Abstract.
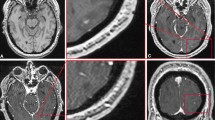
Seventy-four patients with one to eight proven intraaxial brain metastases received a total cumulative dose of 0.2 mmol/kg bodyweight gadobenate dimeglumine, administered as sequential injections of 0.05, 0.05 and 0.1 mmol/kg over a 20-min period. MR imaging was performed before the first administration (T2- and T1-weighted sequences) and after each injection of contrast agent (T1-weighted sequences only). Quantitative assessment of images revealed significant (P<0.01) dose-related increases in lesion-to-brain (L/B) ratio and percent enhancement of lesion signal intensity. Qualitative assessment by two independent, blinded assessors revealed additional lesions in 22%, 25% and 38% (assessor 1) and 29%, 32% and 34% (assessor 2) of patients after each cumulative dose when compared with combined T1- and T2-weighted pre-contrast images. Significantly more lesions (P≤0.01) were noted by both assessors after the first injection and by one assessor after each subsequent injection. For patients with just one lesion observed on unenhanced T1- and T2-weighted images, additional lesions were noted in 12%, 16% and 28% of patients by assessor 1 following each dose and in 24%, 27% and 30% of patients by assessor 2. Contemporaneously, diagnostic confidence was increased and lesion conspicuity improved over unenhanced MRI. For patients with one lesion observed after an initial dose of 0.05 mmol/kg, additional lesions were noted by assessors 1 and 2 in 9.1% and 11.8% of patients, respectively, after a cumulative dose of 0.1 mmol/kg and in a further 9.1% and 5.9% of patients, respectively, after a cumulative dose of 0.2 mmol/kg. No safety concerns were apparent.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Balériaux, D., Colosimo, C., Ruscalleda, J. et al. Magnetic resonance imaging of metastatic disease to the brain with gadobenate dimeglumine. Neuroradiology 44, 191–203 (2002). https://doi.org/10.1007/s002340100636
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s002340100636