Abstract
Introduction
Magnetic resonance (MR) diffusion-weighted imaging (DWI), dynamic susceptibility contrast-enhanced perfusion imaging (DSC), and MR spectroscopy (MRS) techniques provide specific physiologic information that may distinguish malignant glioma progression from post-radiation change, yet no single technique is completely reliable. We propose a simple, multiparametric scoring system to improve diagnostic accuracy beyond that of each technique alone.
Methods
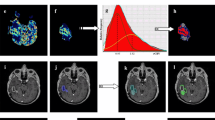
Fifteen subjects with lesions suspicious for glioma progression following radiation therapy who had also undergone 3-tesla DWI, DSC, and MRS studies of the lesion were retrospectively reviewed. Minimum apparent diffusion coefficient (ADC) ratio, maximum regional cerebral blood volume (rCBV) ratio, and maximum MRS choline/creatine (Cho/Cr) and choline/N-acetyl-aspartate (Cho/NAA) metabolic peak-height ratios were quantified within each lesion. Each parameter (ADC ratio, rCBV ratio, and combined Cho/Cr and Cho/NAA ratios) was scored as either glioma progression (one point) or radiation change (zero point) based upon thresholds derived from our own data. For each lesion, the combined parameters yielded a multiparametric score (0 to 3) for prediction of tumor progression or post-radiation change.
Results
Optimum thresholds for ADC ratio (1.30), rCBV ratio (2.10), and either combined Cho/Cr (1.29) and Cho/NAA (1.06) yielded diagnostic accuracies of 86.7%, 86.7%, and 84.6%, respectively (p < 0.05). A combined multiparametric score threshold of 2 improved diagnostic accuracy to 93.3% (p < 0.05).
Conclusion
In this small series combining 3-T DWI, DSC, and MRS diagnostic results using a simple, multiparametric scoring system has potential to improve overall diagnostic accuracy in distinguishing glioma progression from post-radiation change beyond that of each technique alone.



Similar content being viewed by others
References
Mullins ME, Barest GD, Schaefer PW, Hochberg FH, Gonzalez RG, Lev MH (2005) Radiation necrosis versus glioma recurrence: conventional MR imaging clues to diagnosis. AJNR Am J Neuroradiol 26:1967–1972
Kumar AJ, Leeds NE, Fuller GN, Van Tassel P, Maor MH, Sawaya RE, Levin VA (2000) Malignant gliomas: MR imaging spectrum of radiation therapy- and chemotherapy induced necrosis of the brain after treatment. Radiology 217:377–384
Zeng QS, Li CF, Liu H, Zhen JH, Feng DC (2007) Distinction between recurrent glioma and radiation injury using magnetic resonance spectroscopy in combination with diffusion-weighted imaging. Int J Radiat Oncol Biol Phys 78:151–158
Lam WW, Poon WS, Metreweli C (2002) Diffusion MR imaging in glioma: does it have any role in the pre-operation determination of grading of gliomas? Clin Radiol 57:219–225
Hein PA, Eskey CJ, Dunn JF, Hug EB (2004) Diffusion-weighted imaging in the follow-up of treated high-grade gliomas: tumor recurrence versus radiation therapy. AJNR Am J Neuroradiol 25:201–209
Asao C, Korogi Y, Kitajima M et al (2005) Diffusion-weighted imaging of radiation-induced brain injury for differentiation from tumor recurrence. AJNR Am J Neuroradiol 26:1455–1460
Sugahara T, Korogi Y, Tomiguchi S, Shigematsu Y, Ikushima I, Kira T, Liang L, Ushio Y, Takahashi M (2000) Post therapeutic intra axial brain tumor: the value of perfusion-sensitive contrast-enhanced MR imaging for differentiating tumor recurrence from nonneoplastic contrast-enhancing tissue. AJNR Am J Neuroradiol 21:901–909
Cha S, Knopp EA, Johnson G, Litt A, Glass J, Gruber ML, Lu S, Zagzag D (2000) Dynamic contrast-enhanced T2*-weighted MR imaging of recurrent malignant gliomas treated with thalidomide and carboplatin. AJNR Am J Neuroradiol 21:881–890
Sugahara T, Korogi Y, Kochi M, Ushio Y, Takahashi M (2001) Perfusion sensitive MR imaging of gliomas: comparison between gradient-echo and spin-echo echo-planar imaging techniques. AJNR Am J Neuroradiol 22:1306–1315
Liu G, Sobering G, Duyn J, Moonen CT (1993) A functional MRI technique combining principles of echo-shifting with a train of observations (PRESTO). Magn Reson Med 30:764–768
Manka C, Träber F, Gieseke J, Schild HH, Kuhl CK (2005) Three-dimensional dynamic susceptibility-weighted perfusion MR imaging at 3.0T: feasibility and contrast agent dose. Radiology 234:869–877
van Gelderen P, Grandin C, Petrella JR, Moonen CT (2000) Rapid three-dimensional MR imaging method for tracking a bolus of contrast agent through the brain. Radiology 216:603–608
Plotkin M, Eisenacher J, Bruhn H, Wurm R, Michel R, Stockhammer F, Feussner A, Dudeck O, Wust P, Felix R, Amthauer H (2004) 123I-IMT SPECT and 1HMR-spectroscopy at 3.0T in the differential diagnosis of recurrent or residual gliomas: a comparative study. J Neurooncol 70:49–58
Ando K, Ishikura R, Nagami Y, Morikawa T, Takada Y, Ikeda J, Nakao N, Matsumoto T, Arita N (2004) Usefulness of Cho/Cr ratio in protonMR spectroscopy for differentiating residual/recurrent glioma from non-neoplastic lesions. Nippon Igaku Hoshasen Gakkai Zasshi 64:121–126
Zeng QS, Li CF, Kai Z, Liu H, Kang XS, Zhen JH (2007) Multivoxel 3D proton MR spectroscopy in the distinction of recurrent glioma from radiation injury. J Neurooncol 84:63–69
Rock JP, Hearshen D, Scarpace L, Croteau D, Gutierrez J, Fisher JL, Rosenblum ML, Mikkelsen T (2002) Correlations between magnetic resonance spectoroscopy and image-guided histopathology, with special attention to radiation necrosis. Neurosurgery 51:912–920
Weybright P, Sundgren PC, Maly P, Hassan DG, Nan B, Rohrer S, Junck L (2005) Differentiation between brain tumor recurrence and radiation injury using MR spectroscopy. AJR Am J Roentgenol 185:1471–1476
Lichy MP, Henze M, Plathow C, Bachert P, Kauczor HU, Schlemmer HP (2004) Metabolic imaging to follow stereotactic radiation of gliomas the role of 1H MR spectroscopy in comparison to FDG PET and IMT-SPECT. Rofo 176:1114–1121
Schlemmer HP, Bachert P, Herfarth KK, Zuna I, Debus J, van Kaick G (2001) Proton MR spectroscopic evaluation of suspicious brain lesions after stereotactic radiotherapy. AJNR Am J Neuroradiol 22:1316–1324
Di Chiro G, Oldfield E, Wright DC (1988) Cerebral necrosis after radiotherapy and/or intraarterial chemotherapy for brain tumors: PET and neuropathologic studies. AJR Am J Roentgenol 150:189–197
Langleben DD, Segall GM (2000) PET in differentiation of recurrent brain tumor from radiation injury. J Nucl Med 41:1861–1867
Wang SX, Boethius J, Ericson K (2006) FDG-PET on irradiated brain tumor: ten years’ summary. Acta Radiol 47:85–90
Kim JH, Chang KH, Na DG, Song IC, Kwon BJ, Han MH, Kim K (2006) 3T H-MR spectroscopy in grading of cerebral gliomas: comparison of short and intermediate echo time sequences. AJNR Am J Neuroradiology 27:1412–1418
Conflict of interest statement
K. Maravilla is a research support speaker and consultant for Bracco and offers research support and is a consultant to Bayer healthcare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsusue, E., Fink, J.R., Rockhill, J.K. et al. Distinction between glioma progression and post-radiation change by combined physiologic MR imaging. Neuroradiology 52, 297–306 (2010). https://doi.org/10.1007/s00234-009-0613-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-009-0613-9