Abstract
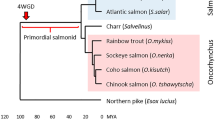
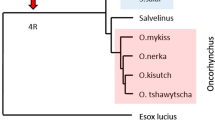
The genes of the major histocompatibility complex (MHC) are amongst the most variable in vertebrates and represent some of the best candidates to study processes of adaptive evolution. However, despite the number of studies available, most of the information on the structure and function of these genes come from studies in mammals and birds in which the MHC class I and II genes are tightly linked and class II alpha exhibits low variability in many cases. Teleost fishes are among the most primitive vertebrates with MHC and represent good organisms for the study of MHC evolution because their class I and class II loci are not physically linked, allowing for independent evolution of both classes of genes. We have compared the diversity and molecular mechanisms of evolution of classical MH class II α and class II β loci in farm populations of three salmonid species: Oncorhynchus kisutch, Oncorhynchus mykiss and Salmo salar. We found single classical class II loci and high polymorphism at both class II α and β genes in the three species. Mechanisms of evolution were common for both class II genes, with recombination and point mutation involved in generating diversity and positive selection acting on the peptide-binding residues. These results suggest that the maintenance of variability at the class IIα gene could be a mechanism to increase diversity in the MHC class II in salmonids in order to compensate for the expression of one single classical locus and to respond to a wider array of parasites.

Similar content being viewed by others
References
Aguilar A, Garza JC (2006) A comparison of variability and population structure for major histocompatibility complex and microsatellite loci in California coastal steelhead (Oncorhynchus mykiss Walbaum). Mol Ecol 15:923–937
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Automat Contr 19:716–723
Anisimova M, Nielsen R, Yang Z (2003) Effect of recombination on the accuracy of the likelihood method for detecting positive selection at amino acid sites. Genetics 164:1229–1236
Bartl S, Baltimore D, Weissman IL (1994) Molecular evolution of the vertebrate immune system. Proc Natl Acad Sci USA 91:10769–10770
Belich M, Madrigal J, Hildebrand W, Zemmour J, Williams R, Luz R, Petzl-Erler M, Parham P (1992) Unusual HLA-B alleles in two tribes of Brazilian Indians. Nature 357:326–329
Bell J, Estess P, John T, Saiki R, Watling D, Erlich H, McDevitt H (1985) DNA sequence and characterization of human class II major histocompatibility complex ss chains from the DR1 haplotype. Proc Natl Acad Sci USA 82:3405–3409
Bernatchez L, Landry C (2003) MHC studies in nonmodel vertebrates: what have we learned about natural selection in 15 years? J Evol Biol 16:363–377
Bingulac-Popovic J, Figueroa F, Sato A, Talbot W, Johnson S, Gates M, Postlethwait J, Klein J (1997) Map** of Mhc class I and class II regions to different linkage groups in the zebrafish, Danio rerio. Immunogenetics 46:129–134
Bjorkman PJ, Saper MA, Samraoui B, Bennett WS, Strominger JL, Wiley DC (1987) Structure of the human class I histocompatibility antigen, HLA-A2. Nature 329:506–512
Bonneaud C, Mazuc J, Chastel O, Westerdahl H, Sorci G (2004) Terminal investment induced by immune challenge and fitness traits associated with major histocompatibility complex in the house sparrow. Evolution 58:2823–2830
Bontrop RE, Otting N, de Groot NG, Doxiadis GGM (1999) Major histocompatibility complex class II polymorphisms in primates. Immunol Rev 167:339–350
Boyson J, Shufflebotham C, Cadavid L, Urvater J, Knapp L, Hughes A, Watkins D (1996) The MHC class I genes of the rhesus monkey. Different evolutionary histories of MHC class I and II genes in primates. J Immunol 156:4656–4665
Conejeros P, Phan A, Power M, Alekseyev S, O’Connell M, Dempson B, Dixon B (2008) MH class IIα polymorphism in local and global adaptation of Arctic charr (Salvelinus alpinus L.). Immunogenetics 60:325–337
Consortium TMS (1999) Complete sequence and gene map of a human major histocompatibility complex. Nature 401:921–923
Consuegra S, Megens HJ, Leon K, Stet RJM, Jordan WC (2005a) Patterns of variability at the major histocompatibility class II alpha locus in Atlantic salmon contrast with those at the class I locus. Immunogenetics 57:16–24
Consuegra S, Megens HJ, Schaschl H, Leon K, Stet RJM, Jordan WC (2005b) Rapid evolution of the MH class I locus results in different allelic compositions in recently diverged populations of atlantic salmon. Mol Biol Evol 22:1095–1106
Croisetière S, Tarte P, Bernatchez L, Belhumeur P (2008) Identification of MHC class II resistance/susceptibility alleles to Aeromonas salmonicida in brook charr (Salvelinus fontinalis). Mol Immunol 45:3107–3116
Dixon B, van Erp S, Rodrigues P, Egberts E, Stet R (1995) Fish major histocompatibility complex genes: an expansion. Dev Comp Immunol 19:109–133
Dorschner M, Duris T, Bronte C, Burnham Curtis M, Phillips R (2000) High levels of MHC class II allelic diversity in lake trout from Lake Superior. J Hered 91:359
Edwards SV, Hedrick PW (1998) Evolution and ecology of MHC molecules: from genomics to sexual selection. Trends Ecol Evol 13:305–311
Egea R, Casillas S, Barbadilla A (2008) Standard and generalized McDonald-Kreitman test: a website to detect selection by comparing different classes of DNA sites. Nucleic Acids Res 36:W157–W162
Flajnik M, Ohta Y, Namikawa-Yamada C, Nonaka M (1999) Insight into the primordial MHC from studies in ectothermic vertebrates. Immunol Rev 167:59
Fu Y, Li W (1993) Statistical tests of neutrality of mutations. Genetics 133:693–709
Garrigan D, Hedrick PW (2003) Perspective: detecting adaptive molecular polymorphism: lessons from the MHC. Evolution 57:1707–1722
Graser R, Vincek V, Takami K, Klein J (1998) Analysis of zebrafish Mhc using BAC clones. Immunogenetics 47:318–325
Grimholt U, Hordvik I, Fosse VM, Olsaker I, Endresen C, Lie Ø (1993) Molecular cloning of major histocompatibility complex class I cDNAs from Atlantic salmon (Salmo salar). Immunogenetics 37:469–473
Grimholt U, Getahun A, Hermsen T, Stet RJM (2000) The major histocompatibility class II alpha chain in salmonid fishes. Dev Comp Immunol 24:751–763
Grimholt U, Drabløs F, Jørgensen S, Høyheim B, Stet R (2002) The major histocompatibility class I locus in Atlantic salmon (Salmo salar L.): polymorphism, linkage analysis and protein modelling. Immunogenetics 54:570–581
Grimholt U, Larsen S, Nordmo R, Midtlyng P, Kjoeglum S, Storset A, Saebo S, Stet RJM (2003) MHC polymorphism and disease resistance in Atlantic salmon (Salmo salar); facing pathogens with single expressed major histocompatibility class I and class II loci. Immunogenetics 55:210–219
Günther E, Walter L (2001) The major histocompatibility complex of the rat (Rattus norvegicus). Immunogenetics 53:520–542
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hansen J, Strassburger P, Thorgaard G, Young W, Du Pasquier L (1999) Expression, linkage, and polymorphism of MHC-related genes in rainbow trout, Oncorhynchus mykiss. J Immunol 163:774
Harstad H, Lukacs M, Bakke H, Grimholt U (2008) Multiple expressed MHC class II loci in salmonids; details of one non-classical region in Atlantic salmon (Salmo salar). BMC Genomics 9:193
Hedrick PW (2002) Pathogen resistance and genetic variation at MHC loci. Evolution 56:1902–1908
Hordvik I, Grimholt U, Fosse VM, Lie Ø, Endresen C (1993) Cloning and sequence analysis of cDNAs encoding the MHC class II β chain in Atlantic salmon (Salmo salar). Immunogenetics 37:437–441
Horton R, Wilming L, Rand V, Lovering R, Bruford E, Khodiyar V, Lush M, Povey S, Talbot C, Wright M (2004) Gene map of the extended human MHC. Nat Rev Genet 5:889–899
Hudson R, Kaplan N (1985) Statistical properties of the number of recombination events in the history of a sample of DNA sequences. Genetics 111:147–164
Hughes AL, Nei M (1988) Pattern of nucleotide substitution at major histocompatibility complex class I loci reveals overdominant selection. Nature 335:167–170
Hughes AL, Nei M (1989) Nucleotide substitution at major histocompatibility complex class II loci: evidence for overdominant selection. Proc Natl Acad Sci USA 86:958–962
Hughes AL, Yeager M (1998) Natural selection at the major histocompatibility complex loci of vertebrates. Annu Rev Genet 32:415–435
Hughes AL, Hughes MK, Howell CY, Nei M (1994) Natural selection at the class II major histocompatibility complex loci of mammals. Philos Trans R Soc Lond 345:359–367
Hughes C, Miles S, Walbroehl J (2008) Support for the minimal essential MHC hypothesis: a parrot with a single, highly polymorphic MHC class II B gene. Immunogenetics 60:219–231
Hurt P, Walter L, Sudbrak R, Klages S, Muller I, Shiina T, Inoko H, Lehrach H, Günther E, Reinhardt R, Himmelbauer H (2004) The genomic sequence and comparative analysis of the rat major histocompatibility complex. Genome Res 14:631–639
Kaufman J, Milne S, Göbel T, Walker B, Jacob J, Auffray C, Zoorob R, Beck S (1999) The chicken B locus is a minimal essential major histocompatibility complex. Nature 401:923–925
Kekäläinen J, Vallunen J, Primmer C, Rättyä J, Taskinen J (2009) Signals of major histocompatibility complex overdominance in a wild salmonid population. Proc Biol Sci 276:3133–3140
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University Press, Cambridge
Klein J (1986) Natural history of the major histocompatibility complex. Wiley, New York
Kosakovsky Pond S, Frost S (2005) Not so different after all: a comparison of methods for detecting amino acid sites under selection. Mol Biol Evol 22:1208–1222
Kosakovsky Pond S, Frost S, Muse S (2005) HyPhy: hypothesis testing using phylogenies. Bioinformatics 21:676–679
Kosakovsky Pond SL, Posada D, Gravenor MB, Woelk CH, Frost SDW (2006) GARD: a genetic algorithm for recombination detection. Bioinformatics 22:3096–3098
Kruiswijk CP, Hermsen T, van Heerwaarden J, Dixon B, Savelkoul HFJ, Stet RJM (2005) Major histocompatibility genes in the Lake Tana African large barb species flock: evidence for complete partitioning of class II B, but not class I, genes among different species. Immunogenetics 56:894–908
Landry C, Bernatchez L (2001) Comparative analysis of population structure across environments and geographical scales at major histocompatibility complex and microsatellite loci in Atlantic salmon (Salmo salar). Mol Ecol 10:2525–2539
Langefors Å, Lohm J, Von Schantz T, Grahn M (2000) Screening of Mhc variation in Atlantic salmon (Salmo salar): a comparison of restriction fragment length polymorphism (RFLP), denaturing gradient gel electrophoresis (DGGE) and sequencing. Mol Ecol 9:215–219
Langefors Å, Lohm J, Grahn M, Andersen O, Von Schantz T (2001) Association between major histocompatibility complex class IIB alleles and resistance to Aeromonas salmonicida in Atlantic salmon. Philos Trans R Soc Lond 268:479–485
Larhammar D, Schenning L, Gustafsson K, Wiman K, Claesson L, Rask L, Peterson P (1982) Complete amino acid sequence of an HLA-DR antigen-like ss chain as predicted from the nucleotide sequence: similarities with immunoglobulins and HLA-A,-B, and-C antigens. Proc Natl Acad Sci USA 79:3687–3691
Li H, Zhang Y, Zhang Y, Fu Y (2003) Neutrality tests using DNA polymorphism from multiple samples. Genetics 163:1147–1151
Li Y, Costello J, Holloway A, Hahn M, Rausher M (2008) “Reverse ecology” and the power of population genomics. Evolution 62:2984–2994
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451
Lukacs M, Harstad H, Grimholt U, Beetz-Sargent M, Cooper G, Reid L, Bakke H, Phillips R, Miller K, Davidson W, Koop B (2007) Genomic organization of duplicated major histocompatibility complex class I regions in Atlantic salmon (Salmo salar). BMC Genomics 8:251–267
Martinsohn J, Sousa A, Guethlein L, Howard J (1999) The gene conversion hypothesis of MHC evolution: a review. Immunogenetics 50:168–200
McDonald J, Kreitman M (1991) Adaptive protein evolution at the Adh locus in Drosophila. Nature 351:652–654
McVean G, Awadalla P, Fearnhead P (2002) A coalescent-based method for detecting and estimating recombination from gene sequences. Genetics 160:1231–1241
Miller K, Withler R (1996) Sequence analysis of a polymorphic Mhc class II gene in Pacific salmon. Immunogenetics 43:337–351
Miller M, Bacon L, Hala K, Hunt H, Ewald S, Kaufman J, Zoorob R, Briles W (2004) Nomenclature for the chicken major histocompatibility (B and Y) complex. Immunogenetics 56:261–279
Miller K, Li S, Ming T, Kaukinen K, Schulze A (2006) The salmonid MHC class I: more ancient loci uncovered. Immunogenetics 58:571–589
Myers R, Maniatis T, Lerman L (1987) Detection and localization of single base changes by denaturing gradient gel electrophoresis. Meth Enzymol 155:501–527
Nielsen R, Yang Z (1998) Likelihood models for detecting positively selected amino acid sites and applications to the HIV-1 envelope gene. Genetics 148:929–936
Noakes M, Reimer T, Phillips R (2003) Genotypic characterization of an MHC class II locus in Lake Trout (Salvelinus namaycush) from lake superior by single-stranded conformational polymorphism analysis and reference strand–mediated conformational analysis. Mar Biotechnol 5:270–278
Ohta T (1995) Gene conversion vs point mutation in generating variability at the antigen recognition site of major histocompatibility complex loci. J Mol Evol 41:115–119
Parham P, Ohta T (1996) Population biology of antigen presentation by MHC Class I molecules. Science 272:67–74
Penn DJ, Damjanovich K, Potts WK (2002) MHC heterozygosity confers a selective advantage against multiple-strain infections. Proc Natl Acad Sci USA 99:11260–11264
Peters M, Turner T (2008) Genetic variation of the major histocompatibility complex (MHC class II β gene) in the threatened Gila trout. Oncorhynchus gilae gilae. Conserv Genet 9:257–270
Piertney SB, Oliver MK (2006) The evolutionary ecology of the major histocompatibility complex. Heredity 96:7–21
Reche P, Reinherz E (2003) Sequence variability analysis of human class I and class II MHC molecules: functional and structural correlates of amino acid polymorphisms. J Mol Biol 331:623–641
Reusch TBH, Wegner KM, Kalbe M (2001) Rapid genetic divergence in postglacial populations of threespine stickleback (Gasterosteus aculeatus): the role of habitat type, drainage, and geographic proximity. Mol Ecol 10:2435–2446
Salomonsen J, Marston D, Avila D, Bumstead N, Johansson B, Juul-Madsen H, Olesen G, Riegert P, Skjødt K, Vainio O (2003) The properties of the single chicken MHC classical class II a chain (B-LA) gene indicate an ancient origin for the DR/E-like isotype of class II molecules. Immunogenetics 55:605–614
Sambrook J, Figueroa F, Beck S (2005) A genome-wide survey of major histocompatibility complex (MHC) genes and their paralogues in zebrafish. BMC Genomics 6:152
Sheffield V, Cox D, Lerman L, Myers R (1989) Attachment of a 40-base-pair G + C-rich sequence (GC-clamp) to genomic DNA fragments by the polymerase chain reaction results in improved detection of single-base changes. Proc Natl Acad Sci USA 86:232–236
Shum B, Guethlein L, Flodin L, Adkison M, Hedrick R, Nehring R, Stet R, Secombes C, Parham P (2001) Modes of salmonid MHC Class I and II evolution differ from the primate paradigm 1. J Immunol 166:3297–3308
Steinmetz M, Hood L (1983) Genes of the major histocompatibility complex in mouse and man. Science 222:727–733
Stern LJ, Brown JH, Jardetzky TS, Gorga JC, Urban RG, Strominger JL, Wiley DC (1994) Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide. Nature 368:215–221
Stet R, de Vries B, Mudde K, Hermsen T, van Heerwaarden J, Shum B, Grimholt U (2002) Unique haplotypes of co-segregating major histocompatibility class II A and class II B alleles in Atlantic salmon (Salmo salar) give rise to diverse class II genotypes. Immunogenetics 54:320–331
Stet R, Kruiswijk C, Dixon B (2003) Major histocompatibility lineages and immune gene function in teleost fishes: the road not taken. Crit Rev Immunol 23:441–471
Sültmann H, Mayer W, Figueroa F, O’hUigin C, Klein J (1994) Organization of Mhc class II B genes in the zebrafish (Brachydanio rerio). Genomics 23:1–14
Tajima F (1989) Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123:585–595
Takahata N (1990) A simple genealogical structure of strongly balanced allelic lines and trans-species evolution of polymorphism. Proc Natl Acad Sci USA 87:2419–2423
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Trowsdale J, Parham P (2004) Mini-review: defense strategies and immunity-related genes. Eur J Immunol 34:7–17
Van den Berg T, Yoder J, Litman G (2004) On the origins of adaptive immunity: innate immune receptors join the tale. Trends Immunol 25:11–16
Van den Berghe E, Gross M (1989) Natural selection resulting from female breeding competition in a Pacific salmon (coho: Oncorhynchus kisutch). Evolution 43:125–140
Watkins D, McAdam S, Liu X, Strang C, Milford E, Levine C, Garber T, Dogon A, Lord C, Ghim S (1992) New recombinant HLA-B alleles in a tribe of South American Amerindians indicate rapid evolution of MHC class I loci. Nature 357:329–333
Yang Z (2007) PAML 4: phylogenetic analysis by maximum likelihood. Mol Biol Evol 24:1586–1591
Yang Z, Swanson W (2002) Codon-substitution models to detect adaptive evolution that account for heterogeneous selective pressures among site classes. Mol Biol Evol 19:49–57
Yang Z, Wong W, Nielsen R (2005) Bayes empirical bayes inference of amino acid sites under positive selection. Mol Biol Evol 22:1107–1118
Acknowledgments
We would like to thank Constanza Cárdenas for her help in the comparative analysis with human MHC. We would also like to thank El Golfo fisheries and ADL Dignostics S.A. for providing reliable fish samples.
Funding
This work was supported by CORFO-Innova (grant number PBT-165 to S.M.); D.G. holds a pre-doctoral scholarship from CONICYT, Chile.
Author information
Authors and Affiliations
Corresponding author
Additional information
The nucleotide sequences reported in this paper were registered in the NCBI database under GenBank accession numbers FJ597489-FJ597492, FJ597494, FJ597495, FJ597498, FJ597500, FJ597501, FJ597504, FJ597509, FJ597510, FJ597512, FJ597514, FJ597515, FJ597517- FJ597519, FJ597521-FJ597530, FJ597532- FJ597538, FJ943245-FJ943255 and GQ375895-GQ375899.
An erratum to this article can be found at http://dx.doi.org/10.1007/s00251-011-0522-z
Rights and permissions
About this article
Cite this article
Gómez, D., Conejeros, P., Marshall, S.H. et al. MHC evolution in three salmonid species: a comparison between class II alpha and beta genes. Immunogenetics 62, 531–542 (2010). https://doi.org/10.1007/s00251-010-0456-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-010-0456-x