Abstract
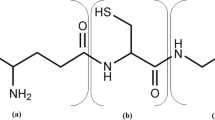
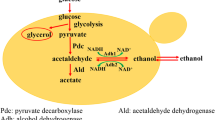
Saccharomyces cerevisiae maintains a redox balance under fermentative growth conditions by re-oxidizing NADH formed during glycolysis through ethanol formation. Excess NADH stimulates the synthesis of mainly glycerol, but also of other compounds. Here, we investigated the production of primary and secondary metabolites in S. cerevisiae strains where the glycerol production pathway was inactivated through deletion of the two glycerol-3-phosphate dehydrogenases genes (GPD1/GPD2) and replaced with alternative NAD+-generating pathways. While these modifications decreased fermentative ability compared to the wild-type strain, all improved growth and/or fermentative ability of the gpd1Δgpd2Δ strain in self-generated anaerobic high sugar medium. The partial NAD+ regeneration ability of the mutants resulted in significant amounts of alternative products, but at lower yields than glycerol. Compared to the wild-type strain, pyruvate production increased in most genetically manipulated strains, whereas acetate and succinate production decreased in all strains. Malate production was similar in all strains. Isobutanol production increased substantially in all genetically manipulated strains compared to the wild-type strain, whereas only mutant strains expressing the sorbitol producing SOR1 and srlD genes showed increases in isoamyl alcohol and 2-phenyl alcohol. A marked reduction in ethyl acetate concentration was observed in the genetically manipulated strains, while isobutyric acid increased. The synthesis of some primary and secondary metabolites appears more readily influenced by the NAD+/NADH availability. The data provide an initial assessment of the impact of redox balance on the production of primary and secondary metabolites which play an essential role in the flavour and aroma character of beverages.






Similar content being viewed by others
References
Albertyn J, Hohmann S, Thevelein JM, Prior BA (1994) GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol Cell Biol 14:4135–4144
Ansell R, Granath K, Hohmann S, Thevelein JM, Adler L (1997) The two isoenzymes for yeast NAD+-dependent glycerol 3-phosphate dehydrogenase encoded by GPD1 and GPD2 have distinct roles in osmoadaptation and redox regulation. EMBO J 16:2179–2187
Bach B, Meudec E, Lepoutre JP, Rossignol T, Blondin B, Dequin S, Camarasa C (2009) New insights into γ-aminobutyric acid catabolism: evidence for γ-hydroxybutyric acid and polyhydroxybutyrate synthesis in Saccharomyces cerevisiae. Appl Environ Microbiol 75:4231–4239
Berben G, Dumont J, Gilliquet V, Bolle PA, Hilger F (1991) The YDp plasmids: a uniform set of vectors bearing versatile gene disruption cassettes for Saccharomyces cerevisiae. Yeast 7:475–477
Boulton RB, Singleton VL, Bisson LF, Kunkee RE (1996) Principle and practices of winemaking. Chapman and Hall, New York
Bro C, Regenberg B, Förster J, Nielsen J (2006) In silico aided metabolic engineering of Saccharomyces cerevisiae for improved bioethanol production. Metab Eng 8:102–111
Camarasa C, Grivet JP, Dequin S (2003) Investigation by 13C-NMR and tricarboxylic acid (TCA) deletion mutant analysis of pathways for succinate formation in Saccharomyces cerevisiae during anaerobic fermentation. Microbiol 149:2669–2678
Eriksson P, Andre L, Ansell R, Blomberg A, Adler L (1995) Cloning and characterization of GPD2, a second gene encoding glycerol 3-phosphate dehydrogenase (NAD+) in Saccharomyces cerevisiae and its comparison with GPD1. Mol Microbiol 17:95–107
Gietz RD, Schiestl RH (2007) High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat Protoc 2:31–34
Gordon JH, Dubos R (1970) The anaerobic bacterial flora of the mouse cecum. J Exp Med 132:251–260
Gururajan VT, Gorwa-Grauslund MF, Hahn-Hägerdal B, Pretorius IS, Cordero Otero RR (2007) A constitutive catabolite repression mutant of a recombinant Saccharomyces cerevisiae strain improves xylose consumption during fermentation. Ann Microbiol 57:85–92
Harju S, Fedosyuk H, Peterson KR (2004) Rapid isolation of yeast genomic DNA: Bust n' Grab. BMC Biotech 4:8
Hazelwood LA, Daran JM, van Maris AJA, Pronk JT, Dickinson JR (2008) The Ehrlich pathway for fusel alcohol production: a century of research on Saccharomyces cerevisiae metabolism. Appl Environ Microbiol 74:2259–2266
Jain VK, Divol B, Prior BA, Bauer FF (2011) Elimination of glycerol and replacement with alternative products in ethanol fermentation by Saccharomyces cerevisiae. J Ind Microbiol Biotechnol. doi:10.1007/s10295-010-0928-x
Kalapos MP (1999) Methylglyoxal in living organisms: chemistry, biochemistry, toxicology and biological implications. Toxicol Lett 110:145–175
Kuhn A, van Zyl C, van Tonder A, Prior BA (1995) Purification and partial characterization of an aldo-keto reductase from Saccharomyces cerevisiae. Appl Environ Microbiol 61:1580–1585
Lambrechts MG, Pretorius IS (2000) Yeast and its importance to wine aroma: a review. S Afr J Enol Vitic 21:97–129
Larsson C, Pahlman IL, Ansell R, Rigoulet M, Adler L, Gustafsson L (1998) The importance of the glycerol 3-phosphate shuttle during aerobic growth of Saccharomyces cerevisiae. Yeast 14:347–357
Louw L, Roux K, Tredoux A, Tomic O, Naes T, Nieuwoudt HH, van Rensburg P (2009) Characterization of selected South African young cultivar wines using FTMIR spectroscopy, gas chromatography, and multivariate data analysis. J Agric Food Chem 57:2623–2632
Malherbe DF (2010) Characterization and evaluation of glucose oxidase activity in recombinant Saccharomyces cerevisiae strains. PhD Dissertation, Stellenbosch University
Mulligan KJ (1996) A procedure to determine diethylene glycol (2,2'-oxybisethanol) and ethylene glycol (1,2-ethanediol) in glycerin and selected products. Lab Inform Bull 12:1–7
Murata K, Saikusa T, Fukuda Y, Watanabe K, Inoue Y, Shimosaka M, Kimura A (1986) Metabolism of 2-oxoaldehydes in yeasts. Possible role of glycolytic bypath as a detoxification system in L-threonine catabolism by Saccharomyces cerevisiae. Eur J Biochem 157:297–301
Ng LK (2002) Analysis by gas chromatography/mass spectrometry of fatty acids and esters in alcoholic beverages and tobaccos. Anal Chim Acta 465:309–318
Novotny MJ, Reizer J, Esch F, Saier MH (1984) Purification and properties of D-mannitol-1-phosphate dehydrogenase and D-glucitol-6-phosphate dehydrogenase from Escherichia coli. J Bacteriol 159:986–990
Pronk JT, Yde Steensma H, Van Dijken JP (1996) Pyruvate metabolism in Saccharomyces cerevisiae. Yeast 12:1607–1633
Rossouw D, Naes T, Bauer FF (2008) Linking gene regulation and the exo-metabolome: a comparative transcriptomics approach to identify genes that impact on the production of volatile aroma compounds in yeast. BMC Genomics 9:530
Saint-Prix F, Bonquist L, Dequin S (2004) Functional analysis of the ALD gene family of Saccharomyces cerevisiae during anaerobic growth on glucose: the NADP+-dependent Ald6p and Ald5p isoforms play a major role in acetate formation. Microbiol 150:2209–2220
Schoondermark-Stolk SA, Tabernero M, Chapman J, Ter Schure EG, Verrips CT, Verkleij AJ, Boonstra J (2005) Bat2p is essential in Saccharomyces cerevisiae for fusel alcohol production on the non-fermentable carbon source ethanol. FEMS Yeast Res 5:757–766
van Dijken JP, Scheffers WA (1986) Redox balances in the metabolism of sugars by yeasts. FEMS Microbiol Rev 32:199–224
Vanderhaegen B, Neven H, Coghe S, Verstrepen KJ, Verachtert H, Derdelinckx G (2003) Evolution of chemical and sensory properties during aging of top-fermented beer. J Agric Food Chem 51:6782–6790
Xu Y, Zhao GA, Wang LP (2006) Controlled formation of volatile components in cider making using a combination of Saccharomyces cerevisiae and Hanseniaspora valbyensis yeast species. J Ind Microbiol Biotechnol 33:192–196
Acknowledgements
This work was supported by the National Research Foundation, South Africa. We would like to thank Dr. D. F. Malherbe and Dr. V. T. Gururajan for kindly providing the pDMPM and pSTAH vectors, respectively, for cloning purposes. The authors would also like to thank Dr. A. Tredoux for technical assistance and Prof. Martin Kidd for statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jain, V.K., Divol, B., Prior, B.A. et al. Effect of alternative NAD+-regenerating pathways on the formation of primary and secondary aroma compounds in a Saccharomyces cerevisiae glycerol-defective mutant. Appl Microbiol Biotechnol 93, 131–141 (2012). https://doi.org/10.1007/s00253-011-3431-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3431-z