Abstract
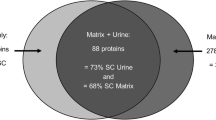
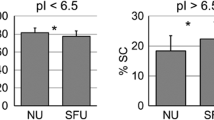
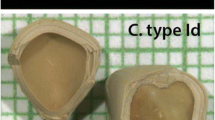
Drug-related kidney stones are a diagnostic problem, since they contain a large matrix (protein) fraction and are frequently incorrectly identified as matrix stones. A urine proteomics study patient produced a guaifenesin stone during her participation, allowing us to both correctly diagnose her disease and identify proteins critical to this drug stone-forming process. The patient provided three random midday urine samples for proteomics studies; one of which contained stone-like sediment with two distinct fractions. These solids were characterized with optical microscopy and Fourier transform infrared spectroscopy. Immunoblotting and quantitative mass spectrometry were used to quantitatively identify the proteins in urine and stone matrix. Infrared spectroscopy showed that the sediment was 60 % protein and 40 % guaifenesin and its metabolite guaiacol. Of the 156 distinct proteins identified in the proteomic studies, 49 were identified in the two stone-components with approximately 50 % of those proteins also found in this patient’s urine. Many proteins observed in this drug-related stone have also been reported in proteomic matrix studies of uric acid and calcium containing stones. More importantly, nine proteins were highly enriched and highly abundant in the stone matrix and 8 were reciprocally depleted in urine, suggesting a critical role for these proteins in guaifenesin stone formation. Accurate stone analysis is critical to proper diagnosis and treatment of kidney stones. Many matrix proteins were common to all stone types, but likely not related to disease mechanism. This protocol defined a small set of proteins that were likely critical to guaifenesin stone formation based on their high enrichment and high abundance in stone matrix, and it should be applied to all stone types.





Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- FTIR:
-
Fourier transform infrared spectroscopy
- GC:
-
Guaifenesin.guaiacol crystal mixture
- GF:
-
Orange stone sediment containing guaifenesin and guaiacol crystals)
- IRB:
-
Institutional Review Board
- LC–MS:
-
Liquid chromatography coupled with mass spectrometry
- MCW:
-
Medical College of WISCONSIN
- MIS.MAC:
-
Mandel International Stone and Molecular Analysis Center
- MS:
-
Mass spectrometry
- MX:
-
Matrix strand stone sediment containing few drug crystals
- P/C:
-
Protein/creatinine ratio in urine (mg/g)
- SG:
-
Specific gravity
- UA:
-
Uric acid stone matrix
- UM:
-
Urine macromolecules
- VA:
-
US Department of Veterans Affairs
- % SC:
-
Percentage of total assigned scan counts attributable to an individual or group of proteins
All protein abbreviations are defined in Table 3
References
Boyce Garvey FK (1956) The amount and nature of the organic matrix in urinary calculi: a review. J Urol 76:213–227
Aggarwal KP, Narula S, Kakkar M, Tandon C (2013) Nephrolithiasis: molecular mechanism of renal stone formation and the critical role played by modulators. Biomed res int. doi:10.1155/2013/292953
Mandel NS, Mandel GS (1989) Urinary tract stone disease in the united states veteran population. I. Geographical frequency of occurrence. J Urol 142:1513–1515
Krambeck AE, Lingeman JE, McAteer JA, Williams JC Jr (2010) Analysis of mixed stones is prone to error: a study with US laboratories using micro CT for verification of sample content. Urol Res 38:469–475. doi:10.1007/s00240-010-0317-y
Gage H, Beal HW (1908) V. fibrinous calculi in the kidney. Ann Surg 48:378–387
Bani-Hani AH, Segura JW, Leroy AJ (2005) Urinary matrix calculi: our experience at a single institution. J Urol 173:120–123
Dussol B, Geider S, Lilova A, Leonetti F, Dupuy P, Daudon M, Berland Y, Dagorn JC, Verdier JM (1995) Analysis of the soluble organic matrix of five morphologically different kidney stones. evidence for a specific role of albumin in the constitution of the stone protein matrix. Urol Res 23:45–51
Khan SR, Atmani F, Glenton P, Hou Z, Talham DR, Khurshid M (1996) Lipids and membranes in the organic matrix of urinary calcific crystals and stones. Calcif Tissue Int 59:357–365
Canales BK, Anderson L, Higgins L, Ensrud-Bowlin K, Roberts KP, Wu B, Kim IW, Monga M (2010) Proteome of human calcium kidney stones. Urology 76:1017.e13–1017.e20. doi:10.1016/j.urology.2010.05.005
Jou YC, Fang CY, Chen SY, Chen FH, Cheng MC, Shen CH, Liao LW, Tsai YS (2012) Proteomic study of renal uric acid stone. Urology 80:260–266. doi:10.1016/j.urology.2012.02.019
Kaneko K, Kobayashi R, Yasuda M, Izumi Y, Yamanobe T, Shimizu T (2012) Comparison of matrix proteins in different types of urinary stone by proteomic analysis using liquid chromatography-tandem mass spectrometry. Int J Urol 19:765–772. doi:10.1111/j.1442-2042.2012.03005.x
Assimos DG, Langenstroer P, Leinbach RF, Mandel NS, Stern JM, Holmes RP (1999) Guaifenesin and ephedrine induced stones. J Endourol 13:665–667
Bennett S, Hoffman N, Monga M (2004) Ephedrine- and guaifenesin-induced nephrolithiasis. J Altern Complement Med 10:967–969
Pickens CL, Milliron AR, Fussner AL, Dversdall BC, Langenstroer P, Ferguson S, Fu X, Schmitz FJ, Poole EC (1999) Abuse of guaifenesin-containing medications generates an excess of a carboxylate salt of beta-(2-methoxyphenoxy)-lactic acid, a guaifenesin metabolite, and results in urolithiasis. Urology 54:23–27
Kolbach AM, Afzal O, Halligan B, Sorokina E, Kleinman JG, Wesson JA (2012) Relative deficiency of acidic isoforms of osteopontin from stone former urine. Urol Res 40:447–454. doi:10.1007/s00240-012-0459-1
Mani N, Jun HW, Beach JW, Nerurkar J (2003) Solubility of guaifenesin in the presence of common pharmaceutical additives. Pharm Dev Technol 8:385–396
Mandel IC, Mandel NS (2007) Chapter 5. Structural and compositional analysis of kidney stones. In: Stoller ML, Meng MV (eds) Urinary stone disease: the practical guide to medical and surgical management. Humana Press, USA, pp 69–81
Viswanathan P, Rimer JD, Kolbach AM, Ward MD, Kleinman JG, Wesson JA (2011) Calcium oxalate monohydrate aggregation induced by aggregation of desialylated tamm–horsfall protein. Urol Res 39:269–282. doi:10.1007/s00240-010-0353-7
Yu H, Wakim B, Li M, Halligan B, Tint GS, Patel SB (2007) Quantifying raft proteins in neonatal mouse brain by ‘tube-gel’ protein digestion label-free shotgun proteomics. Proteome Sci 5:17
Lemann J Jr, Pleuss JA, Worcester EM, Hornick L, Schrab D, Hoffmann RG (1996) Urinary oxalate excretion increases with body size and decreases with increasing dietary calcium intake among healthy adults. Kidney Int 49:200–208
Li M, Gray W, Zhang H, Chung CH, Billheimer D, Yarbrough WG, Liebler DC, Shyr Y, Slebos RJ (2010) Comparative shotgun proteomics using spectral count data and quasi-likelihood modeling. J Proteome Res 9:4295–4305. doi:10.1021/pr100527g
Canales BK, Anderson L, Higgins L, Frethem C, Ressler A, Kim IW, Monga M (2009) Proteomic analysis of a matrix stone: a case report. Urol Res 37:323–329. doi:10.1007/s00240-009-0213-5
Okumura N, Tsujihata M, Momhara C, Yoshioka I, Suto K, Nonomura N, Okuyama A, Toshifumi T (2013) Diversity in protein profiles of individual calcium oxalate kidney stones. PLoS One 8(7):e68624. doi:10.1371/journal.pone.0068624
Brahm NC, Yeager LL, Fox MD, Farmer KC, Palmer TA (2010) Commonly prescribed medications and potential false-positive urine drug screens. Am J Health Syst Pharm 67:1344–1350. doi:10.2146/ajhp090477
Ramsdell CM, Postlethwaite AE, Kelley WN (1974) Uricosuric effect of glyceryl guaiacolate. J Rheumatol 1:114–116
Bellei E, Cuoghi A, Monari E, Bergamini S, Fantoni LI, Zappaterra M, Guerzoni S, Bazzocchi A, Tomasi A, Pini LA (2012) Proteomic analysis of urine in medication-overuse headache patients: possible relation with renal damages. J Headache Pain 13:45–52. doi:10.1007/s10194-011-0390-9
Riaz S, Alam SS, Srai SK, Skinner V, Riaz A, Akhtar MW (2010) Proteomic identification of human urinary biomarkers in diabetes mellitus type 2. Diabetes Technol Ther 12:979–988. doi:10.1089/dia.2010.0078
Merchant ML, Cummins TD, Wilkey DW, Salyer SA, Powell DW, Klein JB, Lederer ED (2008) Proteomic analysis of renal calculi indicates an important role for inflammatory processes in calcium stone formation. Am J Physiol Renal Physiol 295:F1254–F1258. doi:10.1152/ajprenal.00134.2008
Matlaga BR, Shah OD, Assimos DG (2003) Drug-induced urinary calculi. Rev urol 5:227–231
Riese RJ, Kleinman JG, Wiessner JH, Mandel GS, Mandel NS (1990) Uric acid crystal binding to renal inner medullary collecting duct cells in primary culture. J Am Soc Nephrol 1:187–192
Riese RJ, Mandel NS, Wiessner JH, Mandel GS, Becker CG, Kleinman JG (1992) Cell polarity and calcium oxalate crystal adherence to cultured collecting duct cells. Am J Physiol (Renal Fluid Electrolyte Physiol) 262(31):F117–F184
Cockerill PA, de Cogain MR, Krambeck AE (2013) Acute bilateral ureteral obstruction secondary to guaifenesin toxicity. Can J Urol 20:6971–6973
Seagrave J, Albrecht H, Park YS, Rubin B, Solomon G, Kim KC (2011) Effect of guaifenesin on mucin production, rheology, and mucociliary transport in differentiated human airway epithelial cells. Exp Lung Res 37:606–614. doi:10.3109/01902148.2011.623116
Acknowledgments
We gratefully acknowledge the primary financial support provided in part with resources and the use of facilities at the Clement J. Zablocki Department of Veterans Affairs Medical Center, Milwaukee, WI, and in part by the National Institutes of Health/National Institute for Diabetes, Digestive, and Kidney Diseases (DK 82550) (JAW). Additional financial support was provided by the Medical College of Wisconsin and in part by the National Institutes of Health/National Institute for Diabetes, Digestive, and Kidney Diseases (DK 74741) (JGK). We also gratefully acknowledge the technical support MIS.MAC (Mandel International Stone and Molecular Analysis Center), Milwaukee, WI, for stone analysis and the technical support of Brian Halligan, PhD, for proteomic analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was primarily funded with resources and the use of facilities at the Clement J. Zablocki Department of Veterans Affairs Medical Center, Milwaukee, WI, and in part by a grant from the National Institutes of Health (NIDDK, DK 82550-JAW). Additional financial support was provided by the Medical College of Wisconsin and in part by the National Institutes of Health (NIDDK, DK 74741-JGK).
Conflict of interest
None of the authors has any conflicts of interest to report.
Human studies
The participating patient in this study was recruited with informed consent to an established study under VA IRB approval (VA-IRB protocol: 9305-01P). All procedures performed in these studies were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments.
Additional information
The contents do not represent the views of the US Department of Veterans Affairs or the United States Government.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kolbach-Mandel, A.M., Mandel, N.S., Cohen, S.R. et al. Guaifenesin stone matrix proteomics: a protocol for identifying proteins critical to stone formation. Urolithiasis 45, 139–149 (2017). https://doi.org/10.1007/s00240-016-0907-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-016-0907-4