Abstract
Background
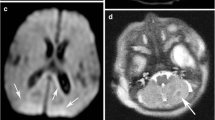
Morbidity attributable to hypoxic-ischaemic injury (HIE) in the perinatal period remains problematic, and timely and accurate assessment of the degree of injury is required for clinical management and prognosis. Conventional MR sequences typically appear normal in the first 48 h post HIE. While diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) maps register the injury earlier, perhaps within the first 24 h, it has been suggested that there may be a propensity at that early stage to underestimate the lesion severity or extent.
Objective
To assess whether MR imaging that included DWI, measured ADC values and T1- and T2-weighted sequences ultimately correlated with either neurodevelopmental outcome or with late MR imaging at 2 years of age. In addition, we wished to compare the performance of MR imaging with cranial US imaging.
Materials and methods
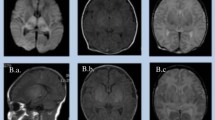
All infants presenting with HIE who had an MRI within 10 days of life were eligible for enrollment and subsequently underwent a full neurodevelopmental assessment at 2 years of age. All children underwent repeat MRI at this time. All neonates had at least one cranial US study. The US findings were categorized as normal, abnormalities confined to the cerebral cortex and subcortical white matter, isolated central grey matter hyperechogenicity, and central hyperechogenicity combined with cerebral cortical/subcortical changes. All MRI studies were retrospectively reviewed by three radiologists. The patterns of injury on the early DWI and ADC maps and early T1- and T2-W studies were recorded as diffuse, central, watershed or atypical. The patterns of signal abnormality were assessed using a scoring system that yielded four separate scores [basal ganglia (BG), watershed (W), BG/W and summation (S)] for the three sets of images, a total of 12 scores in all. The appearance of the posterior limb of the internal capsule (PLIC) on T1-W inversion recovery sequences and of the corpus callosum on all sequences was also documented. After detailed neurodevelopmental assessment at 2 years of age, infants were classified into two groups according to whether they had a favourable or unfavourable outcome.
Results
Of the 26 infants, 6 infants died before formal assessment at the age of 2 years. A further 5 infants had moderate to severe cerebral palsy in addition to severe cognitive impairment. The remaining 15 infants were categorized in the favourable outcome group. The US appearance performed well in terms of predicting final outcome (P = 0.005). The pattern of ischaemia seen on early MRI was a significant predictor of outcome (P < 0.0001). The BG, BG/W and S scores of the diffusion imaging were significantly associated with outcome (P < 0.0001, P < 0.0001 and P = 0.0005 respectively). DWI was predictive of outcome group (P < 0.0001), as were the early T1- and T2-W sequences (P = 0.002) and cranial US (P = 0.005). Assessment of the PLIC in infants with watershed or atypical patterns of ischaemia was found to be less reliable in predicting outcome. The measured ADC value in the PLIC was significantly reduced in those children who had an unfavourable outcome (P = 0.03).
Conclusion
While early MRI performed better than cranial US, the sonography findings were useful. The pattern of ischaemia on early MRI was a good predictor of prognosis. All infants with watershed or atypical patterns had a favourable outcome. The majority of infants with central patterns of ischaemia had an unfavourable outcome and all infants with a diffuse pattern had an unfavourable outcome. DWI was predictive of outcome group, as were early T1- and T2-W sequences and cranial US.




Similar content being viewed by others
References
Barkovich AJ, Miller SP, Bartha A et al (2006) MR imaging, MR spectroscopy, and Diffusion tensor imaging of sequential studies in neonates with encephalopathy. AJNR 27:533–547
McKinstry RC, Miller JH, Snyder AZ et al (2002) A prospective, longitudinal diffusion tensor imaging study of brain injury in newborns. Neurology 59:824–833
Barkovich AJ, Westmark KD, Bedi HS et al (2001) Proton spectroscopy and diffusion imaging on the first day of life after perinatal asphyxia: preliminary report. AJNR 22:1786–1794
Robertson RL, Ben-Sira L, Barnes PD et al (1999) MR line-scan diffusion-weighted imaging of term neonates with perinatal brain ischemia. AJNR 20:1658–1670
Sarnat HB, Sarnat MS (1976) Neonatal encephalopathy following fetal distress. A clinical and electroencephalographic study. Arch Neurol 33:696–705
Levene MI, Kornberg J, Williams TH (1985) The incidence and severity of post-asphyxial encephalopathy in full-term infants. Early Hum Dev 11:21–26
Barkovich AJ, Hajnal BL, Vigneron D et al (1998) Prediction of neuromotor outcome in perinatal asphyxia: evaluation of MR scoring systems. AJNR 19:143–149
Blankenberg FG, Loh NN, Bracci P et al (2000) Sonography, CT, and MR imaging: a prospective comparison of neonates with suspected intracranial ischemia and hemorrhage. AJNR 21:213–218
Barkovich AJ, Westmark K, Partridge C et al (1995) Perinatal asphyxia: MR findings in the first 10 days. AJNR 16:427–438
Daneman A, Epelman M, Blaser S et al (2006) Imaging of the brain in full-term neonates: does sonography still play a role? Pediatr Radiol 36:636–646
Miller SP, Ramaswamy V, Michelson D et al (2005) Patterns of brain injury in term neonatal encephalopathy. J Pediatr 146:453–460
Vermeulen RJ, van Schie PE, Hendrikx L et al (2008) Diffusion-weighted and conventional MR imaging in neonatal hypoxic ischemia: two-year follow-up study. Radiology 249:631–63
Miller SP, Newton N, Ferriero DM et al (2002) Predictors of 30-month outcome after perinatal depression: Role of proton MRS and socioeconomic factors. Pediatr Res 52:71–77
Aida N, Nishimura G, Hachiya Y et al (1998) MR imaging of perinatal brain damage: comparison of clinical outcome with initial and follow-up MR findings. AJNR 19:1909–1921
Biagioni E, Mercuri E, Rutherford M et al (2001) Combined use of electroencephalogram and magnetic resonance imaging in full-term neonates with acute encephalopathy. Pediatrics 107:461–468
Rutherford MA, Pennock JM, Counsell SJ et al (1998) Abnormal magnetic resonance signal in the internal capsule predicts poor neurodevelopmental outcome in infants with hypoxic-ischemic encephalopathy. Pediatrics 102:323–328
Hunt RW, Neil JJ, Coleman LT et al (2004) Apparent diffusion coefficient in the posterior limb of the internal capsule predicts outcome after perinatal asphyxia. Pediatrics 114:999–1003
Boichot C, Walker PM, Durand C et al (2006) Term neonate prognoses after perinatal asphyxia: contributions of MR imaging, MR spectroscopy, relaxation times, and apparent diffusion coefficients. Radiology 239:839–848
Li AM, Chau V, Poskitt KJ et al (2009) White matter injury in term newborns with neonatal encephalopathy. Pediatr Res 65:85–89
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Twomey, E., Twomey, A., Ryan, S. et al. MR imaging of term infants with hypoxic-ischaemic encephalopathy as a predictor of neurodevelopmental outcome and late MRI appearances. Pediatr Radiol 40, 1526–1535 (2010). https://doi.org/10.1007/s00247-010-1692-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-010-1692-9