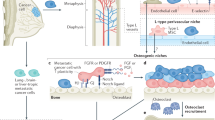
Abstract
Metastasis is defined as a very inefficient process, since less than 0.01% of cancer cells injected into the circulation will engraft in a distant organ, where they must acquire the ability to survive and proliferate inside a “foreign” environment. In bone metastases, the interaction with the host organ is much more favoured if tumour cells gain “osteomimicry”, that is the ability to resemble a resident bone cell (i.e. the osteoblast), thus intruding in the physiology of the bone. This is accomplished by the expression of osteoblast markers (e.g. alkaline phosphatase) and the production of bone matrix proteins and paracrine factors which deregulate the physiology of bone, fuelling the so-called “vicious cycle”. The main challenge of researchers is therefore to identify the genetic profile determining the osteotropism of tumour cells, which would eventually lead to bone colonisation. This could likely provide the answer to a quite intriguing question, that is why some cancers, such as prostate and breast, have a specific predilection to metastasise to the bone. Therefore, it is important to completely address the molecular mechanisms underlying this aspect of bone oncology, identifying relevant pathways, the targeting of which could make any type of bone metastasis curable or avoidable.




Similar content being viewed by others
References
Fidler IJ (1970) Metastasis: quantitative analysis of the distribution and fate of tumor emboli labeled with 125I-5-iodo-2′-deoxyuridine. J Natl Cancer Inst 45:773–782
Paget S (1889) The distribution of secondary growths in cancer of the breast. Lancet 1:571–573
Ewing J (1928) A treatise on tumours, 3rd edn. W.B. Saunders, Philadelphia
Bubendorf L, Schopfer A, Wagner U (2000) Metastatic patterns of prostate cancer: an autopsy study of 1589 patients. Hum Pathol 31:578–583
Coleman RE (2016) Impact of bone-targeted treatments on skeletal morbidity and survival in breast cancer. Oncology 30(8):695 (Williston Park)
Roodman GD (2004) Mechanisms of bone metastasis. N Engl J Med 350:1655–1664
Lacey D, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T, Elliott R, Colombero A, Elliott G, Scully S, Hsu H, Sullivan J, Hawkins N, Davy E, Capparelli C, Eli A, Qiann YX, Kaufman S, Sarosi I, Shalshoub V, Senaldi G, Guo J, Delaney J, Boyle WJ (1998) Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93:165–176
Simonet WS, Lacey DL, Dunstan CR, Kelley M, Chang MS, Luthy R, Nguyen HQ, Wooden S, Bennett L, Boone T, Shimamoto G, DeRose M, Elliott R, Colombero A, Tan HL, Trail G, Sullivan J, Davy E, Bucay N, Renshaw-Gegg L, Hughes TM, Hill D, Pattison W, Campbell P, Sander S, Van G, Tarpley J, Derby P, Lee R, Program Amgen EST, Boyle WJ (1997) Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89:309–319
**ong J, Piemontese M, Onal M, Campbell J, Goelner JJ, Dusevich V, Bonewald L, Manolagas SC, O’Brian CA (2015) Osteocytes not osteoblasts or lining cells are the main source of the RANKL required for osteoclast formation in remodelling bone. PLoS ONE 10:e0138189
Fernandez-Valdivia R, Mukherjee A, Ying Y, Li J, Paquet M, DeMayo FJ, Lydon JP (2009) The RANKL signaling axis is sufficient to elicit ductal side-branching and alveologenesis in the mammary gland of the virgin mouse. Dev Biol 328:127–139
Kong YY, Boyle WJ, Penninger JM (1999) Osteoprotegerin ligand: a common link between osteoclastogenesis, lymph node formation and lymphocyte development. Immunol Cell Biol 77:188–193
Anderson DM, Maraskovsky E, Billigsley WL, Dougall WC, Tometsko ME, Roux ER, Teepe MC, DuBose RF, Cosman D, Galibert L (1997) A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature 390:175–179
Kim W, Takyar FM, Swan K, Jeong J, vanHouten J, Sullivan CA, Dann P, Yu H, Fiaschi-Taesch N, Chang W, Wysolmerski J (2016) Calcium-sensing receptor (CaSR) promotes breast cancer by stimulating intracrine actions of parathyroid hormone-related protein. Cancer Res 76:5348–5360
Byun MR, Hwang JH, Kim AR, Kim KM, Hwang ES, Yaffe MB, Hong JH (2014) Canonical Wnt signalling activates TAZ through PP1A during osteogenic differentiation. Cell Death Differ 21:854–863
Ducy P, Zhang R, Geoffroy V, Ridall AI, Karsenty G (1997) Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell 89:747–754
Kim WK, Meliton V, Bourquard N, Hahn TJ, Parhami F (2010) Hedgehog signaling and osteogenic differentiation in multipotent bone marrow stromal cells are inhibited by oxidative stress. J Cell Biochem 111:1199–1209
Curatolo C, Ludovico GM, Correale M, Pagliarulo A, Abbate I, Cirrillo Marucco E, Barletta A (1992) Advanced prostate cancer follow-up with prostate-specific antigen, prostatic acid phosphatase, osteocalcin and bone isoenzyme of alkaline phosphatase. Eur Urol 21(Suppl 1):105–107
Koeneman KS, Yeung F, Chung LW (1999) Osteomimetic properties of prostate cancer cells: a hypothesis supporting the predilection of prostate cancer metastasis and growth in the bone environment. Prostate 39:246–261
Knerr K, Ackermann K, Neidhart T, Pyerin W (2004) Bone metastasis: osteoblasts affect growth and adhesion regulons in prostate tumor cells and provoke osteomimicry. Int J Cancer 111:152–159
Bellahcène A, Bachelier R, Detry C, Lidereau R, Clézardin P, Castronovo V (2007) Transcriptome analysis reveals an osteoblast-like phenotype for human osteotropic breast cancer cells. Breast Cancer Res Treat 101:135–148
Yuen HF, Kwok WK, Chan KK, Chua CW, Chan YP, Chu YY, Wong YC, Wang X, Chan KW (2008) TWIST modulates prostate cancer cell-mediated bone cell activity and is upregulated by osteogenic induction. Carcinogenesis 29:1509–1518
Tan CC, Li GX, Tan LD, Du X, Li XQ, He R, Wang QS, Feng YM (2016) Breast cancer cells obtain an osteomimetic feature via epithelial/mesenchymal transition that have undergone BMP2/RUNX2 signaling pathway induction. Oncotarget 7:79688–79705
Kang Y, Siegel PM, Shu W, Drobnjak M, Kakonen SM, Cordon-Cardo C, Guise T, Massagué J (2003) A multigenic program mediating breast cancer metastasis to bone. Cancer Cell 3:537–549
Kim JB, Leucht P, Lam K, Luppen C, Ten Berge D, Nusse R, Helms JA (2007) Bone regeneration is regulated by wnt signalling. J Bone Miner Res 22:1913–1923
Moverare-Skrtic S, Henning P, Liu X, Nagano K, Saito H, Borjesson AE, Sjogren K, Windhal SH, Farman H, Kindlund B, Engdahl C, Koskela A, Zhang FP, Eriksson EE, Zaman F, Hammarstedt A, Isaksson H, Bally M, Kassem A, Lindholm C, Sandberg O, Aspenberg P, Savendahl L, Feng JQ, Tuckermann J, Tuukkanen J, Poutanen M, Baron R, Lerner UH, Gori F, Ohlsson C (2014) Osteoblast-derived WNT16 represses osteoclastogenesis and prevents cortical bone fragility fractures. Nat Med 20:279–1288
Hall CL, Bafico A, Dai J, Aaronson SA, Kellet ET (2005) Prostate cancer cells promote osteoblastic bone metastases through Wnt. Cancer Res 65:7554–7560
Chen G, Shukeir N, Potti A, Sircar K, Aprikian A, Goltzman D, Rabbani SA (2004) Up-regulation of Wnt-1 and -catenin production in patients with advanced metastatic prostate carcinoma: potential pathogenetic and prognostic implications. Cancer 101:1345–1356
Logothetis CJ, Lin SH (2005) Osteoblasts in prostate cancer metastasis to bone. Nat Rev Cancer 5:21–28
Chu CYG, Zhau HE, Wang R, Rogatko A, Feng X, Zayzafoon M, Liu Y, Farach-Carson MC, You S, Kim J, Freeman MR, Chung WK (2014) RANK-and c-Met-mediated signal network promotes prostate cancer metastatic colonization. Endocr Relat Cancer 21:311–326
Holen H, Croucher PI, Hamdy FC, Eaton CL (2002) Osteoprotegerin (OPG) is a survival factor for human prostate cancer cells. Cancer Res 62:1619–1623
Neville-Webbe HL, Cross NA, Eaton CL, Nyambo R, Evans CA, Coleman RE, Holen I (2004) Osteoprotegerin (OPG) produced by bone marrow stromal cells protects breast cancer cells from TRAIL-induced apoptosis. Breast Cancer Res Treat 86:269–279
Higgs JT, Jarboe JS, Lee JH, Chanda D, Lee CM, Deivanayagam C, Ponnazhagan S (2015) Variants of osteoprotegerin lacking TRAIL binding for therapeutic bone remodeling in osteolytic malignancies. Mol Cancer Res 13:819–882
Fisher LW, Fedarko NS (2003) Six genes expressed in bone and teeth encode the current members of the SIBLING family proteins. Connect Tissue Res 44(Suppl1):33–40
Thalmann GN, Sikes RA, Devoll RE, Kiefer JA, Markwalder R, Klima I, Farach-Carson CM, Studer UE, Chung LW (1999) Osteopontin: possible role in prostate cancer progression. Clin Cancer Res 5:2271–2277
Carlinfante G, Vassilioul D, Svensson O, Wendel M, Heinegard D, Andersson G (2003) Differential expression of osteopontin and bone sialoprotein in bone metastasis of breast and prostate carcinoma. Clin Exp Metastasis 20:437–444
Shevde LA, Das S, Clark DW, Samant RS (2010) Osteopontin: an effector and an effect of tumor metastasis. Curr Mol Med 10:71–81
Das S, Tucker JA, Khullar S, Samant RS, Shevde LA (2012) Hedgehog signalling in tumor cells facilitates osteoblast-enhanced osteolytic metastases. PLoS ONE 7:e34374
Bellahcène A, Merville MP, Castronovo V (1994) Expression of bone sialoprotein, a bone matrix protein, in human breast cancer. Cancer Res 54:2823–2826
Waltregny D, Bellahcène A, Van Riet I, Fisher LW, Young M, Fernandez P, Dewé W, de Leval J, Castronovo V (1998) Prognostic value of bone sialoprotein expression in clinically localized human prostate cancer. J Natl Cancer Inst 90:1000–1008
Zhang JH (2004) Bone sialoprotein promotes bone metastasis of a non-bone-seeking clone of human breast cancer cells. Anticancer Res 24:1361–1368
Reufsteck C, Lifshitz-Shovali R, Zepp M, Bäuerle T, Kübler D, Golomb G, Berger MR (2012) Silencing of skeletal metastasis-associated genes impairs migration of breast cancer cells and reduces osteolytic bone lesions. Clin Exp Metastasis 29:441–456
Yeung F, Law WK, Yeh CH, Westendorf JJ, Zhang Y, Wang R, Kao C, Chung LW (2002) Regulation of human osteocalcin promoter in hormone-independent human prostate cancer cells. J Biol Chem 277:2468–2476
Huang WC, **e Z, Konaka H, Sodek J, Zhau HE, Chung LWK (2005) Human osteocalcin and bone sialoprotein mediating osteomimicry of prostate cancer cells: role of cAMP-dependent protein kinase A signalling pathway. Cancer Res 65:2303–2313
Jacob K, Webber M, Benayahu D, Kleinman HK (1999) Osteonectin promotes prostate cancer cell migration and invasion: a possible mechanism for metastasis to bone. Cancer Res 59:4453–4457
Josson S, Nomura T, Lin JT, Huang WC, Wu D, Zhau HE, Zayzafoon M, Weizmann MN, Gururajan M, Chung WKL (2011) β2-Microglobulin induces epithelial to mesenchymal transition and confers cancer lethality and bone metastasis in human cancer cells. Cancer Res 71:2600–2610
Hassan MQ, Maeda Y, Taipaleenmaki H, Zhang W, Jafferji M, Gordon JA, Li Z, Croce CM, van Wijnen AJ, Stein JL, Stein GS, Lian JB (2012) miR-218 directs a Wnt signaling circuit to promote differentiation of osteoblasts and osteomimicry of metastatic cancer cells. J Biol Chem 287:42084–42092
Raposo G, Stoorvogel W (2013) Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol 200:373–383
Kaiser J (2016) Malignant messengers. Science 352:164–166
Itoh T, Ito Y, Ohtsuki Y, Ando M, Tsukamasa Y, Yamada N, Naoe T, Akao Y (2012) Microvesicles released from hormone-refractory prostate cancer cells facilitate mouse pre-osteoblast differentiation. J Mol Histol 43:509–515
Karlsson T, Lundholm M, Widmark A, Persson E (2016) Tumor-derived exosomes from the prostate cancer cell line TRAMP-C1 impair osteoclast formation and differentiation. PLoS ONE 11:e0166284
Andersen TL, Boissy P, Sondergaard TE, Kupisiewicz K, Plesner T, Rasmussen T, Haaber J, Kølvraa S, Delaissé JM (2007) Osteoclast nuclei of myeloma patients show chromosome translocations specific for the myeloma cell clone: a new type of cancer-host partnership? J Pathol 211:10–17
Vignery A (2005) Macrophage fusion: are somatic and cancer cells possible partners? Trends Cell Biol 15:188–193
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Rucci, N., Teti, A. Osteomimicry: How the Seed Grows in the Soil. Calcif Tissue Int 102, 131–140 (2018). https://doi.org/10.1007/s00223-017-0365-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-017-0365-1