, Aguiar L, Pereira C, Neves P, Azevedo S, Cano M, Proença C, Viegas J, Silva S, Mendes D, Neuparth N, Teixeira JP (2014) Environmental and ventilation assessment in child day care centers in Porto: the Envirh project. J Toxicol Environ Heal Part A 77:931–943. doi:10.1080/15287394.2014.911134
Meulenberg R, Rijnaarts HH, Doddema HJ, Field JA (1997) Partially oxidized polycyclic aromatic hydrocarbons show an increased bioavailability and biodegradability. FEMS Microbiol Lett 152:45–49
Mishra S, Singh SN (2014) Biodegradation of benzo(a)pyrene mediated by catabolic enzymes of bacteria. Int J Environ Sci Technol 11:1571–1580. doi:10.1007/s13762-013-0300-6
Mohee R, Mudhoo A (2012) Bioremediation and sustainability. John Wiley & Sons, Inc., Hoboken, NJ
Mudliar S, Giri B, Padoley K, Satpute D, Dixit R, Bhatt P, Pandey R, Juwarkar A, Vaidya A (2010) Bioreactors for treatment of VOCs and odours—a review. J Environ Manag 91:1039–1054. doi:10.1016/j.jenvman.2010.01.006
Naumova YY, Eisenreich SJ, Turpin BJ, Weisel CP, Morandi MT, Colome SD, Totten LA, Stock TH, Winer AM, Alimokhtari S, Kwon J, Shendell D, Jones J, Maberti S, Wall SJ (2002) Polycyclic aromatic hydrocarbons in the indoor and outdoor air of three cities in the U.S. Environ Sci Technol 36:2552–2559. doi:10.1021/es015727h
Pino-Cortés E, Díaz-Robles LA, Cubillos F, Fu JS, Vergara-Fernández A (2015) Sensitivity analysis of biodiesel blends on benzo[a]pyrene and main emissions using MOVES: a case study in Temuco, Chile. Sci Total Environ 537:352–359. doi:10.1016/j.scitotenv.2015.07.143
Pozo KK, Estellano VH, Harner T, Diaz-Robles L, Cereceda-Balic F, Etcharren P, Pozo KK, Vidal V, Guerrero F, Vergara-Fernández A (2015) Assessing polycyclic aromatic hydrocarbons (PAHs) using passive air sampling in the atmosphere of one of the most wood-smoke-polluted cities in Chile: the case study of Temuco. Chemosphere 134:475–481. doi:10.1016/j.chemosphere.2015.04.077
Prado O, Veiga MC, Kennes C (2004) Biofiltration of waste gases containing a mixture of formaldehyde and methanol. Appl Microbiol Biotechnol. doi:10.1007/s00253-004-1616-4
Rafin C, Veignie E, Woisel P, Cazier F, Surpateanu G (2006) New potential of a Deuteromycete fungus Fusarium solani in benzo[a] pyrene degradation: an eco-physiological hypothesis? In: Glazer MP (ed) New frontiers in environmental research. Nova Science Publishers, Inc., New York, pp 165–181
Revah S, Vergara-Fernández A, Hernández S (2011) Fungal Biofiltration for the Elimination of Gaseous Pollutants from Air. In: Mycofactories. Bentham Science Publishers Ltds, pp 109–120
Sack U, Hofrichter M, Fritsche W (1997) Degradation of polycyclic aromatic hydrocarbons by manganese peroxidase of Nematoloma frowardii. FEMS Microbiol Lett 152:227–234
Sander R (2015) Compilation of Henry’s law constants (version 4.0) for water as solvent. Atmos Chem Phys 15:4399–4981. doi:10.5194/acp-15-4399-2015
Smits THM, Wick LY, Harms H, Keel C (2003) Characterization of the surface hydrophobicity of filamentous fungi. Environ Microbiol 5:85–91. doi:10.1046/j.1462-2920.2003.00389.x
Song X, Xu Y, Li G, Zhang Y, Huang T, Hu Z (2011) Isolation, characterization of Rhodococcus sp. P14 capable of degrading high-molecular-weight polycyclic aromatic hydrocarbons and aliphatic hydrocarbons. Mar Pollut Bull 62:2122–2128. doi:10.1016/j.marpolbul.2011.07.013
Tsapakis M, Lagoudaki E, Stephanou EG, Kavouras IG, Koutrakis P, Oyola P, von Baer D (2002) The composition and sources of PM2.5 organic aerosol in two urban areas of Chile. Atmos Environ 36:3851–3863. doi:10.1016/S1352-2310(02)00269-8
van der Geize R, Hessels GI, Dijkhuizen L (2002) Molecular and functional characterization of the kstD2 gene of Rhodococcus erythropolis SQ1 encoding a second 3-ketosteroid Δ1-dehydrogenase isoenzyme. Microbiology 148:3285–3292. doi:10.1099/00221287-148-10-3285
Veignie E, Rafin C, Woisel P, Sahraoui AL-H, Cazier F (2002) Metabolization of the polycyclic aromatic hydrocarbon benzo(a)pyrene by a non-white rot fungus (Fusarium solani) in a batch reactor. Polycycl Aromat Compd 22:87–97. doi:10.1080/10406630210372
Veignie E, Vinogradov E, Sadovskaya I, Coulon C, Rafin C (2012) Preliminary characterizations of a carbohydrate from the concentrated culture filtrate from Fusarium solani and its role in benzo[a]pyrene solubilization. Adv Microbiol 2:375–381. doi:10.4236/aim.2012.23047
Verdin A, Lounès-Hadj Sahraoui A, Newsam R, Robinson G, Durand R (2005) Polycyclic aromatic hydrocarbons storage by Fusarium solani in intracellular lipid vesicles. Environ Pollut 133:283–291. doi:10.1016/j.envpol.2004.05.040
Vergara-Fernández A, Van Haaren B, Revah S (2006) Phase partition of gaseous hexane and surface hydrophobicity of Fusarium solani when grown in liquid and solid media with hexanol and hexane. Biotechnol Lett 28:2011–2017. doi:10.1007/s10529-006-9186-4
Vergara-Fernández A, Hernández S, Revah S (2008) Phenomenological model of fungal biofilters for the abatement of hydrophobic VOCs. Biotechnol Bioeng 101:1182–1192. doi:10.1002/bit.21989
Vergara-Fernández A, Hernández S, San Martín-Davison J, Revah S (2011) Morphological characterization of aerial hyphae and simulation growth of Fusarium solani under different carbon source for application in the hydrophobic VOCs biofiltration. Rev Mex Ing Química 10:225–233
Vergara-Fernandez A, Soto-Sanchez O, Vasquez J (2011) Effects of packing material type on n-pentane/biomass partition coefficient for use in fungal biofilters. Chem Biochem Eng Q 25:439–444
Vergara-Fernández A, Scott F, Moreno-Casas P, Diaz-Robles L, Muñoz R (2016) Elucidating the key role of the fungal mycelium on the biodegradation of n-pentane as a model hydrophobic VOC. Chemosphere. doi:10.1016/j.chemosphere.2016.05.034
Vigueras G, Shirai K, Martins D, Franco TT, Fleuri LF, Revah S (2008) Toluene gas phase biofiltration by Paecilomyces lilacinus and isolation and identification of a hydrophobin protein produced thereof. Appl Microbiol Biotechnol 80:147–154. doi:10.1007/s00253-008-1490-6
Wessels JGH (1996) Hydrophobins: proteins that change the nature of the fungal surface. Adv Microb Physiol 38:1–45. doi:10.1016/S0065-2911(08)60154-X
Williams ES, Mahler BJ, Van Metre PC (2013) Cancer risk from incidental ingestion exposures to PAHs associated with coal-tar-sealed pavement. Environ Sci Technol 47:1101–1109. doi:10.1021/es303371t
Yu C, Crump D (1998) A review of the emission of VOCs from polymeric materials used in buildings. Build Environ 33:357–374. doi:10.1016/S0360-1323(97)00055-3
Acknowledgements
The present research has been sponsored by CONICYT—Chile (National Commission for Scientific and Technological Research) (FONDEF IDeA in two stages, No. ID4i10130). We thank Dr. Sergio Revah, Metropolitan Autonomous University, Mexico, for providing the Fusarium solani B1 (CBS 117476) strain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Morales, P., Cáceres, M., Scott, F. et al. Biodegradation of benzo[α]pyrene, toluene, and formaldehyde from the gas phase by a consortium of Rhodococcus erythropolis and Fusarium solani . Appl Microbiol Biotechnol 101, 6765–6777 (2017). https://doi.org/10.1007/s00253-017-8400-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8400-8