Abstract
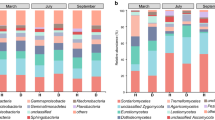
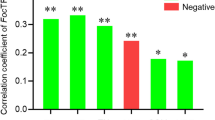
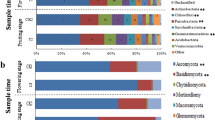
The continuous crop** of banana in the same field may result in a serious soil-borne Fusarium wilt disease and a severe yield decline, a phenomenon known as soil sickness. Although soil microorganisms play key roles in maintaining soil health, the alternations of soil microbial community and relationship between these changes and soil sickness under banana monoculture are still unclear. Bacterial and fungal communities in the soil samples collected from banana fields with different monoculture spans were profiled by sequencing of the 16S rRNA genes and internal transcribed spacer using the MiSeq platform to explore the relationship between banana monoculture and Fusarium wilt disease in the present study. The results showed that successive crop** of banana was significantly correlated with the Fusarium wilt disease incidence. Fungal communities responded more obviously and quickly to banana consecutive monoculture than bacterial community. Moreover, a higher fungal richness significantly correlated to a higher banana Fusarium wilt disease incidence but a lower yield. Banana fungal pathogenic genus of Fusarium and Phyllosticta were closely associated with banana yield depletion and disease aggravation. Potential biocontrol agents, such as Funneliformis, Mortierella, Flavobacterium, and Acidobacteria subgroups, exhibited a significant correlation to lower disease occurrence. Further networks analysis revealed that the number of functionally interrelated modules decreased, the composition shifted from bacteria- to fungi-dominated among these modules, and more resources-competitive interactions within networks were observed after banana long-term monoculture. Our results also showed that bacterial and fungal communities were mainly driven by soil organic matter. Overall, the findings indicated that the bacterial and fungal community structures altered significantly after banana long-term monoculture, and the fungal richness, abundance of Fusarium, interactions between and within bacteria and fungi in ecological networks, and soil organic matter were associated with banana soil-borne Fusarium wilt disease.







Similar content being viewed by others
References
Li X-G, Ding C-F, Zhang T-L, Wang X-X (2014) Fungal pathogen accumulation at the expense of plant-beneficial fungi as a consequence of consecutive peanut monoculturing. Soil Biol. Biochem. 72:11–18
Butler D (2013) Fungus threatens top banana. Nature 504:195–196
Ploetz RC (2006) Fusarium wilt of banana is caused by several pathogens referred to as Fusarium oxysporum f. sp. cubense. Phytopathology 96:653–656
Li C, Li X, Kong W, Wu Y, Wang J (2010) Effect of monoculture soybean on soil microbial community in the Northeast China. Plant Soil 330:423–433
Jaffuel G, Mäder P, Blanco-Perez R, Chiriboga X, Fliessbach A, Turlings TCJ, Campos-Herrera R (2016) Prevalence and activity of entomopathogenic nematodes and their antagonists in soils that are subject to different agricultural practices. Agric. Ecosyst. Environ. 230:329–340
Muellera KE, Eisenhauerb N, Reichd PB, Hobbie SE, Chadwick OA, Chorover J, Dobies T, Hale CM, Jagodziński AM, Kałucka I (2016) Light, earthworms, and soil resources as predictors of diversity of 10 soil invertebrate groups across monocultures of 14 tree species. Soil Biol. Biochem. 92:184–198
Ling N, Huang Q, Guo S, Shen Q (2011) Paenibacillus polymyxa SQR-21 systemically affects root exudates of watermelon to decrease the conidial germination of Fusarium oxysporum f. sp. niveum. Plant Soil 341:485–493
Yuan J, Raza W, Shen Q, Huang Q (2012) Antifungal activity of Bacillus amyloliquefaciens NJN-6 volatile compounds against Fusarium oxysporum f. sp. cubense. Appl. Environ. Microbiol. 78:5942–5944
Weller DM, Raaijmakers JM, Gardener BBM, Thomashow LS (2002) Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu. Rev. Phytopathol. 40:309–348
Langfelder P, Horvath S (2007) Eigengene networks for studying the relationships between co-expression modules. BMC Syst. Biol. 1(1):54
Van der Ent S, Van Hulten M, Pozo MJ, Czechowski T, Udvardi MK, Pieterse CM, Ton J (2009) Priming of plant innate immunity by rhizobacteria and β-aminobutyric acid: differences and similarities in regulation. New Phytol. 183:419–431
Van Elsas JD, Garbeva P, Salles J (2002) Effects of agronomical measures on the microbial diversity of soils as related to the suppression of soil-borne plant pathogens. Biodegradation 13:29–40
Smith FA, Smith SE (1997) Structural diversity in (vesicular)-arbuscular mycorrhizal symbioses. New Phytol. 137:373–388
Vos C, Claerhout S, Mkandawire R, Panis B, De Waele D, Elsen A (2012) Arbuscular mycorrhizal fungi reduce root-knot nematode penetration through altered root exudation of their host. Plant Soil 354:335–345
Yano-Melo AM, Trufem SFB, Maia LC (2003) Arbuscular mycorrhizal fungi in salinized and surrounded areas at the São Francisco Submedium Valley, Brazil. Hoehnea 30:79–87
Anda CCO, Opfergelt S, Declerck S (2016) Silicon acquisition by bananas (c.V. Grande Naine) is increased in presence of the arbuscular mycorrhizal fungus Rhizophagus irregularis MUCL 41833. Plant Soil 409(1):77–85
Van Der Heijden MG, Bardgett RD, Van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 11:296–310
Wu T, Chellemi DO, Graham JH, Rosskopf EN (2008) Assessment of fungal communities in soil and tomato roots subjected to diverse land and crop management systems. Soil Biol. Biochem. 40:1967–1970
Whitman WB, Coleman DC, Wiebe WJ (1998) Prokaryotes: the unseen majority. PNAS 95:6578–6583
Kowalchuk GA, Stephen JR (2001) Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Annu. Rev. Microbiol. 55:485–529
Bever JD (2003) Soil community feedback and the coexistence of competitors: conceptual frameworks and empirical tests. New Phytol. 157:465–473
Rillig MC, Mummey DL (2006) Mycorrhizas and soil structure. New Phytol. 171:41–53
Faust K, Raes J (2012) Microbial interactions: from networks to models. Nat. Rev. Microbiol. 10:538–550
Jeger MJ, Eden-Green S, Thresh JM, Johanson A, Brown WAE (1995) Banana diseases. In: Gowen S (ed) Bananas and plantains. Chapman and Hall, London, pp. 317–381
** spans significantly affect microbial community membership and structure in a vanilla-grown soil as revealed by deep pyrosequencing. Microb. Ecol. 70:209–218
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. PNAS 108:4516–4522
Kozich JJ, Westcott SL, Baxter NT, Highlander SK, Schloss PD (2013) Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79:5112–5120
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73:5261–5267
Huang XQ, Wen T, Zhang JB, Meng L, Zhu TB, Liu LL (2015) Control of soil-borne pathogen Fusarium oxysporum by biological soil disinfestation with incorporation of various organic matters. Eur. J. Plant Pathol. 143:223–235
Zhou J, Deng Y, Luo F, He Z, Yang Y (2011) Phylogenetic molecular ecological network of soil microbial communities in response to elevated CO2. MBio 2:e00122–e00111
Deng Y, Jiang Y-H, Yang Y, He Z, Luo F, Zhou J (2012) Molecular ecological network analyses. BMC Bioinformatics 13:113
Liu X, Zhang J, Gu T, Zhang W, Shen Q, Yin S, Qiu H (2014) Microbial community diversities and taxa abundances in soils along a seven-year gradient of potato monoculture using high throughput pyrosequencing approach. PLoS One 9:e86610
Zhou X, Wu F (2012) Dynamics of the diversity of fungal and Fusarium communities during continuous crop** of cucumber in the greenhouse. FEMS Microbiol. Ecol. 80:469–478
Govaerts B, Mezzalama M, Unno Y, Sayre KD, Luna-Guido M, Vanherck K, Dendooven L, Deckers J (2007) Influence of tillage, residue management, and crop rotation on soil microbial biomass and catabolic diversity. Appl. Soil Ecol. 37:18–30
Luo J, Ran W, Hu J, Yang X, Xu Y, Shen Q (2010) Application of bio-organic fertilizer significantly affected fungal diversity of soils. Soil Sci. Soc. Am. 74:2039–2048
Wong M-H, Crous PW, Henderson J, Groenewald JZ, Drenth A (2012) Phyllosticta species associated with freckle disease of banana. Fungal Divers. 56:173–187
Narisawa K, Tokumasu S, Hashiba T (1998) Suppression of clubroot formation in Chinese cabbage by the root endophytic fungus, Heteroconium chaetospira. Plant Pathol. 47:206–210
Weerakoon DMN, Reardon CL, Paulitz TC, Izzo AD, Mazzola M (2012) Long-term suppression of Pythium abappressorium induced by Brassica juncea seed meal amendment is biologically mediated. Soil Biol. Biochem. 51:44–52
Melo IS, Santos SN, Rosa LH, Parma MM, Silva LJ, Queiroz SC, Pellizari VH (2014) Isolation and biological activities of an endophytic Mortierella alpina strain from the Antarctic moss Schistidium antarctici. Extremophiles 18:15–23
Sanguin H, Sarniguet A, Gazengel K, Moënne-Loccoz Y, Grundmann G (2009) Rhizosphere bacterial communities associated with disease suppressiveness stages of take-all decline in wheat monoculture. New Phytol. 184:694–707
Berendsen RL, Pieterse CM, Bakker PA (2012) The rhizosphere microbiome and plant health. Trends Plant Sci. 17:478–486
Sang M, Kim KD (2012) The volatile-producing Flavobacterium johnsoniae strain GSE09 shows biocontrol activity against Phytophthora capsici in pepper. J. Appl. Microbiol. 113:383–398
Gunasinghe RN, Ikiriwatte CJ, Karunaratne AM (2004) The use of Pantoea agglomerans and Flavobacterium sp. to control banana pathogens. J. Hortic. Sci. Biotechnol. 79:1002–1006
Azcón-Aguilar C, Barea JM (1997) Arbuscular mycorrhizas and biological control of soil-borne plant pathogens-an overview of the mechanisms involved. Mycorrhiza 6:457–464
Borowicz VA (2001) Do arbuscular mycorrhizal fungi alter plant-pathogen relations? Ecology 82:3057–3068
Fath BD, Scharler UM, Ulanowicz RE, Hannon B (2007) Ecological network analysis: network construction. Ecol. Model. 208:49–55
Zhou J, Deng Y, Luo F, He Z, Tu Q, Zhi X (2010) Functional molecular ecological networks. MBio 1:e00169–e00110
Brown KS, Hill CC, Calero GA, Myers CR, Lee KH, Sethna JP, Cerione RA (2004) The statistical mechanics of complex signaling networks: nerve growth factor signaling. Phys. Biol. 3(1):184
Newman ME (2006) Modularity and community structure in networks. PNAS 103:8577–8582
Mau RL, Liu CM, Aziz M, Schwartz E, Dijkstra P, Marks JC, Price LB, Keim P, Hungate BA (2015) Linking soil bacterial biodiversity and soil carbon stability. ISME J. 9:1477–1480
Lauber CL, Strickland MS, Bradford MA, Fierer N (2008) The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biol. Biochem. 40:2407–2415
Cookson WR, Murphy DV, Roper MM (2008) Characterizing the relationships between soil organic matter components and microbial function and composition along a tillage disturbance gradient. Soil Biol. Biochem. 40:763–777
Bausenwein U, Gattinger A, Langer U, Embacher A, Hartmann H-P, Sommer M, Munch J, Schloter M (2008) Exploring soil microbial communities and soil organic matter: variability and interactions in arable soils under minimum tillage practice. Appl. Soil Ecol. 40:67–77
Leon MCC, Stone A, Dick RP (2006) Organic soil amendments: impacts on snap bean common root rot (Aphanomyes euteiches) and soil quality. Appl. Soil Ecol. 31:199–210
Davis J, Huisman O, Everson D, Schneider A (2001) Verticillium wilt of potato: a model of key factors related to disease severity and tuber yield in southeastern Idaho. Am. J. Pota Res. 78:291–300
Garbeva P, Van Veen J, Van Elsas J (2004) Microbial diversity in soil: selection of microbial populations by plant and soil type and implications for disease suppressiveness. Annu. Rev. Phytopathol. 42:243–270
Acknowledgements
We thank the banana orchard owner Mr. Yusheng Li for providing access to the experimental field and facilities. This research was supported by the National Key Basic Research Program of China (2015CB150500), Fundamental Research Funds for the Central Universities (KJQN201746), National Key Research and Development Program (2016YFD0800605 and 2016YFE0101100), National Natural Science Foundation of China (31601836, 31572212 and 31372142), Science and Technology Planning Project of Guangdong Province (2016B020202006), and China Postdoctoral Science Foundation (2016M590469).
Author information
Authors and Affiliations
Corresponding author
Supplementary electronic materials
ESM 1
Eight supplementary tables and five supplementary figures associated with this manuscript are in supplemental material. (DOCX 1058 kb)
Rights and permissions
About this article
Cite this article
Shen, Z., Penton, C.R., Lv, N. et al. Banana Fusarium Wilt Disease Incidence Is Influenced by Shifts of Soil Microbial Communities Under Different Monoculture Spans. Microb Ecol 75, 739–750 (2018). https://doi.org/10.1007/s00248-017-1052-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-017-1052-5